While the specific mechanisms of antidepressant treatment are not known, serotonergic neurons may be important because they are targeted by selective serotonin reuptake inhibitors (SSRIs). In animal models, long-term SSRI administration raises intrasynaptic serotonin (5-HT) levels in the cortex
(1). This could affect different types of 5-HT receptors, but there are several reasons why 5-HT
2A receptors could contribute to SSRI antidepressant effects. First, 5-HT
2A receptors have the potential to activate cAMP-response element-binding protein and/or brain-derived neurotrophic factor through Ca
2+-dependent protein kinases in the cortex
(2,
3). It is possible that elevated 5-HT concentrations after SSRI treatment could elevate mood by means of 5-HT
2A receptors because drugs with 5-HT
2A agonist properties may have euphoriant effects
(4–
8). The euphoria can occur after administration of low doses of 2,5-dimethoxy-4-methyl-amphetamine when hallucinations are not present
(5,
7,
8).
While in theory some SSRI effects could be mediated by 5-HT
2A receptors, it is not known whether SSRIs at clinical doses have any effect at all on 5-HT
2A receptors in humans. Preclinical data with long-term SSRI treatment are not consistent, although there is a tendency toward up-regulation or no change at lower doses
(9–
11) and down-regulation at higher doses
(12,
13). Rats have much faster drug metabolism than humans, and it may be that intraperitoneal dosing of SSRIs in rats differs from oral dosing in humans. We are aware of one published study of humans. Using [
18F]setoperone positron emission tomography (PET), Massou et al.
(14) found that a group of six depressed patients responding to SSRI treatment had higher 5-HT
2A receptor binding potential than a separate group of eight depressed patients who were untreated. This study did not use an intrasubject design, and five of the six treated patients were also taking benzodiazepines. It is reported that benzodiazepines lower serotonin release in animals, and this presents a potential confound
(15–
17).
To our knowledge, the relationship between 5-HT
2A receptors during depression and subsequent response to treatment has not been investigated. Postmortem methods cannot be used, and we know of no imaging study of 5-HT
2A receptors that has examined this question. Platelet 5-HT
2A receptor B
max (receptor density) is another in vivo measurement. Platelet and frontal cortex 5-HT
2A receptors have identical structures; hence, platelet measures may be used as a peripheral index of cortex 5-HT
2A receptors
(18). One such study
(19) showed no association between platelet 5-HT
2A B
max before treatment and the eventual response to SSRIs.
[
18F]Setoperone has several properties favorable for PET imaging of 5-HT
2A receptors in the human cortex
(20). Setoperone is an antagonist with high affinity and specificity for 5-HT
2A receptors in the cortex
(6). The affinity of setoperone is 100 times as great for 5-HT
2A receptors as for 5-HT
2C receptors
(6), and cortex 5-HT
2C receptor density is relatively low
(21,
22). The affinity of setoperone for 5-HT
2A receptors is 10 to 50 times as great as its affinity for D
2 receptors, but D
2 receptors have a low density in the cortex
(23). Both 5-HT
2A,C and D
2 antagonists displace [
18F]setoperone in the striatum, but only 5-HT
2A,C antagonists displace [
18F]setoperone in the cortex; thus, specific binding of [
18F]setoperone in the cortex reflects binding to 5-HT
2A receptors. 5-HT
2A antagonists do not displace [
18F]setoperone in the cerebellum, and the cerebellum is almost devoid of 5-HT
2A receptors, hence it may be used as a reference region
(24,
25). Metabolites of [
18F]setoperone do not show significant brain uptake
(26). Other properties have been previously reviewed
(27).
Results
Nineteen patients (12 male, seven female; mean age=30.8 years, SD=6.1) were scanned before and after treatment. The mean baseline score on the Hamilton Depression Rating Scale of these patients was 21.8 (SD=3.8). All patients scanned before and after treatment had detectable serum paroxetine levels and were included in the analysis (mean=30 μg/liter, SD=24).
Nineteen age-matched healthy subjects were recruited (eight male, 11 female; mean age=31.8, SD=6.9); 18 were matched within 1 year of age, and one was matched within 2 years. There was no significant difference in gender between groups (Fisher’s exact test, p=0.33).
