Saykin and co-workers
(1) reported that patients with schizophrenia are distinguished from healthy comparison subjects by the selectivity and relative severity of the patients’ impairment in verbal learning and memory, compared with other neuropsychological functions. Such an impairment has been found for different types of stimuli (e.g., words, sentences, stories, and nonsense syllables), in a variety of research paradigms (e.g., paired associates, free recall, cued recall, recognition formats, and repetition priming), and within a large range of schizophrenia patient groups (e.g., in patients with acute, chronic, reactive, and late-onset schizophrenia and in patients with tardive dyskinesia)
(2).
Schizophrenic patients have been found to have abnormalities in frontal
(4) and temporal
(5) structures that previous studies have identified as important for human memory performance
(6). Gur and co-workers
(7) reported correlations between frontal and temporal volume deficits and impaired memory performance in schizophrenia. Regarding functional abnormalities, studies of cerebral blood flow (CBF) that have used verbal memory tasks have detected lower levels of prefrontal activation in patients with schizophrenia than in healthy comparison subjects
(8–
10). Hazlett and co-workers
(11) found that patients with schizophrenia have lower relative glucose metabolic rates in both the frontal and the lateral temporal cortices during a serial verbal learning task. In accordance with other investigators
(12), they suggested that an abnormality in the functional connectivity between these structures plays a central role in the pathophysiology of cognitive dysfunction in schizophrenia. Similar observations were made by Ragland and co-workers
(13), who contrasted episodic encoding and recognition of words by measuring CBF with positron emission tomography (PET) in outpatients with schizophrenia and in healthy subjects. In that study, patients had deficits in activation of the left frontotemporal regions during word encoding and deficits in activation of the left frontotemporal regions, anterior cingulate, and right thalamus during word recognition. Approximately one-half of