Neuropsychological studies show that deficits in verbal episodic memory
(1) in schizophrenia occur primarily during encoding and retrieval, with relative sparing of long-term storage
(2,
3). Functional imaging of healthy subjects documents regional differences in activity during encoding and retrieval. A general finding, summarized in the hemispheric encoding/retrieval asymmetry model
(4), is that episodic encoding and semantic retrieval activate the left prefrontal cortex, whereas episodic retrieval activates right prefrontal regions. Subsequent studies supported a distinct role for the right prefrontal cortex during episodic retrieval but found less evidence of hemispheric and task specificity for left prefrontal regions
(5–
8). Mesial temporal lobe distinctions have also been made on the basis of positron emission tomography (PET)
(9–
11): the rostral portion has been implicated in encoding, and the caudal portion has been implicated in retrieval. Functional magnetic resonance imaging has given different results
(12), and some have argued that the mesial temporal lobe is continuously engaged
(13) and that location and degree of mesial temporal lobe activity is related more to success
(14,
15) or stimulus novelty
(16–
19) than to stage of information processing. The purpose of the current study is to examine changes in cerebral blood flow (CBF) during word encoding and retrieval to assess the impact of schizophrenia on frontotemporal function.
Most imaging studies of episodic memory in schizophrenia have examined episodic retrieval and found frontotemporal dysfunction
(20–
22). Disrupted left medial and superior temporal lobe function and atypical middle temporal lobe asymmetries were documented in xenon-133 studies of word and face recognition
(23,
24). A series of PET studies of prose recall demonstrated prefrontal, thalamic, and cerebellar dysfunction
(25–
27). Frontotemporal abnormalities were found in free recall studies of supraspan word lists
(28) and recognition studies of visual paired-associate learning
(29). The mesial temporal lobe was implicated in studies of word-stem completion following shallow and deep encoding
(30,
31). These studies found hippocampal dysfunction across patients and left prefrontal dysfunction primarily in patients with deficit symptoms.
Studies of episodic encoding are more recent. A single photon emission computed tomography study
(32) measured CBF while patients and comparison subjects learned word lists of increasing semantic relatedness. The patients did not use semantic information to organize encoding, and their frontal and anterior cingulate regions were not activated. Degree of left inferior prefrontal activation was associated with patients’ use of semantic information to cluster words. Use of a semantic clustering strategy was also examined in a PET study of glucose metabolism that used a modification of the California Verbal Learning Test
(33). Patients used a serial order rather than semantic clustering strategy and had lower metabolism in frontal and temporal regions. Use of the serial order strategy correlated with reduced frontal and increased temporal metabolism. Finally, CBF was measured while patients learned and recalled lists ranging from one to 12 items in a word-stem completion task
(34). Increased memory demands led to prefrontal increases only in healthy comparison subjects, leading to the conclusion that hypofrontality is found only under conditions of cognitive stress.
To our knowledge, functional imaging studies of episodic memory in schizophrenia have not yet examined encoding and retrieval in the same experiment. This would be particularly informative given evidence that, although encoding deficits are prominent
(35), recognition may be relatively well preserved
(36), particularly for nonrelational recognition tasks requiring simple discrimination between new and old items
(37). In our previous study of word encoding and recognition in healthy subjects using the current paradigm, we found bilateral prefrontal effects, with greater right prefrontal and anterior cingulate activation during recognition
(38). Given these previous findings, we hypothesized that the recognition performance of patients with schizophrenia is relatively intact and that frontotemporal abnormalities are more prominent during word encoding.
Method
Participants
Participants were 23 patients (18 men and five women) and 23 healthy comparison subjects (12 men and 11 women) from the Regional Brain Function in Schizophrenia Center at the University of Pennsylvania. Comparison subjects were selected from a previous study group
(38) and were chosen to match patients in age (comparison subjects’ mean age=30.7 years, SD=10.0; patients’ mean age=35.2, SD=9.9), parental education (comparison subjects’ mean level=14.1 years, SD=3.0; patients’ mean level=12.9 years, SD=2.8), and handedness (there were three left-handers in each group). As expected, the patients had fewer years of education (mean=13.8, SD=2.8, versus mean=15.9, SD=2.2) (t=2.8, df=42, p<0.01). Because illness disrupts educational attainment, participants were not matched in education. To do so would have created a matching fallacy in which patients were compared with undereducated healthy subjects
(39).
Patients had a DSM-IV diagnosis of schizophrenia established by medical, neurological, and psychiatric evaluations
(40,
41). Comparison subjects also underwent standard medical, neurological, and psychiatric evaluations
(42,
43). Participants had no history of substance abuse or other medical, psychiatric, or neurological disorder that might affect brain function.