In the following sections we will first report results of our reassessment, in a larger study group, of previous findings: the age-associated decline in 5-HT
2A binding potential
(27,
43–
45) and the lack of diagnosis effect on 5-HT
2A binding potential
(27). Then the primary question of a treatment effect will be addressed by examining the effect of treatment on cortical 5-HT
2A binding potential and the effect of the interaction between age and treatment on cortical 5-HT
2A binding potential. Finally, we will examine whether initial cortical 5-HT
2A binding potential has any association with eventual treatment response.
Effects of Age
Since age has been previously associated with a decline in both 5-HT
2A B
max and 5-HT
2A binding potential
(27,
43–
45), we assessed the effect of age on 5-HT
2A binding potential. In the healthy subjects, age was a significant covariate globally in the cortex in both type of analyses. In the voxel-wise analysis, analysis of covariance (ANCOVA) at each voxel (86,821 voxels) yielded corrected probability levels of p<0.001 (corrected peak) and p<0.001 (corrected cluster). Similar levels resulted from the ANCOVAs for the regions of interest (F=19–30, df=1, 17, p<0.001).
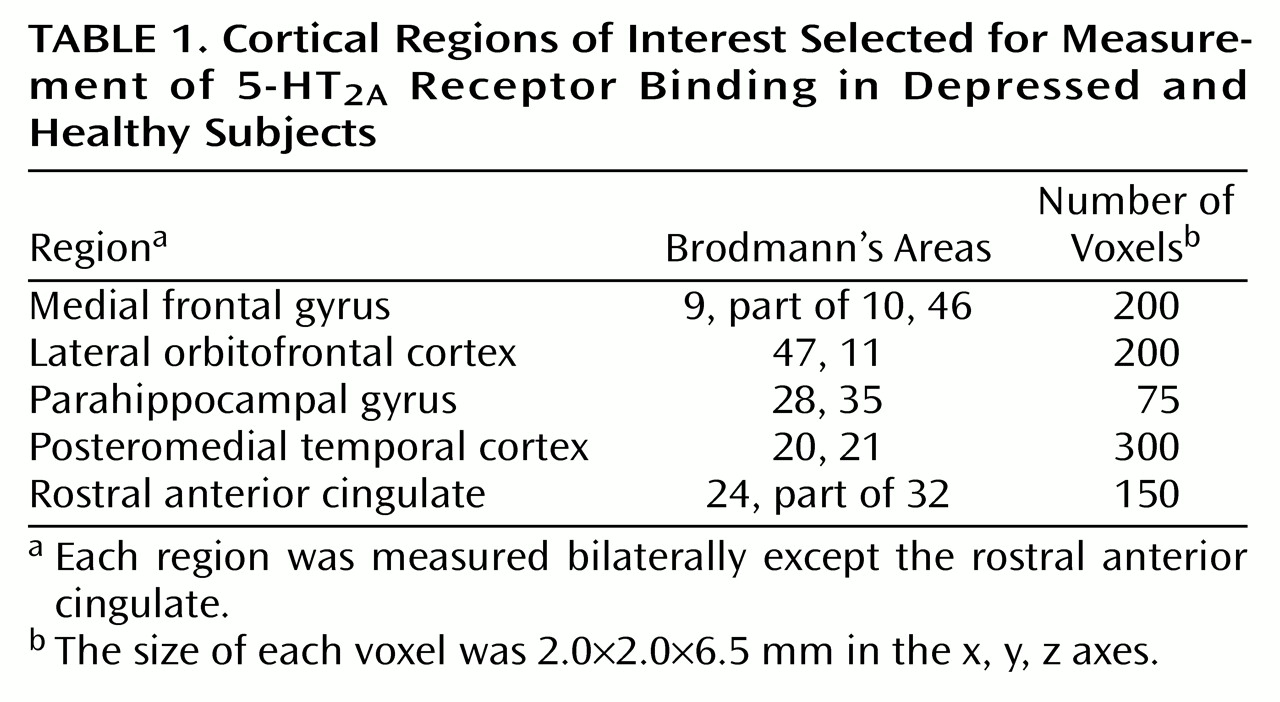
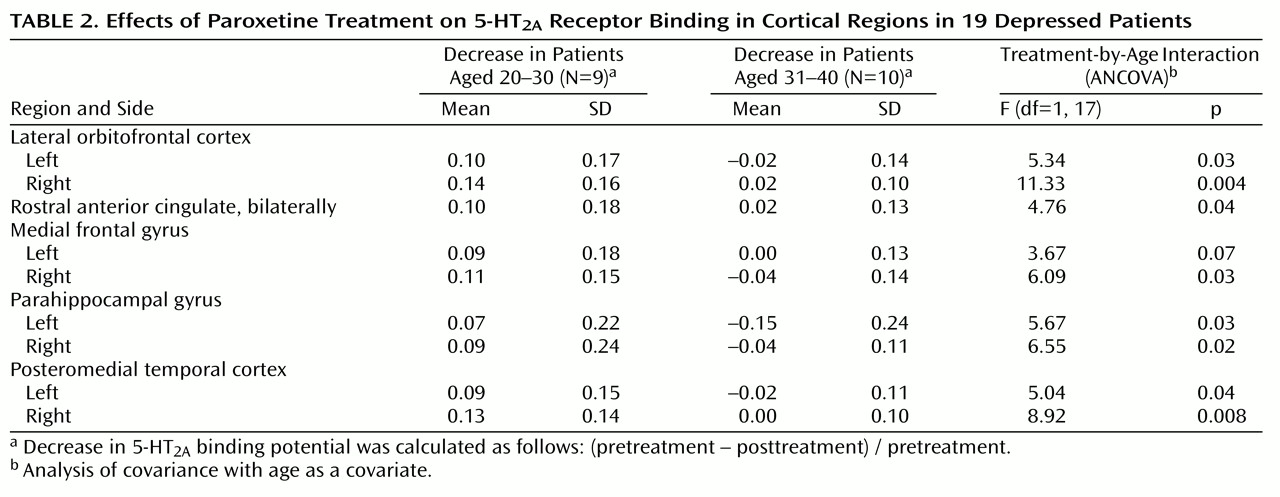
Within the pretreatment scans, all cortical regions showed an association with age (
Figure 1); for the ANCOVAs at each voxel (80,688 voxels), p<0.001 (corrected peak) and p<0.001 (corrected cluster). Within the posttreatment scans, all cortical regions also showed an association with age; for the ANCOVAs at each voxel (80,477 voxels), p<0.001 (corrected peak) and p<0.001 (corrected cluster). Then the relationship between age and 5-HT
2A binding potential was examined in each region within the pretreatment scans and then within the posttreatment scans. Every region showed a significant association with age (ANCOVA, F=15–30, df=1, 17, p<0.001).
