Optimizing the Definition of “Genetic Caseness”
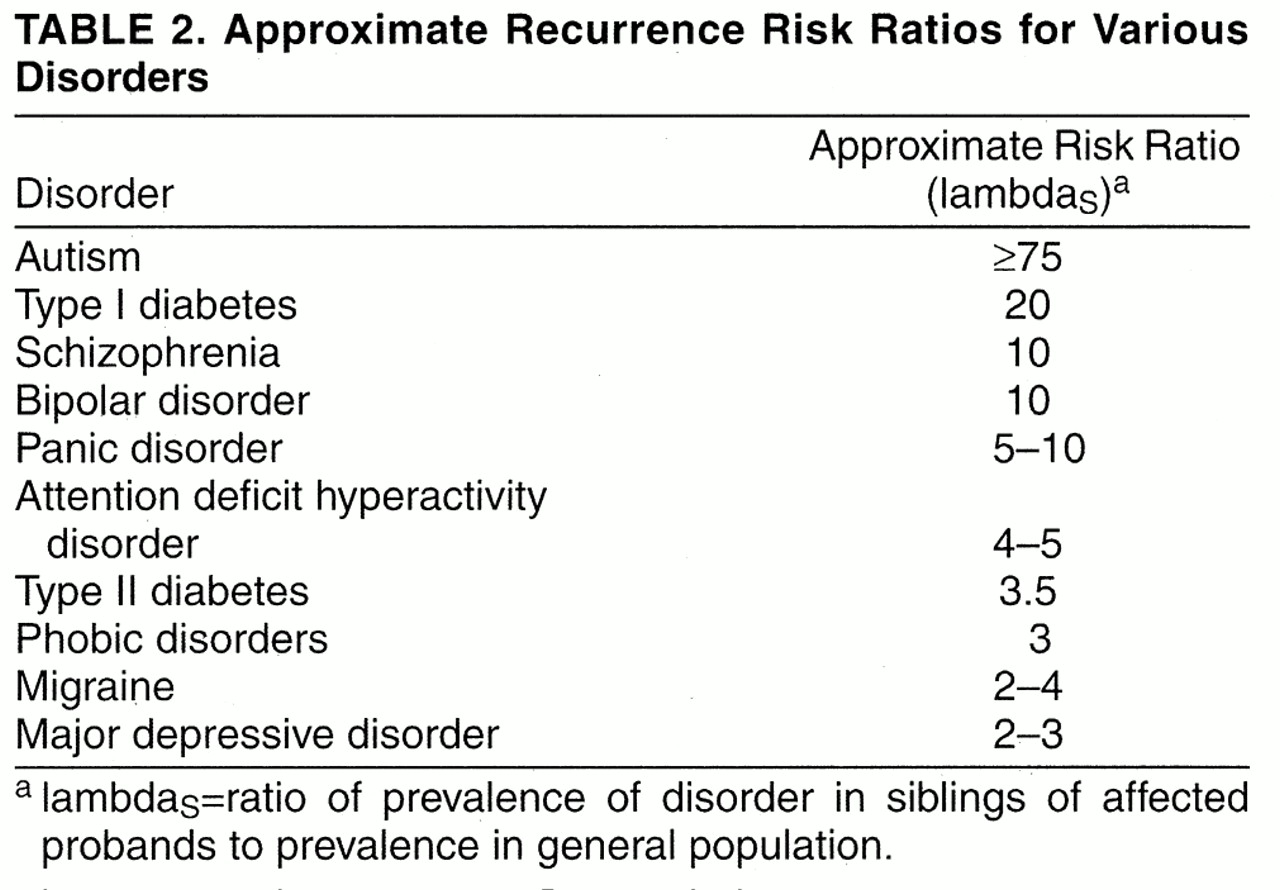
For the purposes of genetic linkage and association studies of categorical diagnoses, identifying a core phenotype that most closely reflects genetic “caseness” is essential. Defining a phenotype that is highly prevalent among relatives of affected probands and rare in the general population (i.e., maximizing lambda
R) increases the power of linkage studies
(4)(35). Among the strategies available to accomplish this, three are discussed here.
