Diseases that affect the basal ganglia, such as Huntington’s disease and Parkinson’s disease, are associated with affective disturbances and psychosis
(1,
2). Involuntary movements, affective disturbance, and catatonia in schizophrenia are phenotypically similar to symptoms found in basal ganglia disorders, suggesting the possibility of basal ganglia pathology in schizophrenia. Postmortem studies suggest both abnormally high and abnormally low basal ganglia volumes in schizophrenia
(3,
4), and some
(5,
6) but not all
(7) magnetic resonance imaging (MRI) studies have shown low caudate volumes in antipsychotic-naive patients.
The effects of treatment with atypical antipsychotics on basal ganglia volumes in a drug-naive population are unclear. We performed a longitudinal study of basal ganglia volumes in patients with first-episode schizophrenia, measuring volumes before and after treatment with risperidone. Comparison groups included patients treated chronically with typical antipsychotics and healthy subjects. The hypotheses were that 1) basal ganglia volumes in first-episode patients would be smaller than those in healthy subjects, 2) basal ganglia volumes in patients treated chronically with typical antipsychotics would be larger than the baseline values for the first-episode patients, and 3) there would be a decrease in volumes following risperidone treatment.
Method
Subjects
Demographic characteristics of the 30 first-episode, 12 chronic, and 23 healthy subjects appear in
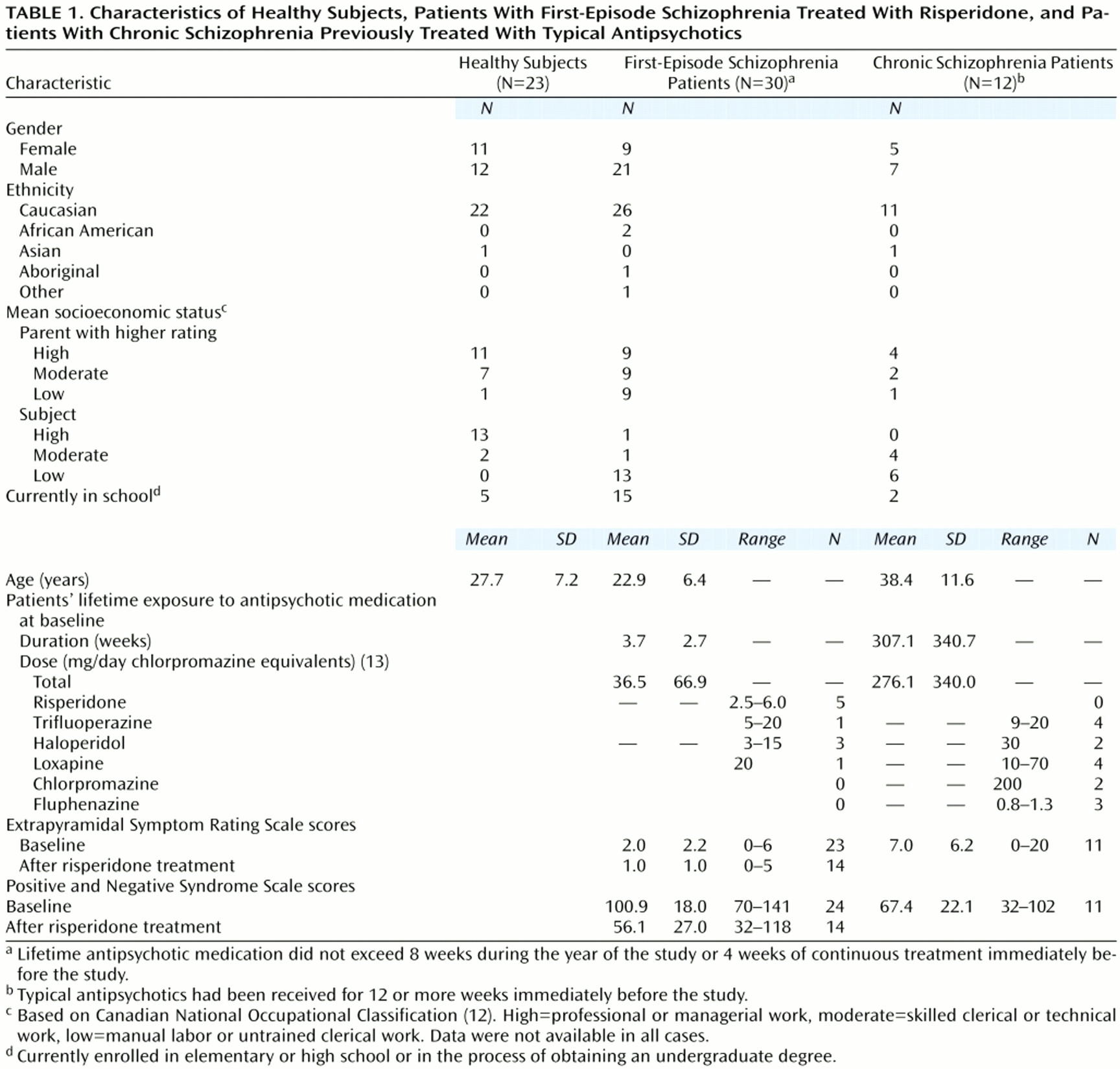
Table 1. The ratings of socioeconomic status were based on the Canadian National Occupational Classification system
(12). The ratings of parental socioeconomic status reflected the higher-rated parent. Diagnoses were made according to the DSM-IV criteria, with information obtained from the Structured Clinical Interview for DSM-IV. All first-episode and chronically treated patients were diagnosed with schizophrenia, except for one first-episode subject diagnosed with schizoaffective disorder.
First-episode patients were included if lifetime exposure to antipsychotic medication did not exceed 8 weeks of treatment in the year of the baseline scan or 4 weeks of continuous treatment immediately before scanning. The first-episode patients’ total lifetime exposure to antipsychotics was less than 12 weeks
(14). The chronically treated patients had received 12 or more weeks of continuous treatment with typical antipsychotic medication immediately before the baseline scan. The dose of antipsychotic medication was converted to chlorpromazine equivalents
