Methamphetamine shares with other drugs of abuse (e.g., cocaine, heroin, alcohol) the ability to enhance release of the neurotransmitter dopamine in the striatum (caudate, putamen, nucleus accumbens)
(4,
5), with the extent of the dopamine increase correlated with the intensity of the “high” in humans
(6,
7). For this reason, it continues to be assumed that some of the effects of methamphetamine exposure in humans are explained in large part by alterations in activity of the pre- and postsynaptic dopamine system in the brain
(8). Thus, we previously reported that striatal tissue levels of dopamine can be low in some users of methamphetamine
(9)—as low as those in Parkinson’s disease in some striatal subdivisions
(10)—which suggests that some of the unpleasant consequences of methamphetamine during drug withdrawal (e.g., depression, cognitive impairment) might be due in part to a striatal dopamine deficiency. It is also possible that short- and long-term adaptive changes due to overactivation of dopamine receptors occurs in methamphetamine-exposed human brain, which might explain some aspects of drug-taking behavior.
Dopamine receptor types can be classified by their ability to stimulate (D
1 receptors) or inhibit (D
2 receptors) adenylyl cyclase (EC 4.6.1.1) through the mediation of the stimulatory G protein (G
s or, in striatum, G
olf [11]) or the inhibitory G protein (G
i, G
o), respectively (see reference
12 for a review). Since the pharmacological literature on the influence of dopamine D
1 receptor agonists/antagonists on the behavior of human psychostimulant users is scanty, the role of the dopamine D
1 receptor in psychostimulant use and abuse in the human has not yet been established. However, the preliminary observation that a single dose of a dopamine D
1 receptor antagonist (SCH 39166, ecopipam) can partially block the euphoric effects of cocaine in chronic users of the psychostimulant
(13) suggests that the dopamine D
1 receptor might mediate part of the pleasurable effects of dopaminergic stimulants. Although no data have yet been provided indicating that dopamine D
1 receptor agonists are rewarding to humans
(14), agonists acting on this receptor are self-administered by nonhuman primates
(15,
16).
Apart from these pharmacological studies, information on the status of the dopamine D
1 receptor system in human psychostimulant users is limited to our postmortem finding of normal levels of dopamine D
1 receptor protein in the dopamine-rich striatal subdivisions of methamphetamine users and users of cocaine, with the exception of higher protein concentration of the receptor restricted to the nucleus accumbens subdivision in the methamphetamine users
(17). Changes or lack of changes in brain neurotransmitter receptor concentrations can, at most, only suggest differences in receptor function. Therefore, we have now extended our investigation to include measurement, in the postmortem brain tissue of chronic methamphetamine users, of a more dynamic index of dopamine D
1 receptor function, namely dopamine-stimulated adenylyl cyclase activity, since adenylyl cyclase is considered to be the key effector of dopamine D
1 receptor function
(12). In order to establish the possible site and specificity of any disturbance in dopamine D
1 receptor, G protein, and adenylyl cyclase coupling, we also measured stimulation of adenylyl cyclase by a nonhydrolyzable GTP analog guanylyl-imidodiphosphate [Gpp(NH)p], which stimulates adenylyl cyclase by direct activation of the stimulatory G protein, i.e., bypassing the dopamine receptor, thereby assessing the G protein and adenylyl cyclase coupling. We report that dopamine D
1-stimulated adenylyl cyclase activity is decreased in striatum of human methamphetamine users, a finding that might explain some of the short- or long-term aspects of drug-taking behavior.
Results
No significant correlations (Pearson) were found in either group between subject age and levels of any of the outcome measures. There were weak, negative correlations between postmortem interval and basal adenylyl cyclase activity in the absence or presence of GTP (comparison subjects: r=–0.35 to 0.01, df=19, p>0.11; methamphetamine users: r=–0.45 to –0.15, df=14, p>0.08) with that in frontal and temporal cortices of the comparison subjects being statistically significant (r=–0.58 to –0.45, df=19, p value range=0.005 to 0.04). For the comparison subjects (df=19), no significant correlations were found between postmortem interval and maximal dopamine stimulation in any of the examined brain areas (nucleus accumbens: r=–0.42, p=0.06; caudate: r=–0.33, p=0.14; putamen: r=0.15, p=0.51; frontal cortex: r=–0.29, p=0.20; temporal cortex: r=–0.23, p=0.31). No significant correlations were seen in the methamphetamine users (df=14) either between postmortem interval and maximal dopamine stimulation in any of the examined brain areas (nucleus accumbens: r=–0.15, p=0.57; caudate: r=–0.20, p=0.47; putamen: r=–0.03, p=0.92; frontal cortex: r=–0.26, p=0.33; temporal cortex: r=–0.20, p=0.46). A negative correlation was observed between postmortem interval and maximal Gpp(NH)p stimulation in all of the brain areas (comparison subjects: r=–0.59 to –0.36, df=19, p value range=0.004 to 0.11; methamphetamine users: r=–0.51 to –0.33, df=14, p value range=0.04 to 0.21), with significant correlations seen in the caudate (r=–0.59, df=19, p<0.005) and frontal cortex (r=–0.54, df=19, p<0.02) of the comparison subjects and in the nucleus accumbens of the methamphetamine users (r=–0.51, df=14, p<0.05).
