Schizophrenia can appear during childhood with psychiatric symptom profiles and neurobiological abnormalities similar to those found in adults
(20–
25). The few available early studies of “schizophrenic children” found neurologic dysfunction in up to 95% of patients
(26–
29) but lacked clear diagnostic criteria, often including children with pervasive developmental disorders, organic brain syndrome, and mental retardation.
In this study, a comprehensive neurologic examination was performed on adolescents with early-onset schizophrenia and an age- and sex-matched healthy comparison group. We proposed that neurologic dysfunction would be more frequent in the patients with schizophrenia than in the healthy comparison adolescents and would be similar in kind to that found in adults with schizophrenia.
Method
The study was approved by the institutional review board of the National Institute for Mental Health. After receiving a full explanation of study procedures, parents or guardians provided written consent; subjects gave their assent to the examination. Psychiatric diagnosis was determined by a review of records, interviews, and psychiatric examinations. Full-scale IQ was estimated from the vocabulary and block design subtests of the WISC-R
(30). Brain structural analysis was performed as previously reported
(31–
34). A standardized neurologic examination was also performed. Individual test items were scored as normal or abnormal, present or absent, or untestable. Patients were examined after at least 1 week with no medication. It was not possible to blind the examiners to patient diagnosis because of active psychotic symptoms in the patients.
We studied 21 adolescents aged 12–17 who met DSM-III-R criteria for schizophrenia with an onset of psychosis before age 13. Exclusionary criteria included a premorbid WISC-R full-scale IQ <70, other neurological or significant medical disorders, active substance abuse, or significant hearing or visual deficits. The subjects were participants in a larger study of refractory childhood-onset schizophrenia
(22,
31,
35–
37). Additionally, 27 psychiatrically healthy adolescents aged 10–17 who met the same exclusionary criteria served as a comparison group.
Means, standard deviations, and ranges are reported. Age was compared by means of a two-tailed t test. Otherwise, cohorts were compared by means of the Wilcoxon ranked sum test, with p<0.05 considered statistically significant. Chi-squares were used to compare frequency of occurrence. Spearman’s correlation coefficients were calculated to detect significant correlations among age, IQ, total brain volume and ventricular volume with individual neurological signs, grouped subcategories of neurologic function, and total number of signs. Correlations were compared by means of Fisher’s r-to-z transformation. Interrater agreement for eight subjects was measured with the kappa statistic and with percent agreement for low-frequency signs.
Results
The cohort with childhood-onset schizophrenia was made up of eight girls and 13 boys with a mean age of 14 years (SD=1). The healthy comparison group was composed of 12 girls and 15 boys with a mean age of 13 years (SD=2). Neither age (t=1.56, N=21, 27, p<0.13) nor sex distribution (χ2=1.96, df=1, p<0.67) differed between the two groups. After onset of symptoms, the subjects with childhood-onset schizophrenia had an estimated mean full-scale IQ of 77 (SD=22, range=48–120), which differed significantly from that of the healthy comparison subjects (estimated mean full-scale IQ=121, SD=12, range=97–141) (Wilcoxon z=4.21, N=48, p<0.0001). Interrater reliability was good; kappa was >0.40, and percent agreement was >75% on all items, except four frontal release signs (snout, palmomental, glabellar tap, and jaw jerk), in which percent agreement ranged from 43% to 57%. Isolated abnormalities of cranial nerves, primary sensation, and strength occurred equally in both groups. Gaze impersistence was more common in the subjects with childhood-onset schizophrenia (χ2=14.15, df=1, p<0.001). Similarly, sensory integration face-hand test results were more frequently abnormal in the patients with childhood-onset schizophrenia (χ2=8.44, df=1, p<0.004). A higher incidence of increased passive tone while reinforcing movements were made with the opposite arm was especially common in healthy comparison adolescents younger than age 15, whereas the adolescents with childhood-onset schizophrenia were significantly more likely to exhibit increased passive tone in the absence of reinforcement, independent of age (χ2=7.31, df=1, p<0.03). Corticospinal tract signs were also significantly more common in the cohort with childhood-onset schizophrenia (χ2=7.31, df=1, p<0.03). Coordination was often abnormal in both groups; however, all of the healthy comparison adolescents with poor coordination were younger than age 15 (p<0.04, Fisher’s exact test), whereas no grouping by age was present in the cohort with childhood-onset schizophrenia (p>0.99, Fisher’s exact test). Choreiform movements were similarly significantly clustered in the younger healthy comparison adolescents (p<0.02, Fisher’s exact test) but were equally present in both younger and older adolescents with childhood-onset schizophrenia (p>0.99, Fisher’s exact test).