Effects of Diagnosis
While the main aim of this study was to examine the effects of paroxetine treatment in depression, the treatment effect is better appreciated in the context of how 5-HT2A binding potential may differ between depressed and healthy subjects. The scans of the patients before treatment were compared to the scans of the healthy subjects by using ANCOVA with age as a covariate; a similar comparison was conducted with the patients’ posttreatment scans. With the voxel-by-voxel approach, no differences in 5-HT2A binding potential between the healthy subjects and depressed patients (pretreatment or posttreatment scans) were found. Similarly, no differences in 5-HT2A binding potential were found between the depressed and healthy subjects in the region-of-interest analyses. Furthermore, differences in 5-HT2A binding potential between the two groups did not interact with age in the voxel-wise or region-of-interest analyses.
Effects of Treatment
5-HT2A binding potential within a large region composed of bilateral occipital and medial temporal cortex decreased with treatment, but this was not significant when the cluster threshold was applied; for the repeated measures ANCOVA with age as a covariate (3,377 voxels), p=0.48 (corrected peak) and p=0.20 (corrected cluster). The results of region-of-interest analyses of 5-HT2A binding potential using repeated measures (within subject) ANCOVA were not significant. Having a full response (N=10) versus having a partial response or nonresponse (N=9) had no association with change in 5-HT2A binding potential with treatment in voxel-wise or regional analyses. Using the region-of-interest data, we examined a plot of the residual values versus age, and an obvious decrease with treatment was present in younger individuals.