Ascertaining phenotypic extremes. In designing gene mapping studies, selecting probands with early-onset anxiety disorders may enrich the sample for genetic cases. This approach has been used to minimize genetic heterogeneity and phenocopies within a sample
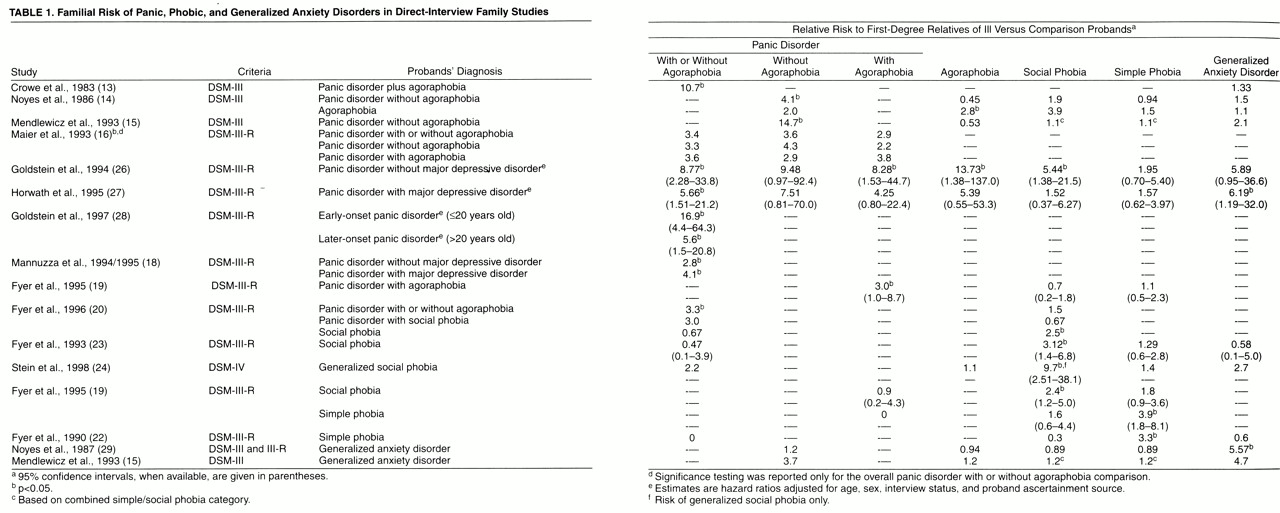
(3). A recent study from Goldstein and colleagues
(28) provided strong evidence that early-onset panic disorder is indeed associated with greater familial loading for the disorder. In a large, direct-interview family study, they found that relatives of probands who developed panic disorder before age 20 had a 17-fold higher risk of panic disorder than relatives of comparison subjects, while relatives of probands with later-onset panic disorder had a sixfold higher risk than relatives of comparison subjects. Relatives of the early-onset panic probands had higher rates of agoraphobia but did not differ from the relatives of later-onset panic probands in age at onset of panic or in symptom profiles.
Childhood anxiety disorders have been associated with an earlier age at onset of panic disorder/agoraphobia, more avoidance, more severe disorder, and more comorbidity
(75),
(77)-
(80), possibly reflecting an underlying anxiety diathesis. The data on behavioral inhibition and familial risk for anxiety disorders are also consistent with the idea that early-onset anxiety may define a highly familial form of disorder. Ascertaining probands who have unusually severe disorder may also improve the genetic homogeneity of clinical samples
(81).