Twelve healthy comparison adolescents but only three adolescents with childhood-onset schizophrenia exhibited no primitive reflexes (χ2=0.03, df=1, p<0.056). The number of primitive reflexes was significantly inversely correlated with age in the healthy comparison cohort (rs=–0.57, N=27, p<0.002) but not in the cohort with childhood-onset schizophrenia (rs=0.05, N=21, p<0.84).
At least one sign was present at neurologic examination in all (100%) of the adolescents with childhood-onset schizophrenia and in 26 (96%) of the 27 healthy comparison subjects. The group with schizophrenia had a mean of 6 (SD=2, range=2–10) signs per subject. The healthy comparison group had a mean of 4 (SD=2, range=0–8) signs per subject (Wilcoxon z=–3.30, N=18, 27, p<0.001). Because examination of primitive reflexes can have low interrater reliability
(38), analyses were repeated with those signs excluded. Significant differences persisted.
No significant correlations were found between either the total number or type of abnormalities by specific neurologic subcategory and either ventricular size or total brain volume. Full-scale IQ correlated only inversely with the number of primitive reflexes in the healthy comparison cohort (rs=–0.64, N=27, p<0.02) but not in the patients.
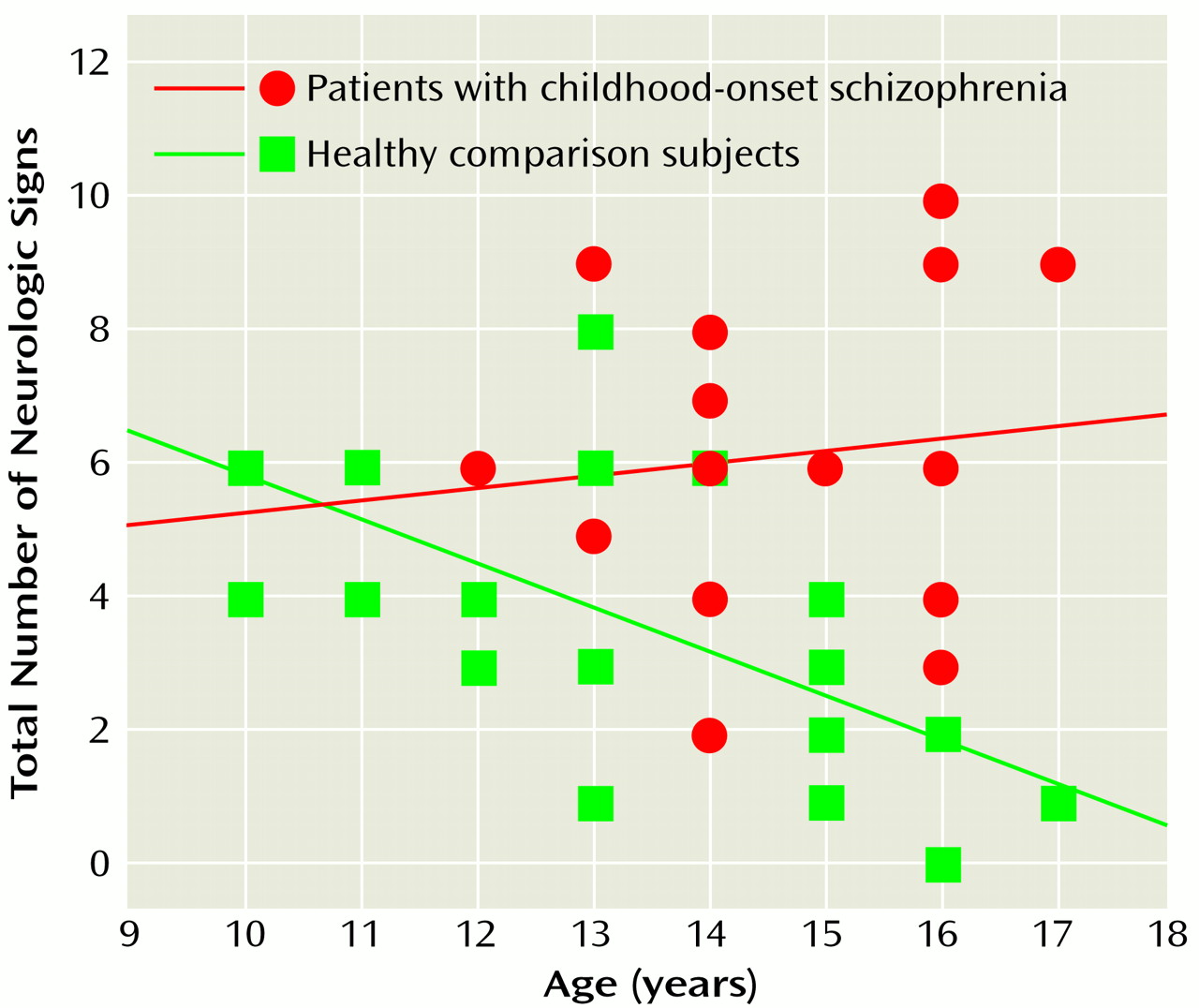
The total number of neurologic signs decreased with age in the healthy comparison group (r
s=–0.73, N=27, p<0.0001) but not in the group with childhood-onset schizophrenia (r
s=–0.07, N=18, p<0.78). These correlations differed significantly (Wilcoxon z=–2.56, p=0.005, Fisher’s r-to-z transformation) (
Figure 1).