All patients were outpatients and ranged between mildly and moderately ill according to their scores on the Scale for the Assessment of Negative Symptoms (SANS)
(44) (mean score=26.4, SD=19.6, range=0–69), Scale for the Assessment of Positive Symptoms (SAPS)
(45) (mean score=16.5, SD=13.0, range=0–39) and Brief Psychiatric Rating Scale (BPRS)
(46) (mean score=31.8, SD=10.1, range=21–63). Clinical scales and PET scans were administered within the same month (median=21 days). Patients were reassessed on the day of the study to determine that no change in clinical status had occurred. One patient had been rated clinically a year before the study, but this patient had stable symptoms.
Patients had received a mean dose of 204.9 mg/day in chlorpromazine equivalents (SD=391.8, range=0–1479.5) of typical neuroleptics and 119.0 mg in chlorpromazine equivalents (SD=267.6, range=0–739.8) of atypical neuroleptics over their lifetime. Their mean age at onset of schizophrenia was 23.5 (SD=7.4), and the mean duration of illness was 12.2 years (SD=9.2). Ten patients were neuroleptic naive, and 13 were receiving medication; six were receiving typical neuroleptics, four atypical, and three both typical and atypical.
There were no differences in education, parental education, age at onset, duration of illness, or BPRS symptoms between medication-naive and medicated patients (all t values <1). However, medication-naive patients had more severe symptoms on the SANS (mean score=40.1, SD=18.5, versus mean score=18.3, SD=15.2) (t=2.9, df=18, p<0.05). Medication-naive patients had nonsignificantly more severe SAPS ratings than medicated patients (mean=22.9, SD=13.9, versus mean=12.4, SD=11.4) (t=1.9, df=19, p=0.07) and were slightly younger (mean=30.6, SD=5.4, versus mean=38.0, SD=11.1) (t=–1.7, df=19, p=0.09). After complete description of the study, written informed consent was obtained from all participants.
Tasks
Images were acquired during four conditions: resting baseline, sequential finger movement, word encoding, and word recognition. Stimuli were visually presented by means of a computer and Power Laboratory software
(47) on a monitor mounted from the ceiling at an angle perpendicular to the subjects’ line of sight. Words were centered on the screen as white block letters against a blue background. Words were presented for 5 seconds with a 2-second interstimulus interval during which a blank blue screen appeared. Word encoding preceded word recognition. The order of resting and motor baselines was variable.
Participants held a computer game pad in their right hand and made a button press every time a word was presented during the encoding condition and every time a word was recognized during the recognition condition. Button presses were visually monitored during encoding to ensure that participants were engaged and responding. Button presses and reaction times were automatically recorded during recognition to permit calculation of performance indexes. Lights were dimmed, and background noise was kept to a minimum.
Each condition was started 30 seconds before injection of 15O-labeled water to assure that participants were fully engaged. Conditions were separated by approximately 15 minutes to permit infused radioactivity to decay before the next infusion, resulting in a total study time of approximately 1 hour and 20 minutes.
The resting baseline was a standard condition
(48) (eyes open, ears unoccluded) during which participants were instructed to “relax with your eyes open, and refrain from talking or movement.” The motor baseline was developed as a reference task to control for motor activation during the acquisition and recognition conditions
(38). During the motor condition, participants sequentially touched their right thumb to the fingers on their right hand using large-amplitude finger movements at a rate of about 60 finger/thumb oppositions per minute.
Encoding and recognition conditions used the Penn Word Recognition Test
(24,
38,
49,
50). Briefly, the imaging version of the Penn Word Recognition Test consists of real words, 20 targets and 20 foils, presented at a rate of 5 seconds each. Targets and foils were selected from a standard word list
(51) and were balanced in frequency, concreteness, and imageability. During the encoding condition participants were instructed, “You will see 20 words, one at a time. When you see a word press the button. Look at the word the whole time it is on the screen and try to remember it. After you see the words once, there will be a blank screen, and then you will see the same words a second time. Continue to look at the words the second time and try to remember them.” Participants were presented with two repetitions of the 20 target items in a fixed order. During recognition, participants were instructed, “Now we are going to show you more words. This time, some of the words will be exactly the same as the words that you just saw, and some of the words will be different. If the word is the same as one you were previously shown push the button as quickly as you can. If the word is not one that you previously saw, do not press the button. If you are unsure, please guess. You will have 5 seconds to respond to each item.”