(13). The mean dose for the chronically treated patients was 276.1 mg/day of chlorpromazine units. The 23 healthy comparison subjects were recruited from hospital staff and the local community.
The exclusion criteria were a history of significant head injury or loss of consciousness exceeding 5 minutes, a history of facial or nasal trauma, a history of DSM-IV substance abuse or seizure disorder, and a family history of psychotic disorders. Written consent was obtained from all participants. The study was approved by the Dalhousie University Research Ethics Committee.
Treatment and Clinical Measures
Ratings of psychiatric and extrapyramidal signs and symptoms were completed by research clinicians. Afterward, 24 of the first-episode patients were treated with risperidone (dose range=1–6 mg/day, mean=2.7 mg/day). Risperidone was the first atypical antipsychotic available in Canada for use with first-episode patients, and Canadian practice guidelines recommended using atypical antipsychotics for this patient group. Risperidone was routinely offered as first-line treatment during the time of enrollment in this study (1995–1998). Of the six patients not treated with risperidone, four refused all treatment, one was treated with olanzapine, and one participated in a controlled trial. Of the 24 patients taking risperidone, 15 were treated continuously for a minimum of 6 months, and these patients were used to study the effects of risperidone on basal ganglia structures. At baseline, one first-episode subject and two of the chronic subjects were receiving concomitant medications (lorazepam, 3 mg/day; benztropine, 2.0 mg/day; and fluvoxamine, 300 mg/day, respectively). At follow-up, one first-episode patient was receiving concomitant medication (sertraline, 25 mg/day). Of the nine patients treated with risperidone initially but excluded from the follow-up study, four did not receive a second MRI (two moved, two refused scan) and five were not taking risperidone at the time of the second scan (three changed medication, two later refused medication).
Extrapyramidal signs and symptoms were rated at baseline, at the time of the follow-up scan, and whenever medication was changed. The ratings were obtained by using the Extrapyramidal Symptom Rating Scale
(15). Interrater reliability, as reflected by intraclass correlation coefficients (ICCs), was high (ICC=0.84). Psychiatric symptoms were assessed by using the Positive and Negative Syndrome Scale
(16). Ratings were made at baseline, with changes in medications, and at follow-up.