Discussion
This study uniquely demonstrates a failure of neurological maturation in patients with early-onset schizophrenia during adolescence. Not only were the patients with childhood-onset schizophrenia more neurologically impaired than the healthy subjects (with a pattern of abnormalities similar to that seen in patients with adult-onset schizophrenia), but they also failed to lose the signs that fade during normal development.
The high frequency of neurologic signs seen in the healthy subjects likely reflected the inclusive nature of the examination and the subjects’ ages. Our results are comparable with those of other studies of healthy subjects. Rossi et al.
(39) found neurologic signs in 95% of normal adults on a 28-item battery of soft signs, with a mean of 4.1 signs per subject. Kennard found neurologic abnormalities in 60% of the healthy children he studied
(28); he and Rutter et al. found chorea in 5%–25% of healthy subjects
(40).
The frequency of neurologic signs found in our subjects with childhood-onset schizophrenia is also comparable to the 92%–100% prevalence reported in other series of both adult
(9,
10,
13,
14) and pediatric
(28) patients with schizophrenia. Our high prevalence of abnormal signs may additionally reflect the fact that our cohort with schizophrenia was severely ill and treatment-resistant. Our high frequency is also consistent with theories that pediatric schizophrenia results from an early cerebral insult and is a more “biologically driven” form of the disease than later-onset schizophrenia
(41,
42).
Gittelman and Birch
(27) found neurologic dysfunction more common in children with schizophrenia with lower IQs than in those with higher IQs. Although the estimated full-scale IQ in our subjects with childhood-onset schizophrenia was significantly lower than that of the healthy comparison group, the only correlation found was an inverse relation with the number of primitive reflexes in the healthy comparison group. It is likely that the lower IQ and higher number of neurologic signs found in the subjects with childhood-onset schizophrenia are both reflections of the underlying illness. It would be difficult to match the individuals in each group by IQ and still consider the healthy comparison cohort neurologically normal.
All patients with childhood-onset schizophrenia were medication-free for at least 1 week before the neurological examination; however, we cannot exclude the possibility that the presence of some signs was medication-related. Although tardive dyskinesia may be less frequent in children than in adults
(43,
44), withdrawal dyskinesias are especially common in children during the first 6 weeks after neuroleptic discontinuation
(45–
48).
Primitive reflexes, present during early life, are suppressed as cortical areas mature
(49). Their reappearance may represent a loss of hemispheric inhibition of the brainstem. Our examination detected a maturational loss of primitive reflexes in the healthy comparison group that was lacking in the cohort with schizophrenia. Similarly, although hypertonia, impaired coordination, chorea, and the total number of neurologic signs decreased with age in the healthy comparison cohort, they persisted in the adolescents with schizophrenia, indicating a failure of normal maturation. We have previously detected other deviations from normal development in subjects with childhood-onset schizophrenia during adolescence, including a progressive loss of cerebral and subcortical volume
(31,
32) and a decline in full-scale IQ due to a lack of new learning
(22,
50). The present study provides additional evidence of an abnormal neurodevelopmental course in patients with early-onset schizophrenia during adolescence.