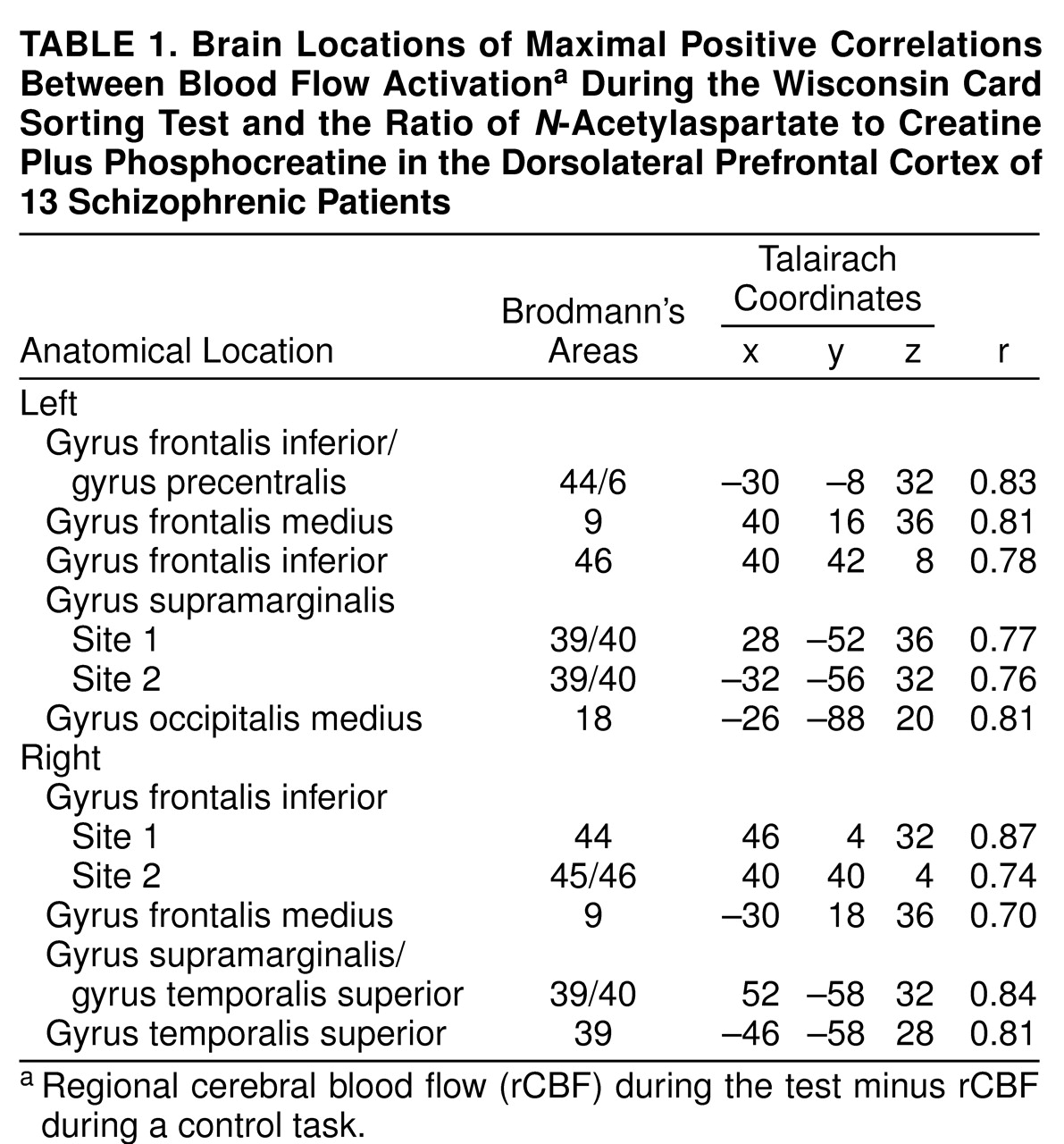
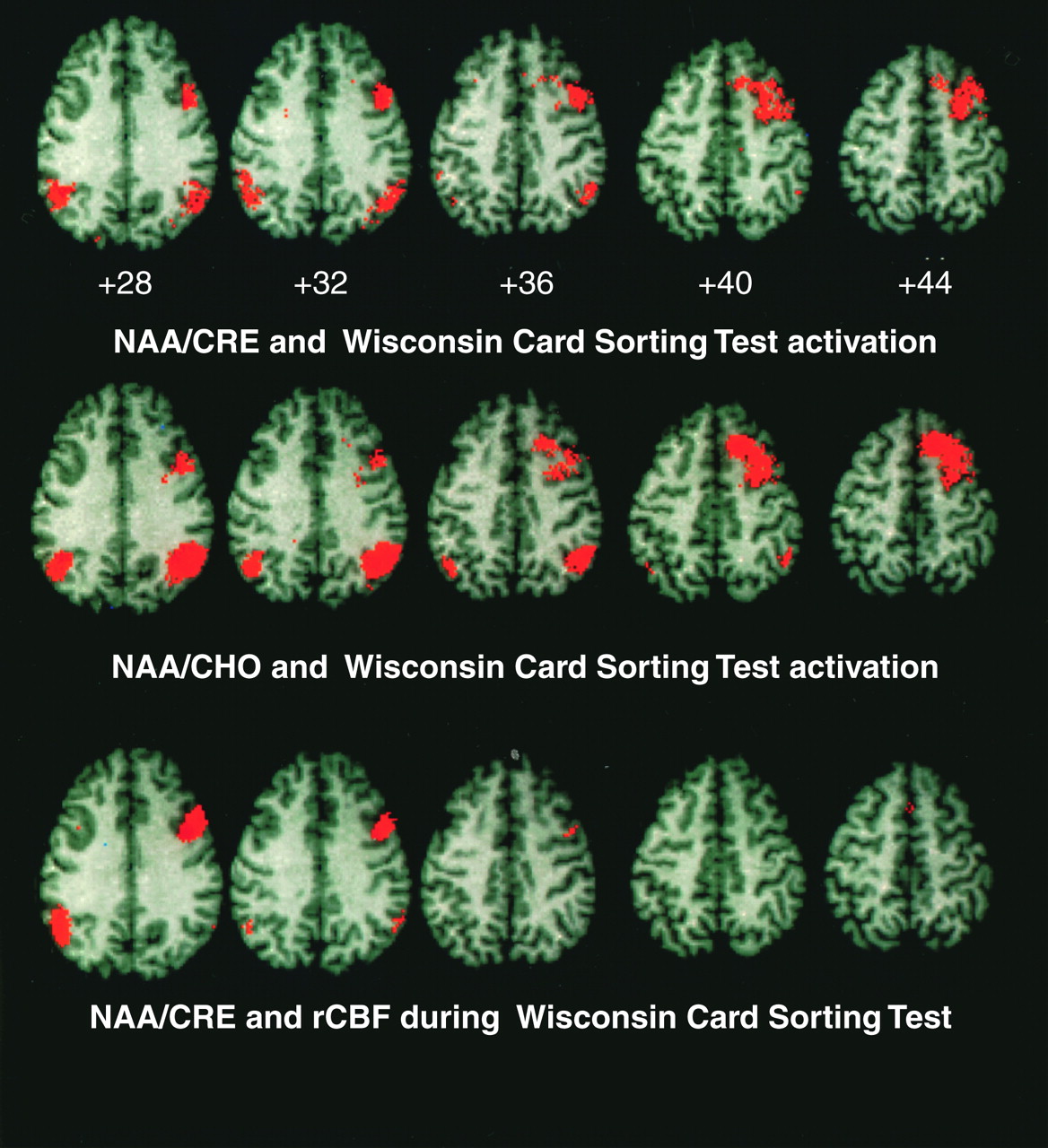
Working memory is a cognitive construct describing the ability to hold information transiently in mind in the service of comprehension, thinking, and planning
(1,
2). Complex cognitive processes such as working memory are thought to be subserved by the functional integration of interconnected regions forming large-scale cortical networks
(1–
5). Data on human and nonhuman primates show that a key cortical region for the execution of working memory tasks is the dorsolateral prefrontal cortex
(6–
12), which has reciprocal anatomical connections with the parietal, temporal, and cingulate cortices, which also participate in the cortical network related to working memory
(1–
5).