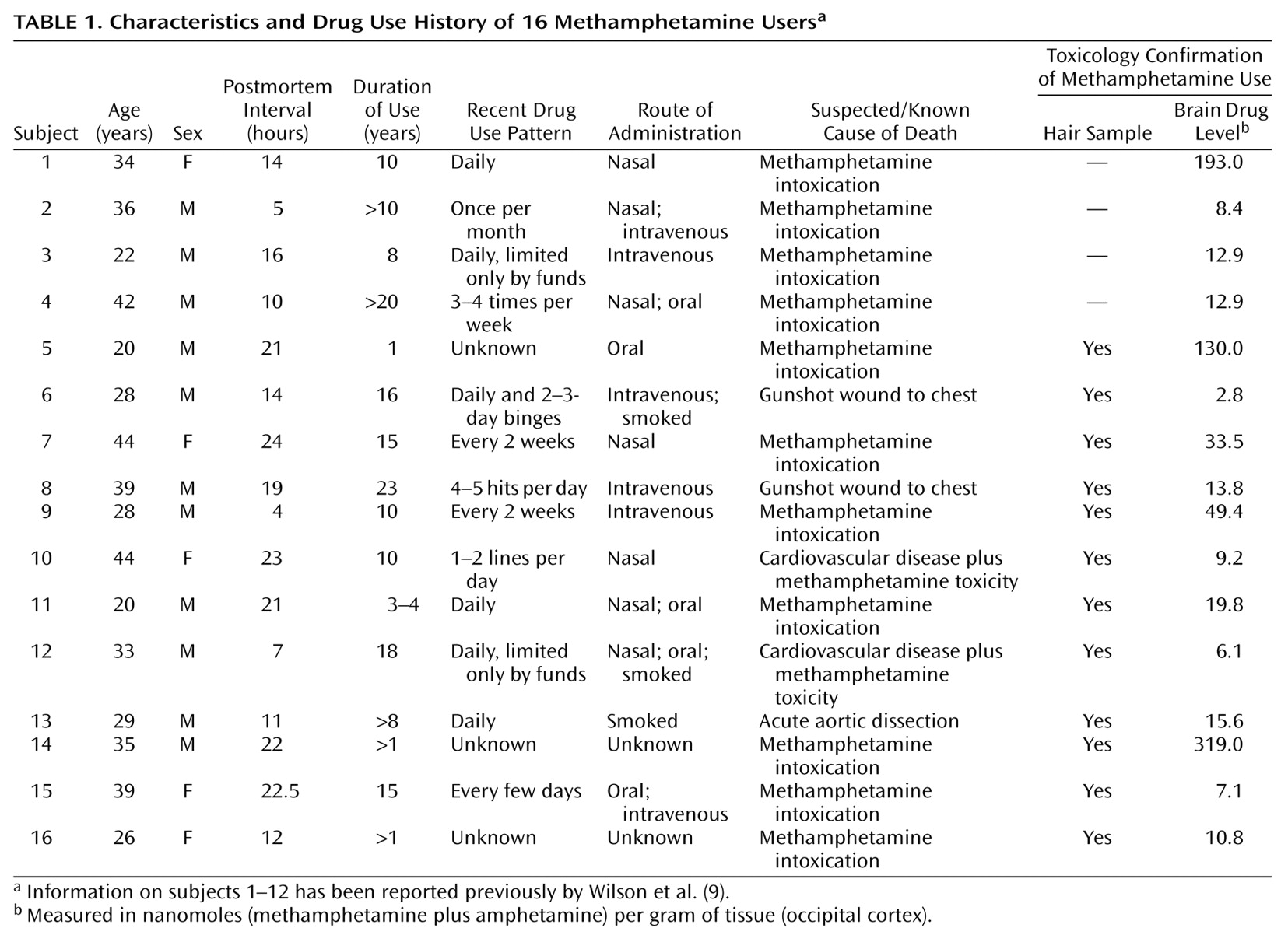
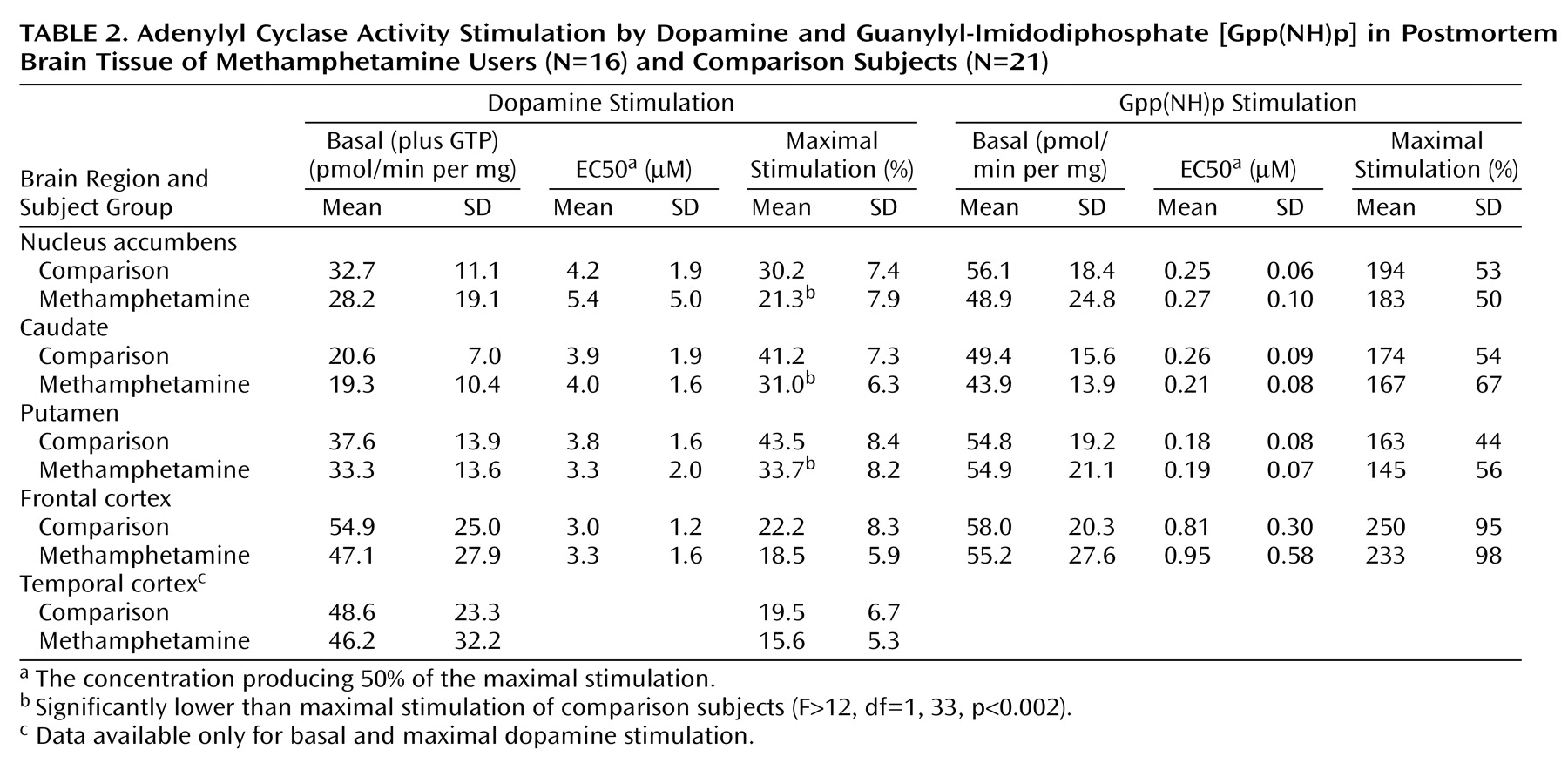
As shown in
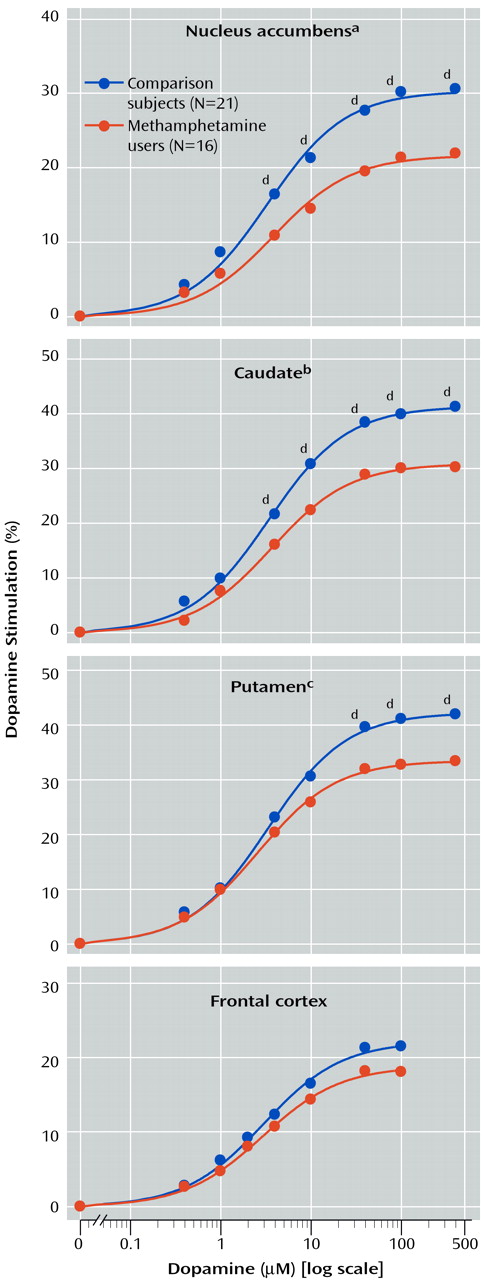
Table 2, basal activity of adenylyl cyclase (in the presence of 10 μM GTP) was normal in all examined brain regions of the methamphetamine users. Dopamine titration (0.4–400 μM) in frontal cortex and striatum showed the expected dose dependence and was highly significant (F>200, df=7, 245, p<0.001) (