MRI Measurements
The subjects were scanned with a Siemens Magneton Vision 1.5-T MRI scanner. A coronal inversion recovery sequence was obtained for each subject. The inversion recovery sequence was obtained as follows: TR/TE=2000/20 msec, field of view=200 cm, and matrix size=168×256, pixel size=1.04×0.78 mm. A total of 18 slices, 4 mm thick with a 1-mm interslice gap, were available. Follow-up scans were acquired a mean of 1.06 years after baseline (SD=0.41). Repeat scans were not available for the chronically treated group. Digitized slices were measured by using a Macintosh G3/350 Mz computer. Additional T2-weighted axial images of the entire brain were acquired to facilitate assessment of total intracranial volume. The axial T2-weighted images were highly sensitive to focal pathology and provided good demarcation of the CSF-containing spaces. Inversion recovery sequences (coronal T1) were obtained for anatomical detail and superior differentiation of gray and white tissue. The white-to-gray pixel intensity ratio in these scans was 1.42, compared with a value of 0.89 obtained by using a spoiled gradient recall acquisition sequence obtained on a 1.5-T GE scanner of similar quality.
Basal ganglia measurements were made by a single rater (D.J.L.) blind to diagnosis, time of scan, and gender. Total intracranial volumes were measured by two raters (D.J.L., V.M.G.). The films were visually inspected for motion artifact, tilt, rotation, and slice matching at follow-up. One subject was excluded because of head tilt. Slice-to-slice matching of the baseline and follow-up scans was assessed by comparison of the placements of the anterior commissure slice and of the first frontal horn slice. This approach was used in a serial scan follow-up study of lesions in multiple sclerosis
(17).
All regions of interest were manually selected with interactive shareware (NIH Image, version 1.61 ppc)
(18). The basal ganglia measures began two slices anterior and ended two slices posterior to the anterior commissure slice in every subject (five slices in each). The slices were determined a priori on the basis of the clarity of edge detection and unequivocal presentation of basal ganglia regions according to Duvernoy’s MRI atlas
(19). All basal ganglia measurements were the mean of four independent trials.
The caudate, putamen, and globus pallidus were manually selected on each slice. The most anterior slice showed the head of the caudate-putamen complex; however, these nuclei could not be separated at this level. The total bilateral caudate-putamen complex on this slice was therefore divided into two halves. The nucleus accumbens was included in the total caudate volume. Intrarater reliability was assessed in a randomly selected subset of 10 scans (caudate, ICC=0.99; putamen, ICC=0.97; globus pallidus, ICC=0.96; intracranial volume, ICC=0.99).
Data Analysis
Continuous demographic variables were analyzed by omnibus analysis of variance (ANOVA). A single ANOVA was performed with age entered as a dependent measure and group (first-episode, chronic, healthy) entered as a main effect. Differences in gender, socioeconomic status, and ethnicity were analyzed by using chi-square goodness-of-fit tests. Measures of total intracranial volume and baseline regional brain volumes were analyzed by an omnibus analysis of covariance (ANCOVA). For analysis of total intracranial volume, group and gender were entered as main effects, with age entered as a covariate. For regional brain volumes, total (left plus right) caudate, putamen, and globus pallidus volumes were entered as dependent measures, with group entered as a main effect and total intracranial volume entered as a covariate. Post hoc analyses of significant main effects were performed by using Bonferroni inequality/Dunn’s tests for comparisons of groups (with significance set to p<0.05)and paired t tests for comparisons of baseline and follow-up volumes. Scores on the Positive and Negative Syndrome Scale and the Extrapyramidal Symptom Rating Scale at week 12 of the medication trial were used as outcome measures. Medication doses and extrapyramidal symptom scores in the first-episode and chronic patients were compared by using unpaired Student t tests. The difference between baseline and follow-up extrapyramidal symptom scores for the risperidone-treated patients was assessed by a paired t test.