Deficits in working memory have been reported to be a cardinal feature of the pathophysiology of schizophrenia
(13,
14). Attempts to anatomically localize these deficits with functional neuroimaging studies in patients performing working memory tasks have often shown subnormal activation of the dorsolateral prefrontal cortex and, to a lesser extent, abnormalities of other regions in the working memory network
(15–
22). There has been considerable debate on issues involving the mechanism of this pattern of hypofunction, including whether it reflects distributed neuronal pathology, is referable to focal cortical pathology, or is, perhaps, an artifact of the test procedure
(15–
22). Postmortem studies of the brains of patients with schizophrenia have shown evidence of abnormalities in a number of cortical areas within the working memory network, including the dorsolateral prefrontal cortex, cingulate, and temporal cortices
(23–
26), although the most extensive data have implicated the dorsolateral prefrontal cortex
(27–
32). Since the overall function of a cortical network presumably relies on the competence of both local information processing within specific local circuits and axonal connections between local circuits and distant cortical areas, a deficit of a single region, for example the dorsolateral prefrontal cortex, could conceivably have functional reverberations throughout the working memory network. The purpose of the present study was to address directly the question of whether the integrity of a population of neurons within the dorsolateral prefrontal cortex, as studied with proton magnetic resonance spectroscopic (
1H-MRS) imaging, preferentially accounts for the distributed pattern of cortical function associated with working memory in schizophrenia, as studied with [
15O]water positron emission tomography (PET) during performance of working memory tasks.
1H-MRS imaging detects signals in multiple brain regions arising from
N-acetyl-containing moieties (mainly
N-acetylaspartate, NAA), choline-containing compounds (CHO), and creatine plus phosphocreatine (CRE)
(33). NAA is an intraneuronal amino acid, the highest concentrations of which occur in pyramidal neurons
(34). Its biological role has yet to be clearly defined. However, it acts through the glutamatergic
N-methyl-
d-aspartic acid (NMDA) receptor to elevate intracellular calcium
(35), and its concentrations are reduced by pharmacological inhibition of mitochondrial energy metabolism
(36) and by a number of pathological processes affecting the integrity of neurons
(37,
38). It is interesting that a recent study
(39) has also shown increased NAA measures in rats during experimental status epilepticus, suggesting that NAA correlates with the functional status of neurons. Relative concentrations of NAA have been previously shown to be lower than normal in the prefrontal cortex of patients with schizophrenia
(40–
44).
[
15O]Water PET identifies changes in regional cerebral blood flow (rCBF) associated with neuronal activity. In the first of two experiments we measured rCBF during performance of the Wisconsin Card Sorting Test, an abstract reasoning task involving the use of previously learned information to formulate a strategy for present and future actions. To the extent that recent memory is essential for achieving the correct action, the test has been considered to involve working memory and to be sensitive to prefrontal pathology
(10,
15,
16,
45,
46). Several earlier studies
(15–
19) have shown reduced rCBF in the dorsolateral prefrontal cortex and other related cortical areas in patients with schizophrenia during performance of the Wisconsin Card Sorting Test and other tasks involving working memory. We also performed a second, post hoc experiment to address the issue of whether the correlations found in the patients during the Wisconsin Card Sorting Test are task specific or related to generic working memory function. In this second experiment, a separate group of patients with schizophrenia performed a less complex working memory task, a version of the “N-back” task
(47). This task has been previously shown to produce activation in a cortical network including the same regions involved in the Wisconsin Card Sorting Test and to reveal similar pathophysiological characteristics in patients with schizophrenia
(22,
48). A “2-back” condition, in which subjects respond according to a number seen two stimuli before, requires continuous updating of the mental set and the use of working memory
(22).