Figure 1). No significant differences were observed in EC50 between the comparison subjects and the methamphetamine users (
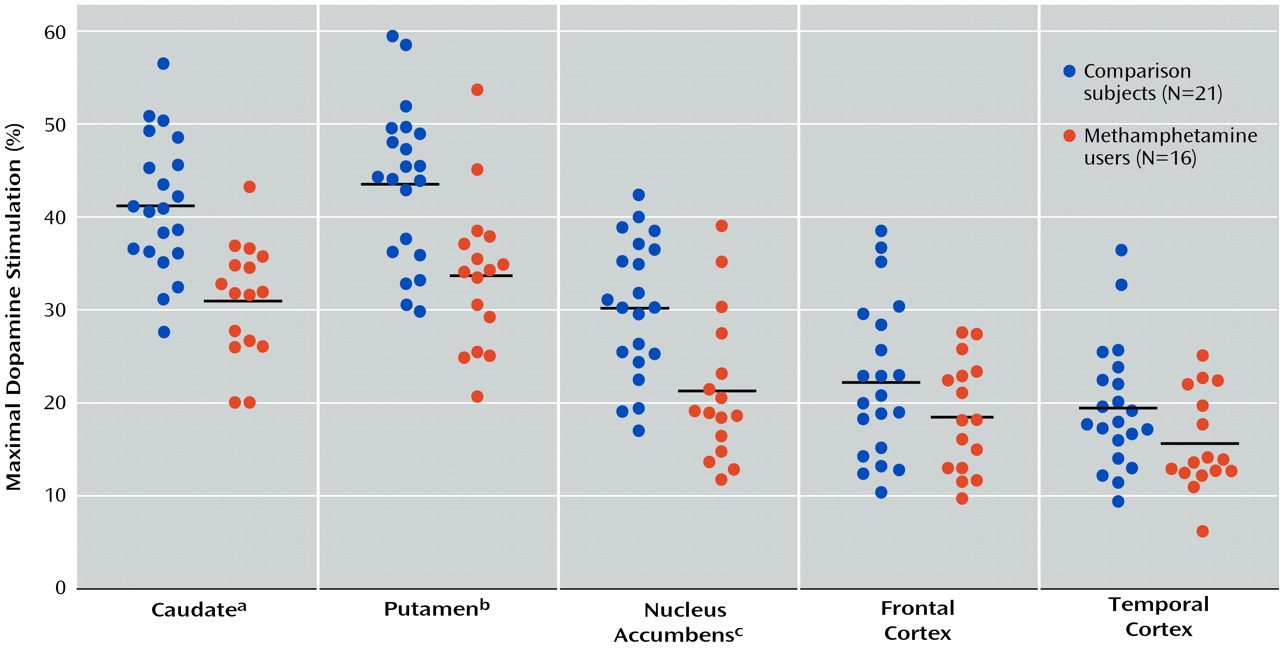
Table 2). However, the magnitude of maximal dopamine stimulation (calculated from the titration curves) in nucleus accumbens, caudate, and putamen of the methamphetamine users was significantly lower than those of the comparison subjects by 25%–30%, with nonsignificantly lower stimulation seen in the frontal cortex (17% decrease) and temporal cortex (20% decrease, calculated from a single saturating concentration of dopamine at 100 μM). As shown in
Figure 2, although there was extensive overlap between the individual comparison and methamphetamine values for maximal dopamine stimulation, most of the methamphetamine user values in the striatum fell lower than the mean level for the comparison subjects.