Forty trials were presented; half of the trials contained targets and half contained foils. Targets and foils were randomly assigned to each trial. Participants had no difficulty understanding task instructions. Practice was given on the computer game pad to ensure that participants were pressing the correct buttons before scanning.
Participants’ responses during recognition were recorded as true positive, false positive, true negative, and false negative. Two primary measures were examined. Discriminability was calculated as a measure of performance success, and true positive reaction time was calculated as a measure of performance effort. A response bias index was also calculated to assess response strategy. Discriminability and response bias index were calculated following the two-high threshold theory
(52), which generates statistically independent signal detection indexes.
Discriminability was calculated by subtracting the false alarm rate ([false positives+0.5]/[number of foils+1.0]) from the hit rate ([true positives+0.5]/[number of targets+1.0]). Values of 0.5 and 1.0 were added to the numerators and denominators to avoid division by zero. Discriminability reflects the proportion of time that an individual is either certain that an item is a target or certain that it is a foil and is an index of memory accuracy.
Response bias was calculated as follows: response bias index=false alarm rate/(1–discriminability). This represents the proportion of items on which individuals say yes when they are uncertain. A value of 0.5 reflects a neutral bias in which there is a 50/50 chance of saying yes or no when uncertain. Response bias index values greater than 0.5 indicate a liberal response bias, and values below 0.5 reveal a conservative response bias.
Reaction time was measured by calculating the median reaction time (in milliseconds) for true positive responses.
PET Data Acquisition
Images were acquired by using the UGM 240H volume imaging scanner
(53) composed of 6 NaI(Tl) crystals in a hexagonal arrangement. The camera has a 12.8-cm axial field of view and produces 64 image slices with an interslice distance of 2 mm. Spatial resolution was 5.5 mm in all directions (UGM Medical Systems
[54]). Participants’ heads were aligned along the orbitomeatal line, and manual head holder restraints reduced movement.
Participants received four intravenous injections of
15O-labeled water according to a modified bolus ramped-infusion technique
(55,
56). Intravenous lines were started approximately 10–15 minutes before scanning. This allowed time for activity to arrive from the cyclotron and time for participants to get used to the intravenous lines. A programmed infusion of 45 mCi of
15O-labeled water was delivered over 3.3 minutes. This results in rapid elevation of tissue activity to the peak of the count rate capability of the camera. The measuring period continues over 4.21 minutes, and dynamic scans were obtained continuously. Tissue activity concentrations peak over the first 1.5 minutes of the measurement period and then leveled because of increased washout
(56). Resulting images were corrected for attenuation, scatter, and dead time and used to obtain regional activity concentration of
15O-labeled water.
Image Analysis
Analysis of PET images was accomplished with SPM 96 statistical parametric mapping software
(57,
58). Images were realigned to the first image to help correct for head movement. Realigned images were normalized into a common space
(59) by using the standard PET template. A customized bounding box (–78:78, –112:76, –40:64) was used to account for a restricted field of view in the z direction. Images were smoothed by using a 15-mm full width at half maximum Gaussian kernel. Proportional scaling was used to adjust values in each voxel by the global mean for each scan. Scaled images were set to a common grand mean of 50 ml/100 g per minute.
Separate SPM 96 paired t test analyses of patient and comparison subject data were used to detect task-related differences within groups. Unpaired SPM 96 t test analyses examined between-group differences These analyses provided an index of task-related activation and yielded a t statistic for the difference at each voxel. Only voxels with a positive signal change were analyzed. These within-group t statistics were expressed as standardized z scores in final projection maps and thresholded at p<0.005, uncorrected, requiring 50 contiguous voxels, corresponding to a minimum t value of 2.8. As noted in previous imaging studies of memory in schizophrenia
(34), a lowered threshold helps avoid type II error and is justified given the large body of research documenting prefrontal dysfunction in schizophrenia. However, results for nonprefrontal regions should be interpreted with caution.
Output coordinates of activated voxels were expressed in Talairach space (x, y, z coordinates)
(59) for identifying functional Brodmann’s areas. Pearson product moment correlations were used to examine the relationship between task performance (discriminability) and differences between task performance and the motor baseline in scaled pixel intensity. Correlation analyses were performed by using MEDx software
(60).