DISCUSSION
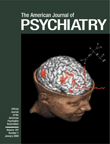
Our results show that in schizophrenia the functional integrity of neurons within the dorsolateral prefrontal cortex (as represented by
NAA measures) has predictable physiological reverberations throughout the entire working memory cortical network. NAA measures in the dorsolateral prefrontal cortex predict activation of cortical regions involved in the execution of working memory tasks, including the dorsolateral prefrontal cortex itself, the parietal cortex, and the temporal association cortex. Moreover, these relationships are regionally specific, involving only the dorsolateral prefrontal cortex as a predictor of network activation. The lack of such relationships in healthy subjects suggests that they emerge in patients because of disease-associated neuronal pathology in the dorsolateral prefrontal cortex. In the present subjects, as in our previous study groups
(41,
43),
NAA measures in the dorsolateral prefrontal cortex of patients (averaged bilateral
NAA/CRE: mean=2.6, SD=0.4) were significantly lower than those of normal comparison subjects (mean=2.9, SD=0.3) (two way ANOVA: F=4.5, df=1, 24, p<0.04; no effect of side or side-by-group interaction). To the extent that low
NAA measures are a reflection of impaired functional integrity of neurons, this putative impairment constrains in a predictable way the functional capacity of the distributed working memory network, as if these dorsolateral prefrontal cortex neurons by virtue of their projections constitute a rate-limiting factor for the degree of network recruitment
(48). These results are consistent with the anatomical and physiological centrality of the dorsolateral prefrontal cortex with respect to working memory function
(6–
12) and, perhaps, with respect to the pathophysiology of schizophrenia.
A traditional criticism of functional neuroimaging studies assessing differences in activation by working memory tasks between patients with schizophrenia and healthy subjects has been that patients usually perform worse on these tests, thus making the comparison unfair. Critics of this approach argue that it is impossible to say whether the abnormal neurobiology causes deficits in performance or vice versa. To address this criticism, we selected patients who could perform the Wisconsin Card Sorting Test well enough to be matched with comparison subjects. Indeed, previous studies
(53–
55) have shown that there is a certain percentage of patients with schizophrenia who perform well on the Wisconsin Card Sorting Test. Moreover, to further address the issue of performance and the related neurobiology, we also selected another group of patients who were not capable of performing a working memory task as well as the comparison subjects. The two cohorts of patients allowed us to assess possible correlations between
NAA in the dorsolateral prefrontal cortex and activation of the working memory network in the presence or absence of impaired performance. It was interesting that the same pattern of relationships emerged during both working memory tasks, irrespective of whether the patients’ performance was normal. This suggests that the relationships reflect the capacity of neurons in the dorsolateral prefrontal cortex to recruit the working memory network and that they are not an epiphenomenon of test score. The fact that task performance was normal in one group of patients during the Wisconsin Card Sorting Test but not in another group during the 2-back test suggests that network capacity, although constrained by the neuronal integrity of the dorsolateral prefrontal cortex, was adequate for the demands of the former condition (the Wisconsin Card Sorting Test) but not for the latter (the N-back task).