Gpp(NH)p titration (10–8–10–4 M) was carried out in the nucleus accumbens, caudate, putamen, and frontal cortex. As described in
Table 2, no significant difference was found between the methamphetamine users and comparison subjects in basal adenylyl cyclase activity, EC50, and the magnitude of Gpp(NH)p stimulation in any of the brain regions.
No significant correlations (Spearman rank) were found between the extent of maximal regional dopamine stimulation reported here and the regional protein concentrations of the dopamine D
1 receptor in the 12 examined methamphetamine users in which both measurements were conducted
(17), or between maximal dopamine stimulation and brain (occipital cortex) levels of methamphetamine plus its metabolite amphetamine in the entire group of 16 methamphetamine users
(18) (data not shown). In addition, there was no significant correlation (Pearson) between dopamine stimulation and duration of drug use in those cases (N=11) for which accurate duration information was available (data not shown).
Discussion
The major finding of our study is that striatal dopamine D1-stimulated adenylyl cyclase activity is decreased in human chronic methamphetamine users.
We attempted to address, as much as possible, potential confounding issues associated with postmortem investigations of drug users. Thus, we obtained forensic evidence in blood, autopsied brain, and, for most of the users, hair, proving that the subjects of our study used methamphetamine both recently (drug positive in blood and brain) and chronically (sequential hair segments). Although the results of drug analyses and retrospective structured interviews suggested that the drug users used only methamphetamine, it is quite possible that the subjects might previously have used other drugs of abuse or might even have had a neurochemical defect before drug taking that could have affected the biochemical outcome measures. Issues surrounding differences in age, postmortem time, and agonal status (sudden versus slow death) were addressed by matching the comparison subjects and methamphetamine users with respect to these variables. We have also previously reported that the extent of maximal dopamine stimulation of adenylyl cyclase activity in biopsied versus autopsied human brain is similar
(19). The absence of significant correlation between postmortem interval and dopamine stimulation was consistent with the reports on rats
(22–
24).
Our finding of decreased striatal dopamine stimulation of adenylyl cyclase in brain tissue of human methamphetamine users is consistent with investigations reporting below-normal dopamine D
1 stimulation of striatal adenylyl cyclase following either acute administration
(25–
27) or repeated exposure plus drug challenge
(28,
29) of amphetamine to rodents. The animal data suggest that impaired dopaminergic stimulation of adenylyl cyclase in humans was not a preexisting abnormality but rather a consequence of exposure to methamphetamine. However, repeated amphetamine administration to rodents can also induce enhanced dopamine D
1 receptor-mediated inhibition of nucleus accumbens neurons
(30,
31). Although the relevance of these experimental animal studies employing different treatment paradigms and outcome measures to human drug users is unknown (see reference
32 for review), this suggests that some dopamine D
1 receptor-mediated functional changes might be differentially affected by methamphetamine.