Discussion
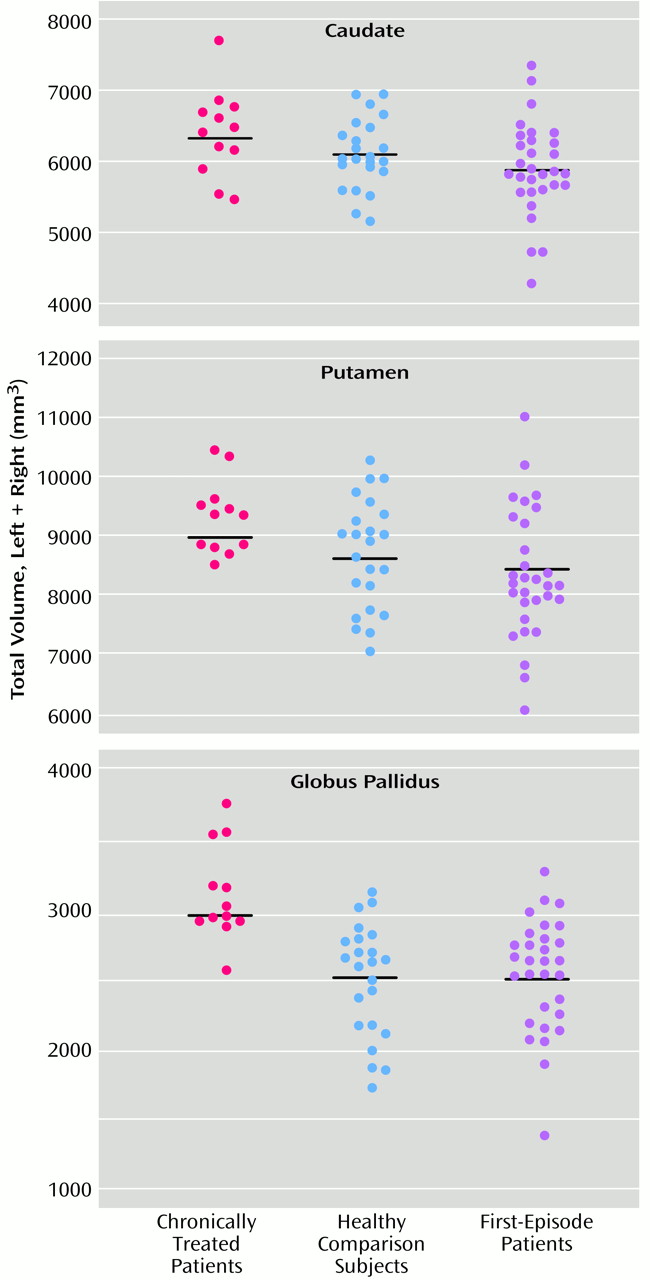
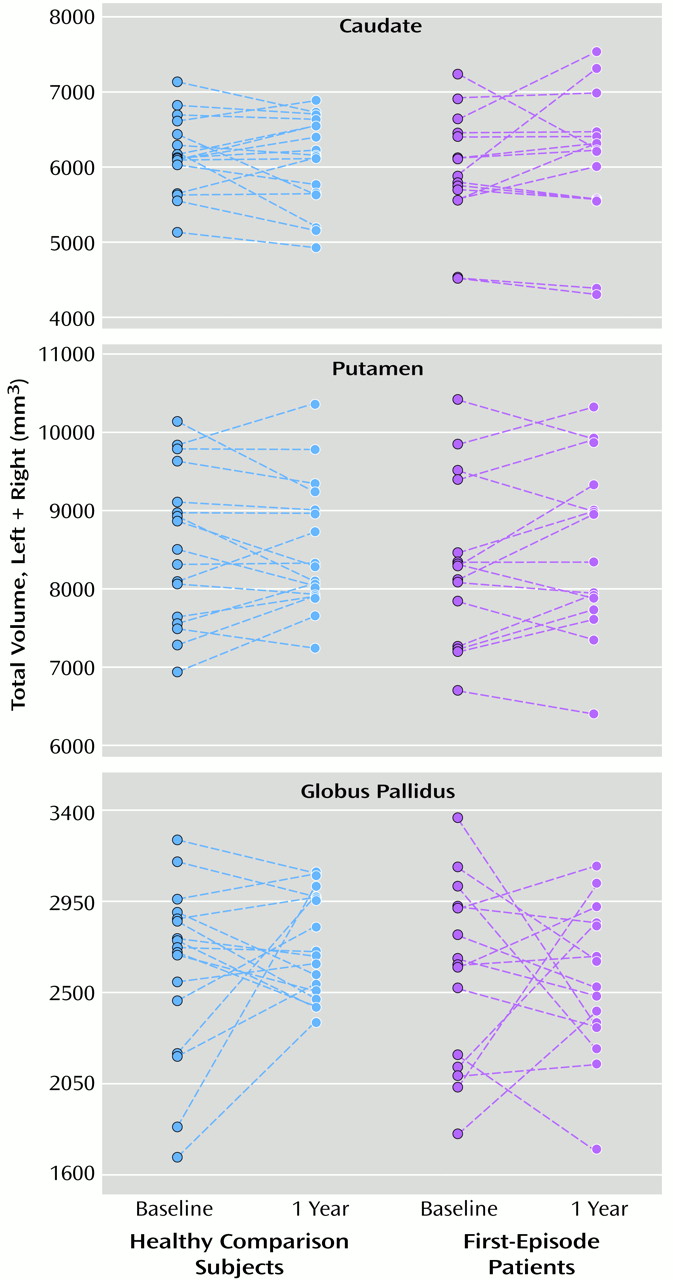
Three main findings emerged from this study. First, we replicated the observation of higher basal ganglia volumes in patients chronically treated with typical antipsychotic medication. Second, contrary to our hypothesis, we found no baseline differences in basal ganglia volumes between patients with first-episode schizophrenia and healthy comparison subjects. Third, we did not detect significant changes in basal ganglia volumes in first-episode patients after a mean of 1.06 years of exposure to risperidone.
In studies of nonhuman basal ganglia tissues
(21,
22), haloperidol treatment increased the number of neurons immunoreactive for the Fos-like protein in the caudate nucleus and putamen. A study of rats
(23) demonstrated basal ganglia enlargement after 1 to 6 months of haloperidol administration. Rat studies
(24,
25) have also demonstrated increased neuronal soma volumes in the prefrontal cortex and striatum after exposure to haloperidol. These investigations suggest neuronal activation in the striatum after exposure to typical antipsychotics. In addition, functional brain studies of humans
(26,
27) have demonstrated haloperidol-induced increases in deoxyglucose metabolism in basal ganglia regions. Both increased neuronal activation and increased neuronal metabolism may contribute to basal ganglia enlargement after exposure to typical antipsychotics.