Discussion
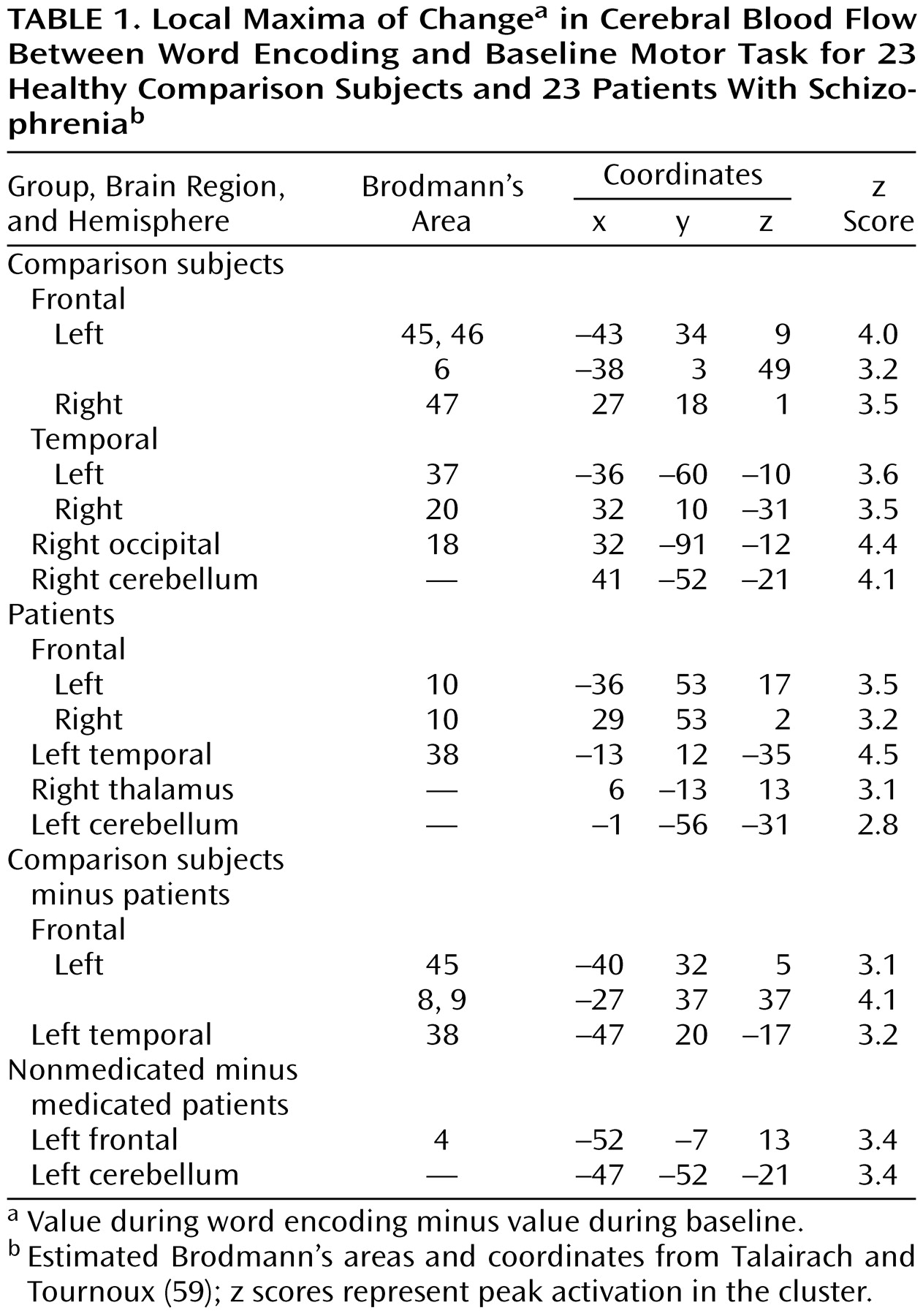
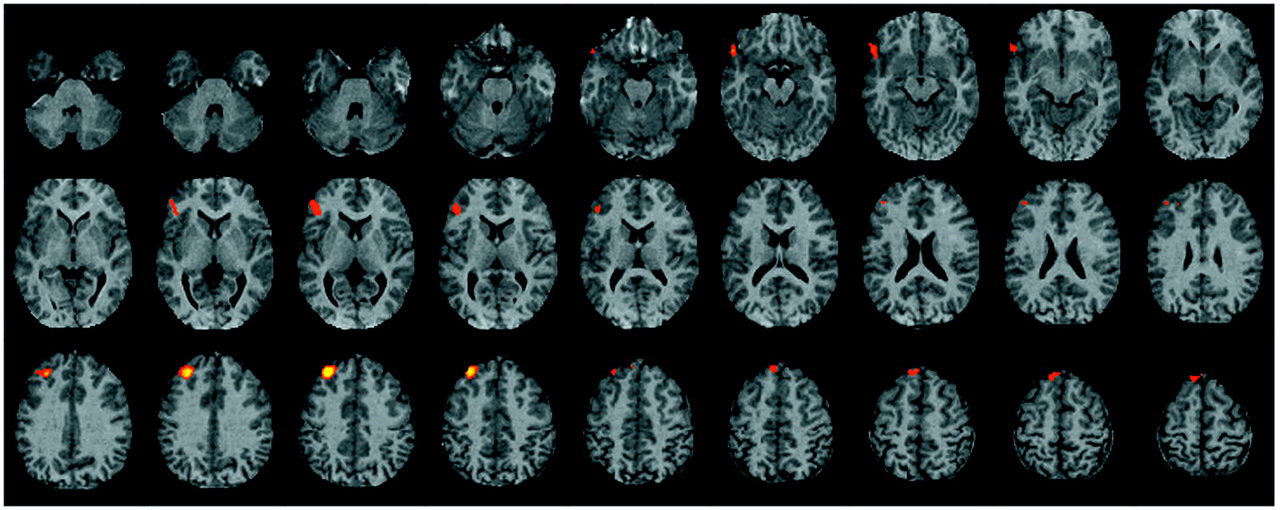
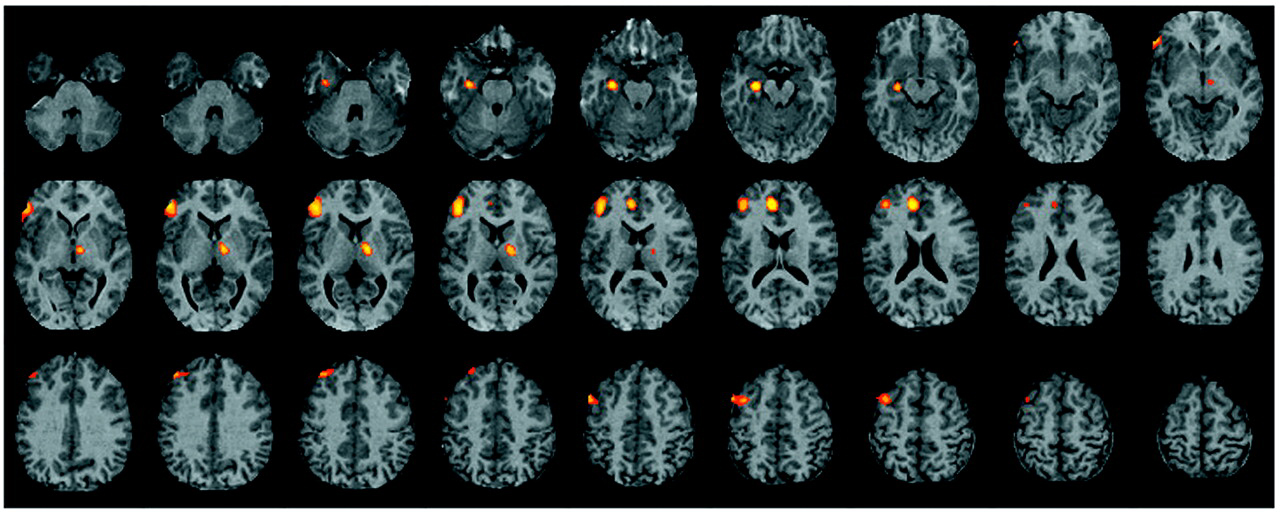
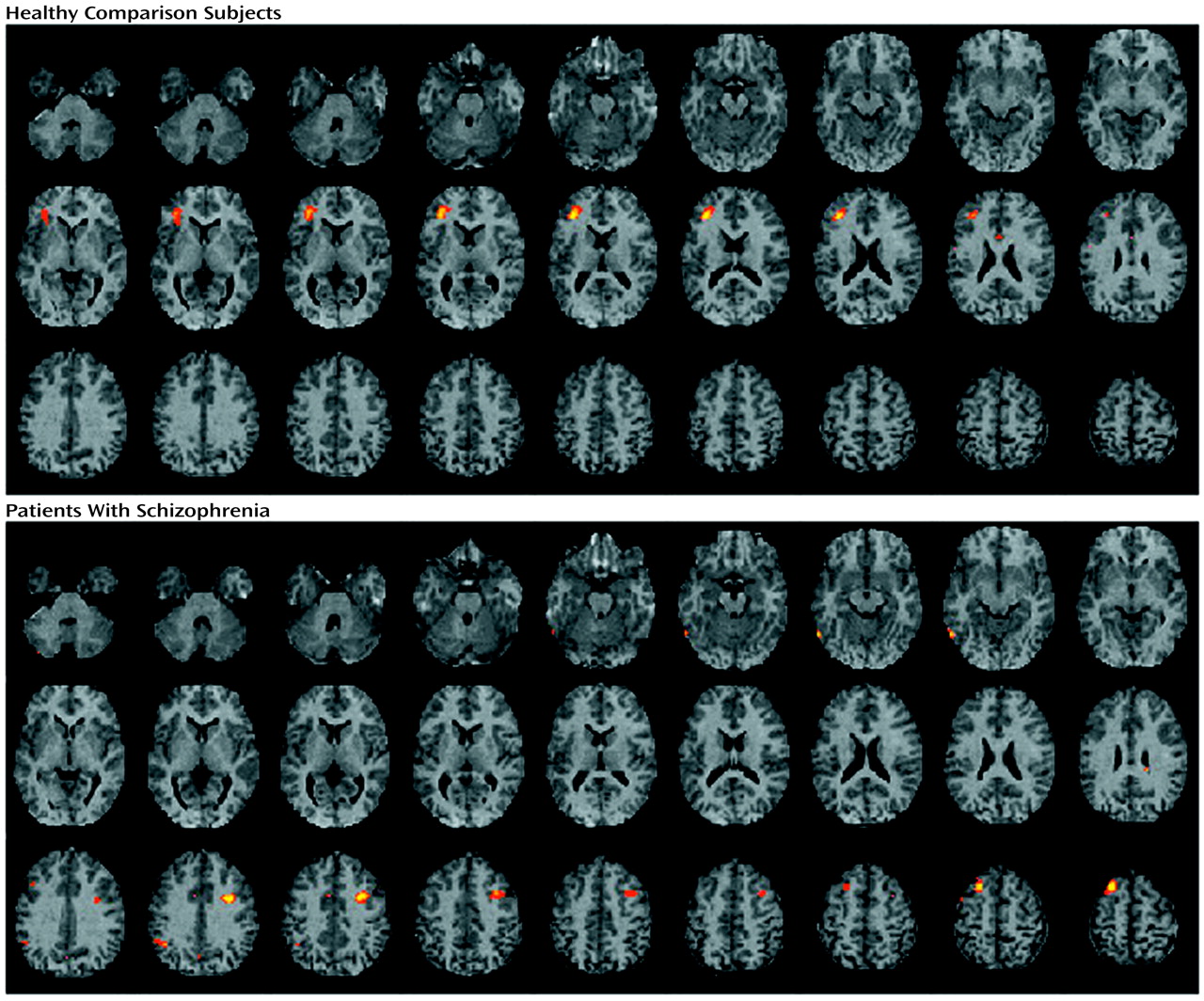
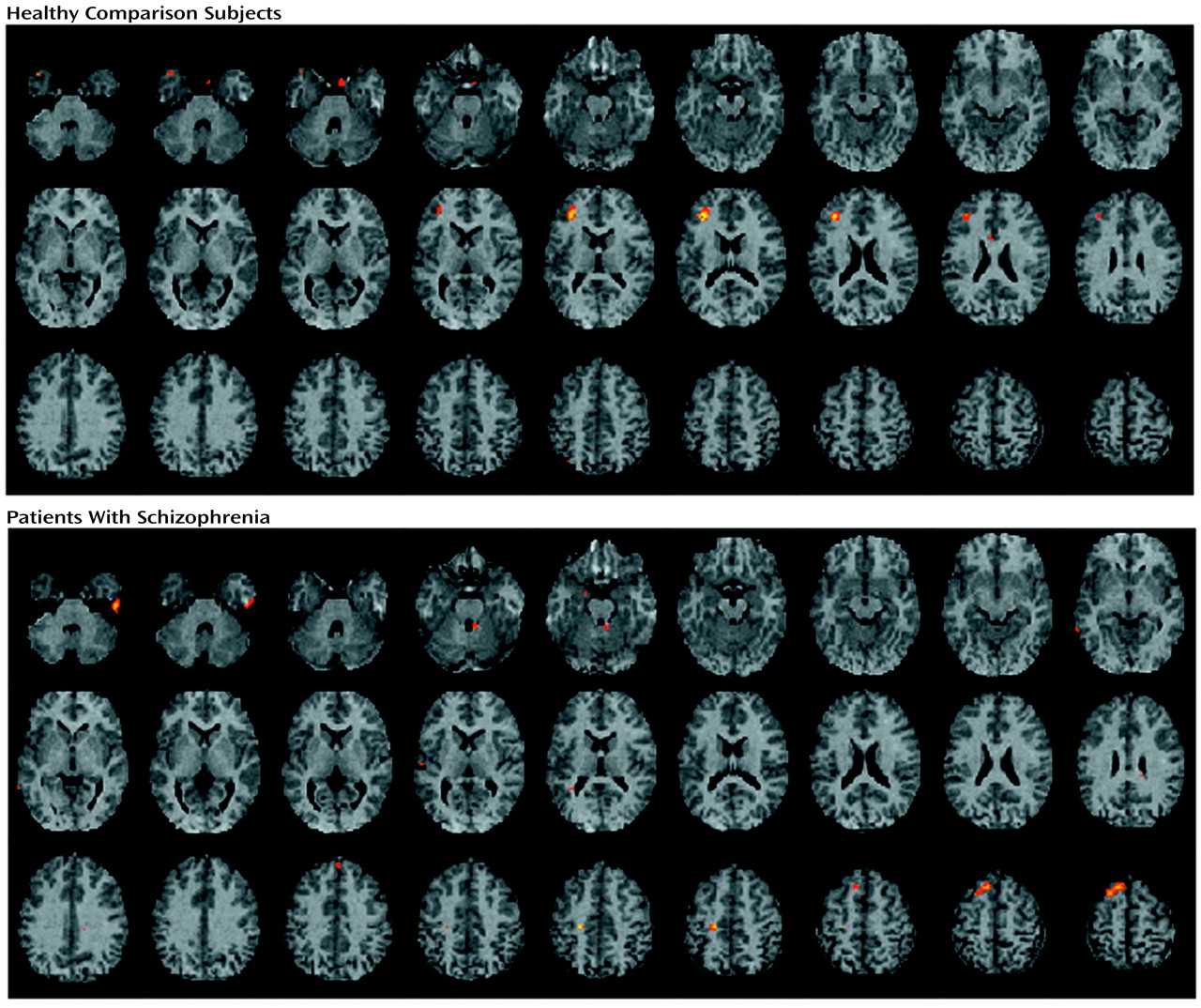
The current study found evidence of reduced left hemisphere frontotemporal activation in patients with schizophrenia during episodic memory encoding and retrieval. Differences between patients and healthy subjects in CBF during encoding were centered in the left inferior prefrontal gyrus and extended to the left middle frontal gyrus, superior frontal gyrus, and left superior temporal cortex. Smaller activations in the left inferior and middle frontal gyrus of patients were also seen during recognition, with additional activation impairments found in the left anterior cingulate, left mesial temporal lobe, and right thalamus. There were no group differences during recognition in a right prefrontal region believed to mediate episodic retrieval