The cause of the decreased dopamine stimulation of adenylyl cyclase in our human investigation is not known but in principle could be explained by decreased levels of or coupling between the dopamine receptor, G protein, and adenylyl cyclase. Our previous observations that striatal levels of dopamine D
1 receptor protein are either normal (caudate, putamen) or elevated (nucleus accumbens) in methamphetamine users
(17), together with our findings that levels of the stimulatory striatal G protein G
olf (33) and basal adenylyl cyclase activity (present investigation) are normal, suggest that decreased dopaminergic stimulation of adenylyl cyclase is unlikely to be explained by low concentration (e.g., due to drug toxicity) of these components of the dopamine D
1 receptor system. Similarly, the demonstration in methamphetamine-exposed rodents
(25–
29) and humans of normal adenylyl cyclase activation by activators (GTP or Gpp[NH]p), which directly stimulate adenylyl cyclase via G
s/G
olf, suggests that G protein and adenylyl cyclase coupling is preserved and indicates specificity of the dopamine-related disturbance. However, the possibility cannot be excluded that G protein and adenylyl cyclase coupling associated with dopamine receptor activity might have represented only a minor part of total G protein and adenylyl cyclase coupling assessed under the conditions of our assay.
We suggest that desensitization of dopamine-stimulated adenylyl cyclase in the methamphetamine users could be explained by impaired coupling between the dopamine D
1 receptor and the stimulatory G protein
(27). Since all of the methamphetamine users used the drug recently as well as chronically (for at least 1 year), receptor desensitization might have occurred consequent to either acute or chronic drug exposure.
Although the relative biological importance of the different dopamine D
1-linked effectors (e.g., adenylyl cyclase, phospholipase C
[34]) is not known, it is reasonable to expect that much, if not most, of dopamine D
1 receptor activity is mediated by G protein activation of adenylyl cyclase
(12). Given that methamphetamine causes release of dopamine from striatal nerve endings, decreased dopamine-stimulated adenylyl cyclase activity in methamphetamine users can reasonably be considered as desensitization (i.e., biochemical tolerance) due to excessive dopaminergic stimulation of the dopamine D
1 receptor. However, as the literature is limited on the nature of the involvement between dopamine D
1 receptor function and drug-taking behavior in the human, the biological significance of impaired dopamine D
1 receptor function in methamphetamine users is uncertain. In this regard, in view of the preliminary evidence that the dopamine D
1 receptor might mediate part of the euphoric effects of dopaminergic psychostimulants in humans, decreased dopamine D
1 receptor function in a limbic brain area (nucleus accumbens) suggests that some D
1 receptor-related tolerance to the euphoric effects of methamphetamine might occur in human users following repeated drug exposure. It is also possible that subnormal dopamine D
1 receptor-adenylyl cyclase activity in this brain area, in addition to the even more severe reduction in tissue stores of striatal dopamine
(9,
10), might explain part of the dysphoria associated with withdrawal from methamphetamine. Finally, the preliminary report describing decreased craving for the psychostimulant cocaine in human users following administration of a selective D
1 agonist
(14) also suggests the much more speculative possibility that decreased dopamine D
1-stimulated adenylyl cyclase activity in limbic brain could explain, in part, compulsive drug craving, which can occur in some chronic methamphetamine users
(1).
Previously, we reported that concentrations of the dopamine D
1 receptor were selectively increased in the postmortem nucleus accumbens subdivision of the striatum of 12 of the 16 methamphetamine users examined in the present study
(17). We now find, however, that despite the increased receptor levels, a functional, and probably more biologically relevant, index of dopamine D
1 receptor activity is below normal in the nucleus accumbens as well as in two other subdivisions in which dopamine D
1 receptor number was normal. This discrepancy between receptor number and function suggests that caution should be employed in predicting functional changes in receptor activity from differences in receptor and G protein concentration. In this regard, the previous reports of changes in several components of the dopamine D
2 receptor system in brain tissue of methamphetamine users, e.g., a trend for decrease in D
2 receptor protein levels
(17,
35) and decreased inhibitory G protein
(33), need to be extended to include assessment of whether these changes actually affect D
2 receptor-mediated activity. Unfortunately, however, because of high intersubject variability in our postmortem brain study, we were unable to develop a valid procedure that could assay dopamine D
2 receptor-inhibited adenylyl cyclase activity in autopsied brain homogenates (Tong and Kish, unpublished observations).
We suspect that changes in activity of different dopamine (e.g., D1, D2) and nondopamine receptor systems underlie some of the behavioral effects of psychostimulant drugs. Our data provide the first functional data in human brain suggesting that the dopamine D1 receptor might be one of the systems involved in mediating as yet undetermined aspects of methamphetamine-induced behavior. Clinical studies of pharmacological agents modifying dopamine D1 receptor function in methamphetamine users will establish the biological significance of our neurochemical findings.