Interaction Between Treatment Effect and Age
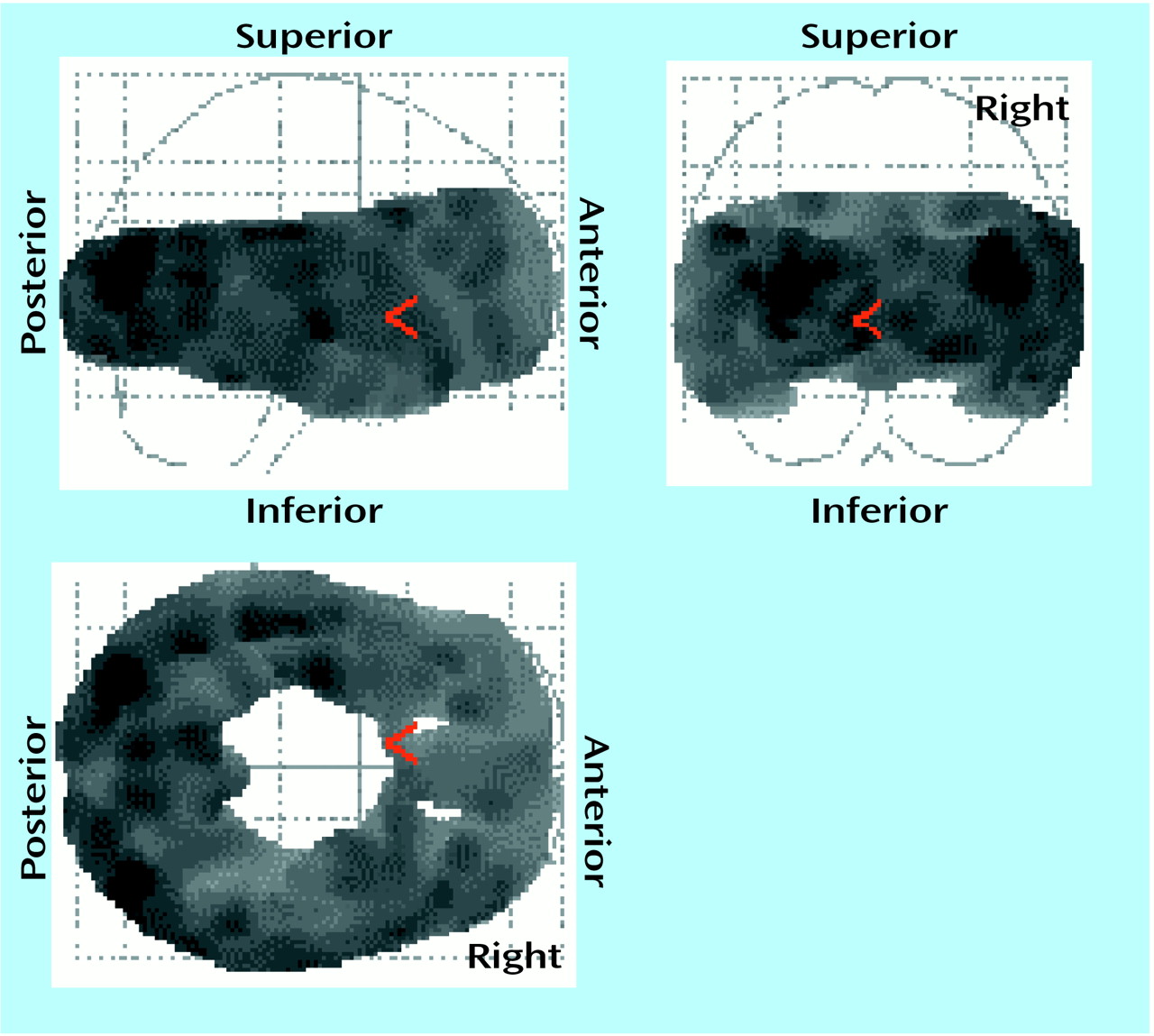
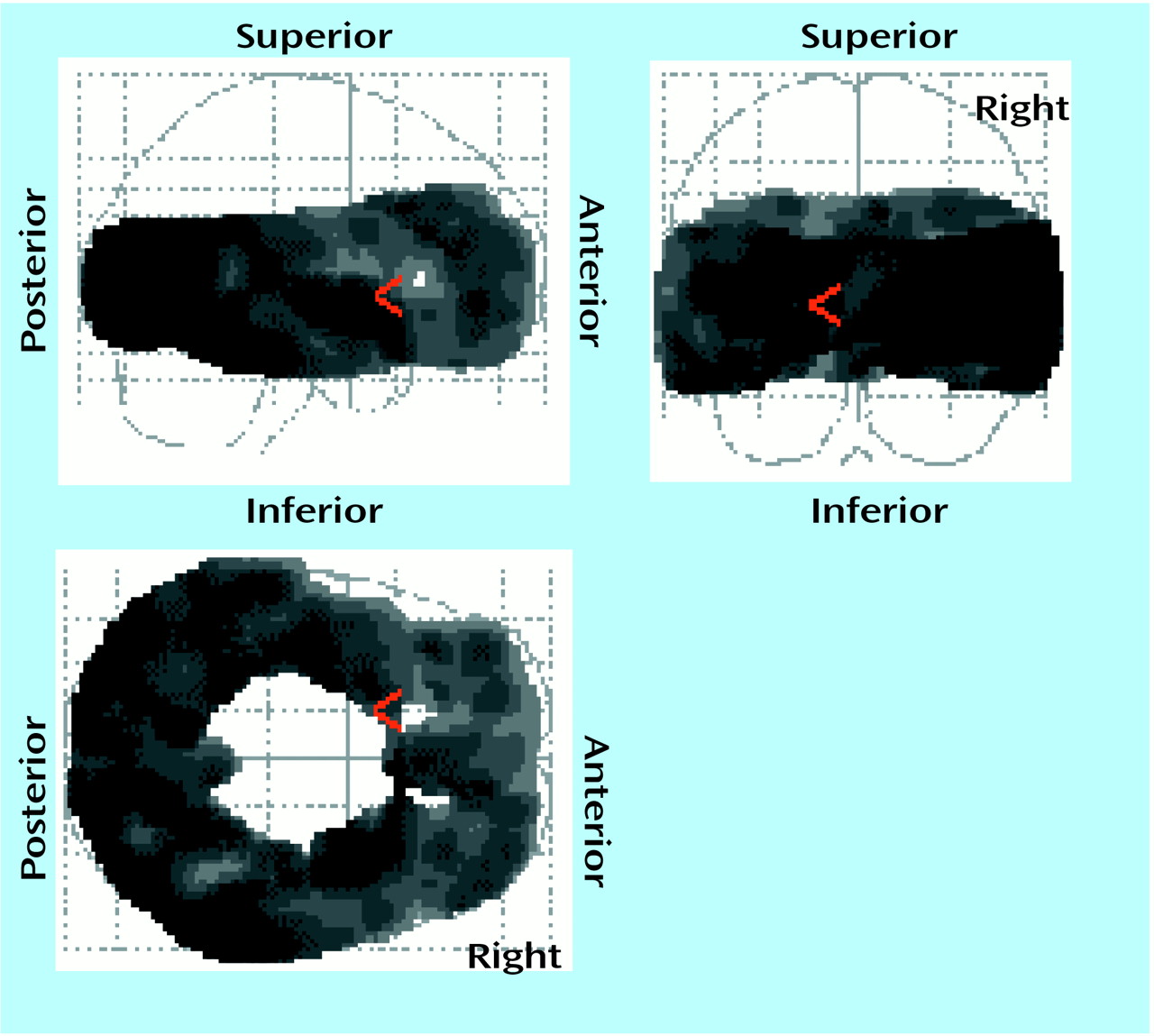
The effect of the interaction between treatment and age on 5-HT
2A binding potential was found globally in the voxel-by-voxel analysis; for the repeated measures ANCOVA of the treatment-by-age interaction (56,928 voxels), p<0.05 (corrected peak) and p<0.001 (corrected cluster) (
Figure 2). The decrease in 5-HT
2A binding potential after treatment became less pronounced as age increased. This finding was also examined with the region-of-interest approach. Using multivariate repeated measures analysis of covariance (MANCOVA), we found the effect of treatment, the treatment-by-age interaction, and the effect of region were significant with respect to 5-HT
2A binding potential (treatment: F=7.71, df=1, 17, p=0.01; treatment by age: F=6.33, df=1, 17, p=0.02; region: F=2.92, df=8, 10, p=0.05). The multiple variables refer to the different regions, and the repeated measure is the within-subject value before and after treatment. Repeated measures ANCOVA with age as a covariate for each region showed that the treatment-by-age interaction was significant for almost every region (
Table 2). The within-subject repeated measure is the region-of-interest 5-HT
2A binding potential. In patients aged 20 to 30 years, the mean decrease was 10% across all regions, whereas in patients aged 30 to 40 the mean change was 0%. For the region-of-interest analysis, there were no outliers, as the Cook’s distances were less than 1 for all observations. Pearson correlation indicated that there was no correlation between age and serum paroxetine level (r=–0.02, N=19, p=0.93).