A more general approach to optimizing the definition of genetic caseness involves analyzing family and twin data to determine which phenotypic features are associated with the greatest familiality and heritability. For example, on the basis of the data reviewed earlier, one might propose that expression of a heritable anxiety diathesis would include anxious temperament, comorbid panic and phobic disorders, continuing anxiety disorders from childhood to adulthood, and multiple affected family members. Although it would require an extensive database, such a hypothesis is testable. Familial recurrence risks or twin concordance rates could be calculated for this composite phenotype (or some subset of it) and compared to recurrence risks for panic or phobic disorders alone. An alternative, and perhaps more fruitful, strategy would be to analyze a range of phenotypic data without a priori hypotheses about which are the most salient. Such an approach, using logistic regression modeling, has been illustrated in studies of bipolar disorder and attention deficit hyperactivity disorder
(82),
(83). By including data on symptoms of panic and phobic disorders (e.g., panic attacks, phobias) as well as anxiety traits (e.g., neuroticism, behavioral inhibition) in such a model, one might empirically construct an improved definition of genetic caseness that incorporates both categorical and dimensional phenotypes. Of course, to do so would require a family or twin study database that comprises such a range of phenotypic data. In one small study
(84), logistic regression modeling indicated that higher levels of the temperamental construct “emotionality” were predictive of caseness among siblings of probands with depression or anxiety disorders.
Identification of elemental phenotypes. A related approach would aim at identifying phenotypic elements of anxiety disorders that are more proximally related to genetic factors than are categorical anxiety diagnoses. The features that make up a clinical diagnosis of panic disorder may combine elements that are under the control of distinct genetic loci, as was recently suggested for obsessive-compulsive disorder
(85). If so, the genetic architecture of the disorder could be much more complicated than that of the elements considered individually. Analysis of clinical data on panic disorder families may reveal distinct symptom clusters that may be under independent genetic influence. A suggestion of this was provided by Briggs and colleagues
(86), who applied principal-components analysis to data from the Cross-National Panic Study and identified two symptom groups distinguished by the presence or absence of prominent respiratory symptoms. These subgroups appeared to be biologically meaningful because the group with prominent respiratory symptoms reported more spontaneous panic attacks and responded preferentially to imipramine, while the other group suffered more situational panic attacks and responded somewhat more to alprazolam.
Trait markers and spectrum phenotypes. The use of biological trait markers that may more closely reflect brain function than do clinical diagnoses has also been recommended as a strategy to simplify the genetic analysis of complex psychiatric disorders
(4). This approach may also identify phenotypic spectra that are genetically related and associated with a higher lambda
S than the clinical diagnosis.
In the case of panic disorder, studies of the biological basis of panic attacks suggest several candidates for trait markers. In fact, panic disorder has emerged as one of the few psychiatric disorders whose symptoms can be provoked and studied under laboratory conditions. Overall, approximately 60%–75% of patients with panic disorder experience panic attacks in response to lactate infusion compared to 10%–15% of control subjects
(87). Several other agents have been reported to provoke panic attacks more frequently in panic disorder patients including carbon dioxide (CO
2), yohimbine, cholescystokinin, and caffeine
(88). The mechanism by which these agents trigger panic attacks is unknown, but treatment with antipanic medications can block provoked panic attacks, suggesting that a neurobiologic abnormality may be involved.
Laboratory provocation tests may be useful in defining biologic subtypes of panic disorder or in identifying panic susceptibility among relatives of panic disorder probands. On the basis of family history data, a link between lactate-induced panic and familial risk of panic or other anxiety disorders has been reported
(89),
(90). However, a methodologically more rigorous direct-interview family study did not support this link
(91).
Studies of CO
2 inhalation have shown more promise
(92)-
(94). Perna and colleagues
(92) found that panic disorder patients and subjects with a history of subsyndromal unexpected panic attacks had similarly elevated rates of panic in response to CO
2 inhalation compared with normal control subjects. A family history of panic disorder was reported by 30% of the panic disorder patients, 31% of subjects with a history of unexpected panic attacks, and only 2% of normal control subjects. In another study
(93), first-degree relatives of panic disorder patients had a rate of CO
2-provoked panic that was significantly lower than that of the panic disorder patients but 12-fold higher than that of normal control subjects. These studies raise the possibility that CO
2-hypersensitivity represents a trait marker for the genetic liability for panic attacks. Whether the CO
2 inhalation test has sufficient specificity to make it a useful tool in genetic studies of panic disorder remains to be established. However, the identification of biological markers of anxiety disorders may provide a crucial handle for gene mapping studies and should continue to be a major focus of research. Because false-positive misclassification can dramatically reduce the power to detect linkage, the emphasis in identifying phenotypic indicators should be on maximizing their specificity
(95).