Our findings are consistent with and amplify an emerging database implicating an abnormality of prefrontal cortical connectivity in schizophrenia. While we have demonstrated this possibility at the level of functional connectivity, others have reported in vivo and postmortem changes consistent with it. Functional neuroimaging studies have suggested that dorsolateral prefrontal cortex dysfunction and connectivity may be responsible for some of the neuropsychological deficits in schizophrenia
(10,
21,
22,
48,
56,
57). Postmortem studies of the prefrontal cortex in schizophrenia have shown diminished neuropil
(27), a low number of dendritic spines on layer III pyramidal neurons
(30), small layer III neurons
(32), abnormal levels of developmental and synaptic proteins such as synaptophysin and growth-associated protein 43
(28), and selective abnormalities in gene expression for glutamate NMDA receptor subunits
(58). The evidence that neuronal connections of layer III neurons may be especially affected
(32,
33) is particularly relevant to our results as these neurons project to other cortical areas, including those recruited during working memory
(1). Consistent with our findings and with this body of literature suggesting abnormal connectivity of the dorsolateral prefrontal cortex in schizophrenia, we have recently reported that the same measure of dorsolateral prefrontal cortex neuronal integrity, i.e.,
NAA-related signals, predicts both steady-state
(59) and amphetamine-induced
(60) subcortical dopamine activity in patients with schizophrenia. Thus, a population of dorsolateral prefrontal cortex neurons identified by low
NAA signals may be critical effectors of both the cortical pathophysiology implicated in the cognitive deficits of schizophrenia and the dopamine-related phenomena implicated in treatment with antipsychotic drugs. We have also shown
(59) that monkeys with developmental prefrontal pathology induced by neonatal lesions of mesial temporal-limbic structures evince analogous relationships between prefrontal
NAA measures and subcortical steady-state and stimulus-induced release of dopamine, further indicating that development of prefrontal neurons and of their connections is a potential mechanism for the determination of these relationships.
It is obvious, however, that since our results were obtained with statistical correlations, they do not intrinsically express a relationship of causality. Therefore, even though the evidence supporting our interpretations is robust, the preceding discussion has to be viewed as conjectural. In fact, another possible interpretation of the present findings is that the
NAA measures in the dorsolateral prefrontal cortex reflect a low abundance of axon terminals from other regions, e.g., the thalamus (as
NAA is also found in neuronal processes). Indeed, in a previous study of rhesus monkeys
(61) we showed that neonatal mesial-temporal limbic lesions can induce
NAA deficits in the dorsolateral prefrontal cortex, perhaps reflecting a loss of inputs from the lesioned areas. However, by either scenario, i.e., low afferent input to the dorsolateral prefrontal cortex or low efferent activity of the dorsolateral prefrontal cortex, it is the net effect on the connectivity of dorsolateral prefrontal cortex neurons and other cortical areas that the correlations implicate.
Some further caution in the interpretation of the results of the present study should be considered. The presence of a statistical correlation in one group but not in another could be caused by greater variance in the former than in the latter. However, this was not the case in our two groups of subjects in the Wisconsin Card Sorting Test experiment, who did not have significant variance differences in either the activation or NAA data (analyzed with Hartley F-max, Cochran C, and Bartlett chi-square tests). Moreover, it is conceivable that the correlation between NAA in the dorsolateral prefrontal cortex and activation in the distributed working memory cortical network in the patients could simply be an epiphenomenon of the fact that activation in all the other regions of the network has a high degree of covariance with activation in the dorsolateral prefrontal cortex. However, if this was the case, the same correlations between NAA in the dorsolateral prefrontal cortex and activation in the cortical network would have been evident also in the comparison group, where we found a similar degree of high covariance between activation in the dorsolateral prefrontal cortex and the other regions of the network (data not shown). Since this was not the case, we can assume that the correlations in the patients are not such an epiphenomenon. Another line of evidence against the correlations being an epiphenomenon of the high degree of covariance of the activation of all regions in the working memory cortical network is the specificity of correlations to NAA measures in the dorsolateral prefrontal cortex. In fact, if the correlations were an epiphenomenon of intracortical rCBF relationships, it would be expected that NAA in other cortical regions of the network would show similar relationships with activation in the entire working memory cortical network. However, this also was not the case, since NAA measures in the superior temporal gyrus and anterior cingulate did not show correlations with activation of the working memory cortical network at all.
In conclusion, the data of the present study show potentially unique relationships between pathology of dorsolateral prefrontal cortical neurons and physiological activation of the whole working memory network in patients with schizophrenia. These data are consistent with current speculation focusing on the role played by development of the dorsolateral prefrontal cortex and its connections in the pathophysiology of schizophrenia
(31,
62).