Relation of Treatment Response to Pretreatment 5-HT2A Binding Potential
The depressed patients were classified according to treatment response, resulting in 10 treatment responders, six partial responders, and three nonresponders. No differences in 5-HT2A binding potential were found between the responders and the remaining patients (partial responders and nonresponders) in either the voxel-based or regional analyses using ANCOVA to examine the effect of being an eventual responder, with age as a covariate. No interaction between age and difference in 5-HT2A binding potential between groups was present. The age of the treatment responders matched that of the non- and partial responders within 2 years, with the exception of an additional 21-year-old subject in the treatment-responding group.
5-HT2A binding potential in the 10 treatment-responding subjects was compared to 5-HT2A binding potential in the age-matched healthy subjects. The voxel-wise analysis showed globally higher 5-HT2A binding potential in the treatment-responding patients; for the ANCOVA of the diagnosis effect in the responders (44,852 voxels), p<0.18 (corrected peak) and p<0.001 (corrected cluster). A region-of-interest MANCOVA showed somewhat greater 5-HT2A binding potential in the treatment-responding patients than in the healthy subjects (diagnosis: F=2.62, df=9, 9, p=0.09; age: F=5.63, df=9, 9, p=0.009). The multiple variables in the MANCOVA were the different regions. Within individual regions, there was no significant difference between the treatment-responding and healthy subjects.
5-HT2A binding potential in the remaining nine subjects—the partial responders and nonresponding patients—was compared to that in a different set of nine age-matched healthy subjects. Using ANCOVA with voxel-based analyses and MANCOVA and ANCOVA for the region-of-interest data, we found no differences between these two groups (and no age interaction was present).
Discussion
To our knowledge, this is the first report of 5-HT2A receptor changes during SSRI treatment shown by a within-subject design in previously drug-free depressed patients. In patients under age 30, we found decreases of 5-HT2A binding potential in all brain regions; however, these decreases attenuated with increasing age.
There are two alternative explanations for the decrease in 5-HT
2A binding potential with paroxetine treatment. One is that paroxetine increased 5-HT secondary to 5-HT transporter blockade
(1) and that 5-HT
2A down-regulation occurred. The second is that paroxetine directly antagonized 5-HT
2A receptors and decreased the detected 5-HT
2A receptor binding potential. This latter possibility can be ruled out. Using [
18F]setoperone PET, we previously demonstrated no change in 5-HT
2A receptor binding potential after single-dose paroxetine administration, thus ruling out a short-term antagonist effect in vivo
(46). Moreover, the affinity of paroxetine for 5-HT
2A receptors is extremely low, being in the nonspecific range
(6). On the other hand, the first explanation is plausible since long-term SSRI exposure is associated with increased synaptic 5-HT
(1).
The binding potential reflects B
max/K
d, and hence the down-regulation observed could be attributed to either decreased B
max (decreased receptor density) or increased K
d (decreased affinity). In ex vivo studies, changes in 5-HT
2A receptors after long-term agonist administration are characterized mostly by decreased 5-HT
2A B
max (47). Thus, it is likely that decreased 5-HT
2A B
max would account for the majority of the change in 5-HT
2A binding potential found in the current study.
The decrease in 5-HT
2A binding potential after treatment became less so with age. It could be that down-regulation of 5-HT
2A receptors decreases with age. This notion is consistent with the well-established finding that 5-HT
2A receptor density decreases with age
(27,
43–
45), since a floor effect would limit down-regulation with age. To our knowledge, the relationship between age and down-regulation of 5-HT
2A receptors has not been investigated in animal models, but some aspects of 5-HT
2A receptor regulation appear to change with age. One effector pathway of 5-HT
2A receptors includes activation of phospholipase C, production of inositol triphosphate, increased intracellular calcium, and activation of protein kinase C
(48,
49). Activation of protein kinase C influences stability of messenger RNA for 5-HT
2A receptors, and the activation of calcium-dependent protein kinase C has been reported to change with age in rodent brains
(50).