the patients were neuroleptic naive, and the others were treated with conventional antipsychotics, with second-generation antipsychotic agents (e.g., clozapine or risperidone), or with both types of agents. As there were also sex differences between the patient and comparison groups in the study by Ragland et al., we attempted to replicate the findings of Ragland et al. by using functional magnetic resonance imaging (fMRI) to investigate the neural circuitry used during episodic encoding and recognition of words in male schizophrenia outpatients, all of whom were treated with second-generation antipsychotics, and in healthy male comparison subjects. We chose to study the pathophysiology of hypothetical core and persistent impairments associated with schizophrenia rather than the transient changes associated with episodes of psychotic relapse. Therefore, the study subjects were stable outpatients with schizophrenia in remission, rather than acutely ill patients, and healthy volunteers.
Discussion
The findings of this fMRI study of episodic memory in schizophrenia, contrasting encoding and recognition of words in the same experiment, replicated previous neuropsychological
(3) and PET findings
(13) showing a lower level of frontotemporal activation in patients with schizophrenia, compared to healthy subjects, despite intact recognition performance.
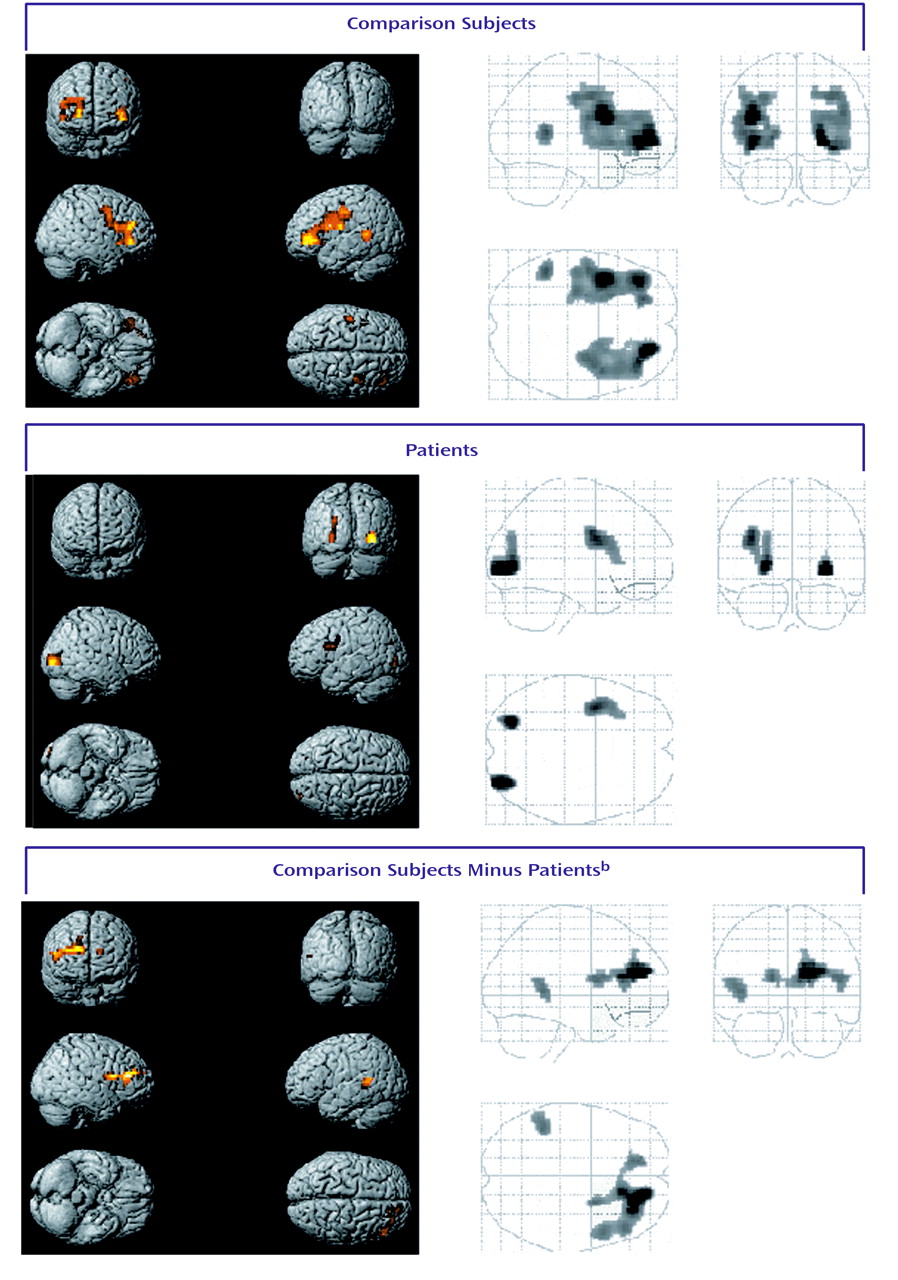
The activation pattern that we observed during episodic encoding in the comparison group was for the most part consistent with previous reports
(24–
31). Only our finding of right hemispheric activations in the frontal cortex did not confirm the hemispheric encoding/retrieval asymmetry model
(32). However, previous studies have shown that the encoding of verbal materials is not invariably left-lateralized. Lee and co-workers
(29) described activations in the bilateral anterior frontal cortices when activation values for verbal retrieval were subtracted from those for verbal encoding, and Otten et al.
(31) found bilateral ventrolateral prefrontal activations when subjects subsequently remembered words they had judged to refer to animate things. As suggested by Mottaghy et al.