The globus pallidus was approximately 25% larger in the chronically treated patients than in the healthy comparison subjects or the first-episode patients. In comparison, the caudate and putamen were larger by approximately 7% and 8%, respectively. Similar magnitudes of pallidal enlargement in patients treated with typical antipsychotics were seen in two previous studies
(28,
29), and Chakos et al.
(14) reported a significant correlation between basal ganglia volume and fluphenazine dose in first-episode schizophrenia. Basal ganglia enlargement may occur relatively rapidly and reach a plateau in the early phases of antipsychotic treatment, and even low-dose exposure to a typical agent may result in hypertrophy
(9).
Our data did not demonstrate abnormalities of basal ganglia volumes in the early phases of illness. Corson et al.
(5) reported low caudate volumes in never-medicated patients with schizophrenia, but Gur et al.
(28) found no differences between drug-naive patients and healthy comparison subjects. Normal brain development involves volumetric changes during adolescence
(30). Both age and gender in normal comparison subjects may require careful matching to determine true differences in subregional brain volumes.
The first-episode patients in this study did not exhibit basal ganglia enlargement after a mean of 1.06 years of continuous risperidone treatment. Studies comparing the effects of typical and atypical antipsychotics suggest that the basal ganglia enlargement induced by typical antipsychotics is reversible
(8,
10,
11) and that differences in the pharmacology of these medications may be related to volume changes.