(66). Greater left inferior prefrontal activation during encoding and recognition was correlated with better task performance only in comparison subjects. These group differences did not appear to be due to medication status or to reduced power to detect activation effects in the patient group.
These results underscore the importance of the dorsolateral portion of the left inferior frontal gyrus (Brodmann’s areas 45 and 46) to episodic memory and to the pathophysiology of schizophrenia
(26,
67–
69). As in our previous study
(38), this region was activated during word encoding and recognition in healthy comparison subjects. This finding is consistent with modifications
(5–
8) of the hemispheric encoding/retrieval asymmetry model
(4), clarifying that, although the right prefrontal cortex appears specific to episodic retrieval, left prefrontal regions are involved in both stages of episodic memory processing. Activation of this region during multiple stages has led to the suggestion that the region is responsible for maintaining a verbal representation of word stimuli to permit encoding and subsequent retrieval
(5,
6).
Differences between patients with schizophrenia and healthy subjects in left inferior prefrontal activation might, therefore, reflect patients’ difficulties with active maintenance and manipulation of verbal representations caused by impaired working memory
(70–
72). This explanation gains support from findings of patient abnormalities in left prefrontal activation during “n-back” working memory tasks despite normal levels of task performance
(65,
73). Statistical control of working memory deficits has also been shown to eliminate free recall deficits in patients with schizophrenia
(74). However, the ability of patients to perform the recognition task suggests that their working memory abilities were sufficient to maintain the words long enough to permit encoding.
An alternative explanation is that patients processed words on a more superficial level
(75) and did not use semantic information to organize encoding and retrieval. Evidence of normal left prefrontal activation in patients during semantic decision tasks
(76) suggests that semantic processing at the lexical level was intact and that prefrontal abnormalities more likely reflected impaired strategic processes caused by executive difficulties
(69,
77–
79). This conclusion is supported by the finding that although patients tend to encode words superficially, when provided with cues
(2,
3), they can successfully use semantic information to improve performance
(36,
79,
80). Imaging data have also shown that left prefrontal activation is maximized when healthy subjects process word associations semantically
(17) and that the degree of left prefrontal activation and metabolism in patients is related to their use of semantic clustering versus serial order strategies
(32,
33). Because encoding strategies were not systematically controlled or measured in the current study, further testing of this hypothesis will require future studies employing depth-of-processing
(81) imaging paradigms
(82,
83).