(28), the use of verbal materials with a high content of object imagery might be one reason for such bilateral prefrontal activations. Furthermore, in the current study all the material was familiar to the subjects, since it had been presented before scanning. Such a condition has been associated with activation of the right ventrolateral prefrontal cortex
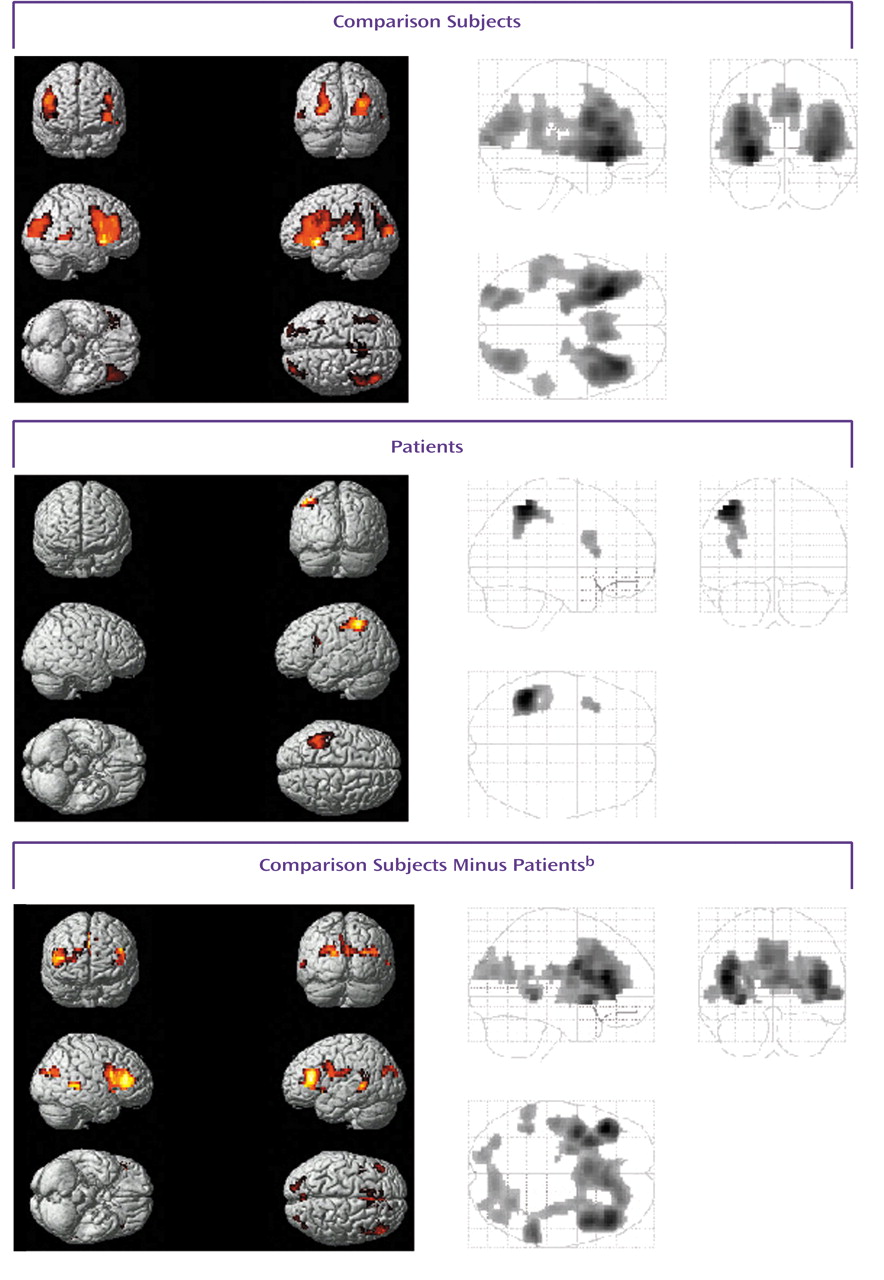
(33). With regard to episodic memory recognition, our findings for the healthy subjects corroborate the findings of other researchers
(13,
25–
28,
30,
34–38).
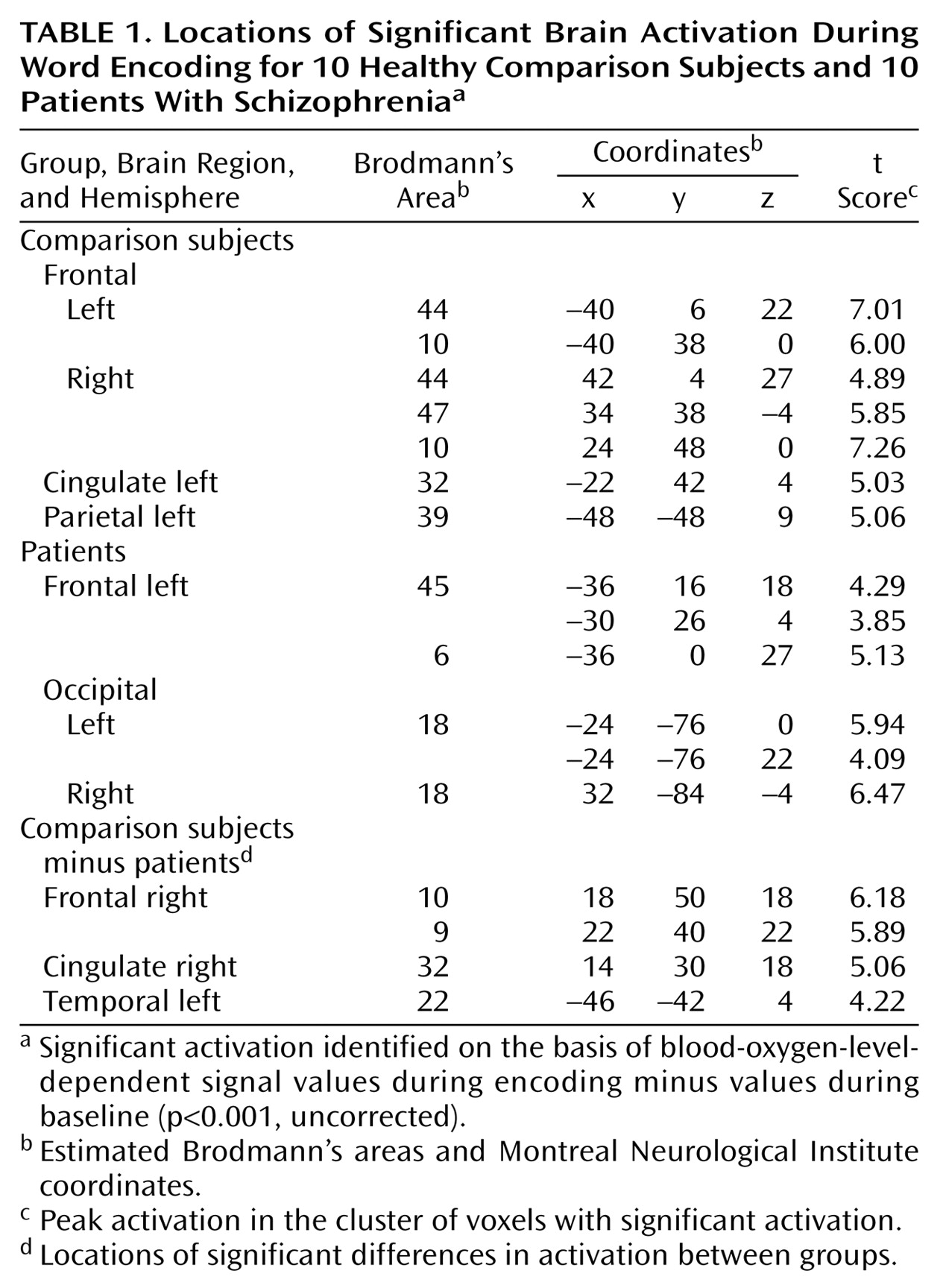
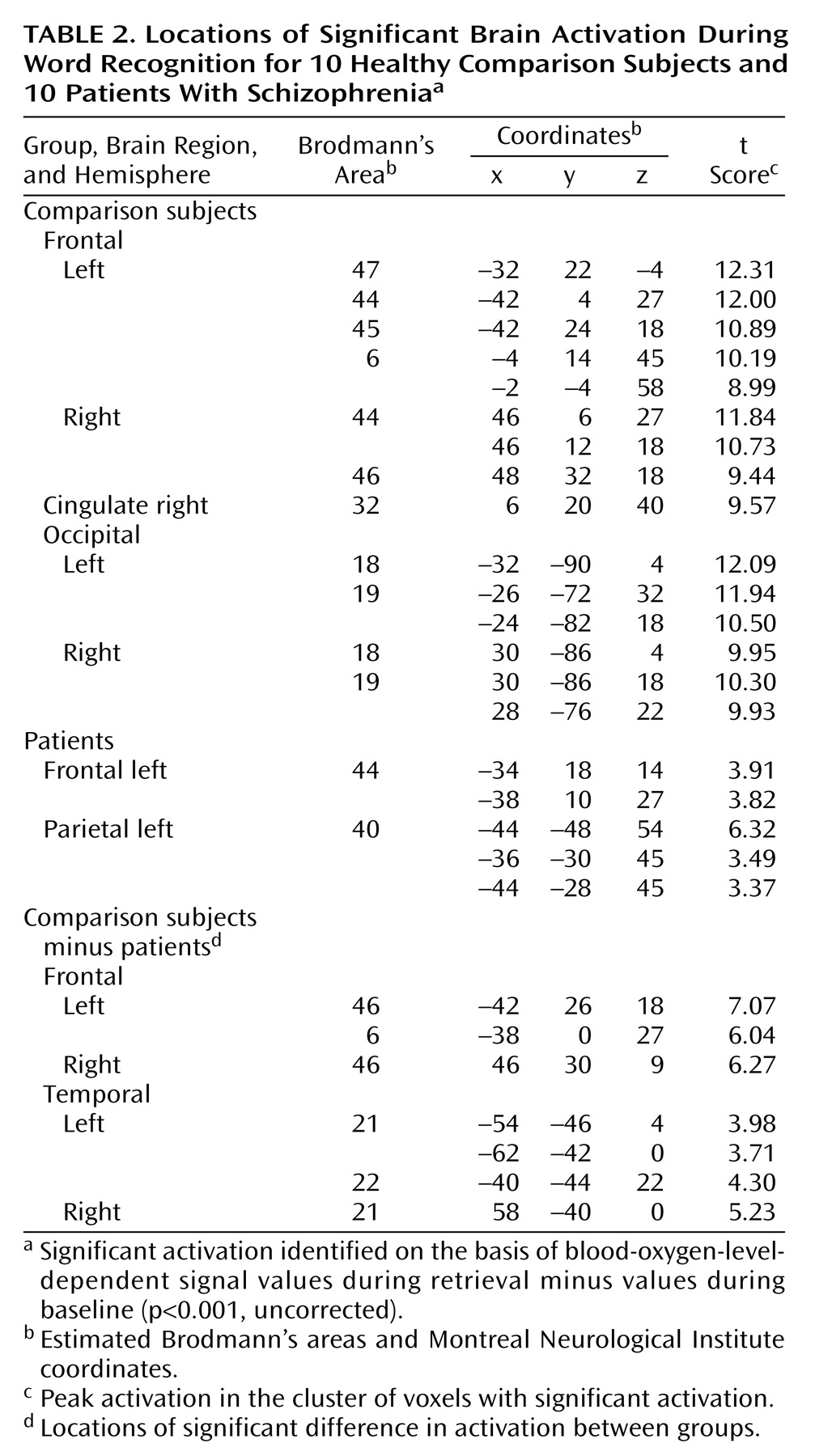
Differences between the patients and the comparison subjects during encoding were found in the right dorsolateral and anterior prefrontal regions, right anterior cingulate, and left lateral temporal cortex. During recognition, patients had impairments in activation in the bilateral dorsolateral prefrontal and lateral temporal cortices.
The left dorsolateral prefrontal and right anterior prefrontal regions have been associated with verbal working memory in healthy individuals