Our finding contrasts with that of Massou et al.
(14), who reported higher 5-HT
2A receptor binding potential in six treated depressed patients than in eight untreated depressed patients. There are several methodological differences between this study and the current one that could explain the discrepancy. The earlier study did not use an intrasubject design, the dose and type of SSRI varied, concomitant benzodiazepines were used, and the timing of the [
18F]setoperone scan varied among subjects. Benzodiazepines may influence 5-HT release
(15–
17). The timing of the second scan relative to the onset of treatment could influence results because preclinical models of SSRI administration demonstrate that both the firing rate of cortical 5-HT neurons and the concentration of 5-HT may fluctuate markedly over the course of treatment
(1,
51,
52).
It has been reported that 5-HT
2A binding potential declines after treatment with desipramine
(53). The authors of that study proposed that decreases in 5-HT
2A binding potential might contribute to a mechanism of therapeutic effect for desipramine. With SSRI treatment it seems unlikely that down-regulation itself is directly therapeutic because in the current study there was no association between down-regulation and treatment response. On the other hand, the power to find this association may be very low for several reasons. The true difference could be small. Placebo responders add variance. The degree of down-regulation could vary among individuals of similar age because of genetic differences, adding more variance. 5-HT agonism on multiple receptors rather than one receptor may be important for therapeutic effect; thus, down-regulation of a single receptor subtype may not associate strongly with treatment response.
Even if down-regulation itself is not a therapeutic effect of SSRIs, this does not rule out the interesting possibility that increased 5-HT agonism on 5-HT2A receptors is therapeutic. While down-regulation of 5-HT2A receptors may occur in the presence of increased 5-HT, down-regulation itself can reduce the agonist effect of 5-HT on 5-HT2A receptors.
The decrease in 5-HT
2A binding potential with treatment in younger subjects was a global cortical change. Given that 5-HT transporter sites are widespread throughout the cortex, it is not surprising that paroxetine would have global effects on 5-HT
2A receptors
(54).
We found no difference between depressed and healthy subjects in any cortical region. This is consistent with findings from studies of medication-free depressed subjects
(27,
45).
It is likely that 5-HT reuptake inhibition resulted in down-regulation of the 5-HT
2A binding potential. In theory, indirect mechanisms, such as occupancy by endogenous 5-HT, could also contribute to a decrease in 5-HT
2A receptor binding potential. The concentration of endogenous 5-HT in the human cortex is about 0.5 to 1.5 nM
(55), and the K
I of 5-HT for cortical 5-HT
2A receptors is 300 to 5000 nM
(6) (K
I is the concentration at which one-half of the receptor sites are displaced by a drug or chemical and is inversely proportional to the affinity of the drug or chemical). If these cortical 5-HT concentrations reflect synaptic concentrations, about 1% of synaptic 5-HT
2A receptors could be occupied by 5-HT at baseline. This is consistent with a finding in our previous study
(46), in which we found that single-dose paroxetine had no effect on the setoperone binding potential of healthy subjects.
Several methodological limitations are present in the study. The resolution of the PET camera does not allow one to distinguish the cell type or cellular location attributable to the change in 5-HT
2A receptors. Candidate cell types likely to account for a large proportion of 5-HT
2A receptors would include interneurons as well as pyramidal cell neurons
(56). Another limitation is that the binding potential reflects B
max/K
d, and we were unable to discern between these two measures. However, down-regulation processes are the predominant responses to agonist stimulation in ex vivo models
(47). With respect to the paroxetine treatment, we took many precautions to enhance compliance, such as informing patients that their serum levels were to be assayed at the time of the second PET scan as part of the study. We also took measurements and detected paroxetine levels in every patient. Even so, it is possible that some patients may have temporarily interrupted their treatment earlier, and this would not be detected in our protocol.
In conclusion, we found a down-regulation of 5-HT2A receptor binding potential after paroxetine treatment in younger depressed patients. This suggests that long-term paroxetine treatment increases 5-HT agonism on 5-HT2A receptors in the cortex of young patients with depression.