Contrary to our hypothesis, prefrontal dysfunction was also prominent during recognition and extended to the left anterior cingulate, left mesial temporal lobe, and right thalamus. This is potentially an incongruous finding, given that we found that patient recognition performance was relatively intact, as has been found in previous studies
(36). However, Danion et al.
(37) demonstrated how recognition tasks can be performed despite a shallow level of processing. In a study of recognition paradigms, these investigators proposed that nonrelational tasks such as the current forced-choice paradigm can be successfully performed on the basis of feelings of familiarity (noetic awareness) rather than conscious recollection (autonoetic awareness). If the task is altered to require conscious recollection (e.g., probe of source memory), then frontal brain potentials increase
(84) and patient recognition performance becomes impaired
(37,
85).
The current correlational findings support the hypothesis that patients and comparison subjects were engaged in different levels of information processing and memory awareness. The strongest correlation in comparison subjects was in the left inferior prefrontal gyrus, consistent with high-level organizational processing during encoding and autonoetic awareness during recognition. In contrast, correlations in patients were more posterior and included a basal temporal area involved in linguistic processing
(86). The patient correlations might reflect operation of a compensatory network used to facilitate language processing
(76).
Finally, lack of group differences in right prefrontal function suggest that retrieval mechanisms were intact, permitting successful performance regardless of level of memory awareness. Future imaging studies contrasting free recall and recognition paradigms with and without source memory probes would help to test these speculative hypotheses.
Reduced patient activation of the anterior cingulate, mesial temporal lobe, and thalamic regions during the Penn Word Recognition Test should be viewed with caution given the relatively low thresholds used for between-group analyses and the fact that comparison subjects did not show mesial temporal lobe and thalamic activation during the within-group analysis. Nevertheless, patient deficits in anterior cingulate activity have been documented in a variety of imaging paradigms
(25,
78,
87–
90) and attributed to problems in attention
(27) or performance monitoring
(91). Patient deficits in mesial temporal lobe function have been reported during episodic retrieval
(30,
31) and attributed to impaired conscious recollection. Abnormal thalamic function has been documented
(25,
27,
92) and discussed in relation to information processing abnormalities
(93). These explanations might all be applicable to the current results. However, there are insufficient data to test these multiple hypotheses, and a more parsimonious explanation may be that an abnormality in the functional connectivity of the prefrontal cortex with temporal-limbic regions accounted for the multifocal abnormalities
(26,
94).
The current study suffered from several limitations. In measuring task effects we chose to contrast encoding and retrieval with a low-level motor baseline task rather than performing serial subtractions of increasingly complex reference conditions
(95). Because of this, we failed to control for linguistic processes and other nonmemory functions that might complicate interpretation. The choice of appropriate reference tasks is a problematic issue in the field, and different arguments have been raised in favor of active versus passive reference conditions
(96). The rationale for the current design was twofold. First, performing serial subtractions of increasingly complex reference tasks has the potential limitation of confounding the interpretation of activation effects if interactions occur between reference tasks at each stage of the serial subtraction procedure
(97). Second, the combination of relatively long scan times (4.2-minute scans) and long (15-minute) decay periods limited the number of scans because of participant fatigue. These time constraints made use of serial subtraction procedures less feasible. Therefore, in the motor task we followed the recommended procedure of establishing similar levels of sensorimotor activation between groups to provide an internal standard to help ensure that subsequent group differences in cognitive effects were not artifactual
(65).
A second limitation is that more than half of the patients were receiving medication at the time of testing. All patients were stable outpatients, and the neuroleptic-naive patients were tested during their first episode. For the stable patients who were receiving medication, discontinuing that medication would have exacerbated symptoms and hindered performance. The clinical benefit of medication was reflected in the fact that patients receiving medication had less severe symptoms. However, there is no evidence that long-term neuroleptic exposure in schizophrenia affects CBF in the prefrontal or mesial temporal lobe regions
(98,
99). Current results support this: there were few regional CBF differences between medicated and unmedicated patients, and no differences occurred in hypothesized regions. There was also no effect of medication on task performance, notwithstanding recent reports that atypical neuroleptics improve memory performance
(100). However, relatively few patients were receiving atypical compounds, and a larger-scale investigation of atypical medication effects on performance and blood flow is warranted.