Risperidone’s dopamine D
2 occupancy rates are dose dependent, and optimal doses (D
2 occupancy of 65%–80%) range from 2 to 5 mg/day
(31). The doses in this study (1–6 mg/day) were mostly within the optimal range. At doses above 6 mg/day, risperidone induces extrapyramidal symptoms without additional improvement in psychotic symptoms
(31,
32). Similarly, optimal doses of haloperidol also have 65%–80% D
2 basal ganglia occupancy rates
(33), and at recommended clinical doses (i.e., at recommended D
2 occupancy levels) haloperidol can increase basal ganglia volumes
(8). These results suggest that the mechanism underlying basal ganglia enlargement cannot be attributed to D
2 blockade alone.
Conversely, the evidence that any atypical antipsychotic causes a reduction in basal ganglia volumes below pretreatment levels remains questionable. Some groups have speculated that clozapine reduces basal ganglia volumes in animals
(34) and humans
(8,
10,
11). The clozapine trials with human subjects were confounded by switches from typical antipsychotics, and the animal study was based on D
2 receptor autoradiography, which may not be optimal for accurate volume estimation. The current data do not support the hypothesis of basal ganglia reduction from a never-treated baseline, at least for risperidone.
We and others
(20, 35–
37) have documented preexisting movement disorders in some drug-naive patients. Preexisting movement disorders in schizophrenia may be masked by antipsychotic drug effects, particularly those of typical antipsychotic medications. In the present study, 10 of 11 of the chronically treated patients had extrapyramidal symptoms. Of the first-episode patients in this study, 10 of 23 had extrapyramidal symptoms at baseline. This is a higher proportion than we reported previously
(20) and suggests a subtle underlying basal ganglia dysfunction in schizophrenia. Overall, the patients experienced improvement of extrapyramidal symptoms with risperidone.
The finding of no difference in basal ganglia volumes between the first-episode patients with and without extrapyramidal symptoms is in contrast to the higher basal ganglia volumes in the chronically treated patients with extrapyramidal symptoms. Enlargement of basal ganglia structures following treatment with typical antipsychotics may be closely related to the likelihood of movement disorders with traditional antipsychotic medications. The relationship of extrapyramidal symptoms and pretreatment basal ganglia volumes remains unclear.
There are several limitations to the present study. First, the results may have been influenced by the MRI acquisition protocol, slice thickness, the presence of interslice gaps, and the clarity of the white-to-gray tissue contrast. Typically, volumes acquired from two-dimensional and three-dimensional MRIs produce lower basal ganglia volumes than are found in postmortem studies
(3,
4,
38). The volumes and error variances obtained in the present study are similar to values determined by imaging protocols with thinner coronal slices
(5,
8,
14,
39). Interstudy comparisons do not indicate that slice thickness affects the accuracy or reliability of basal ganglia volume acquisition. Both accuracy and reliability of basal ganglia volumes are most affected by measurement methods. Our measurement protocols were clearly defined and highly reliable (ICC>0.90 for all regions). Second, subtle differences in basal ganglia morphology are difficult to identify in small study groups. The present study had a relatively small number of subjects because of the inherent difficulty in obtaining appropriate subjects and keeping subjects involved during follow-up. Third, if progressive changes in the basal ganglia are occurring over the early years of illness, we may not have detected differences between the healthy volunteers and patients with schizophrenia in the relatively short follow-up period of 1 year. However, as already noted, studies of nonhuman subjects suggest that changes related to treatment with typical antipsychotics occur relatively rapidly.
There are several implications of the present results. Typical antipsychotics are associated with extrapyramidal symptoms, which appear to be related to enlargement of the basal ganglia. While motor disturbances may also be present in never-medicated patients with schizophrenia, the structural correlates are less clear. Last, the effects of risperidone on basal ganglia structures appear to be different from those of typical antipsychotics. These observations suggest there may be lasting advantages to the use of risperidone (and possibly other atypical antipsychotics) in first-episode schizophrenia.