(39). Correspondingly, impairment of working memory and executive functions in schizophrenia has been associated with decreased blood flow in the dorsolateral prefrontal cortex
(40). Our finding of a lower level of activation in these regions for the patients than for the comparison subjects might, therefore, be related to impaired working memory in schizophrenia. However, levels of activation during recognition were comparable in both groups, which indicates that the patients’ working memory abilities permitted successful encoding. In agreement with Ragland and co-workers
(13), we suggest that the lower level of prefrontal activation during episodic encoding in the patient group may represent executive difficulties. This suggestion is further supported by our finding of a lower level of activity in the anterior cingulate in the schizophrenia group. This structure has been proposed to play a prominent role in the executive control of cognition
(41). The lack of anterior cingulate activation may reflect difficulties in both initiating appropriate responses and suppressing inappropriate ones, especially when attentional demands are high.
During both encoding and recognition, relatively lower activity was seen in the patients’ lateral temporal cortices. Similar findings have been reported by Ganguli et al.
(8), who investigated changes in regional CBF associated with the performance of an auditory verbal supraspan memory task. The lateral temporal cortex (Brodmann’s areas 21 and 22)
(42,
43) has been associated with the semantic processing of words. Thus, difficulties in this area may be associated with the differences between the patients and the healthy subjects that were observed in lateral temporal activation during episodic recognition.
Animal studies have implicated a cortical circuit involving the dorsolateral prefrontal cortex and parietal, temporal, and thalamic regions in working memory
(44). The disruption of an equivalent circuit in humans could be responsible for the executive dysfunction observed in patients with schizophrenia.
Several details of our study design that may limit the generalizability of the results must be considered. First, the study included a small number of subjects, and all subjects were male. Some neuropsychological studies of gender differences in cognition in schizophrenia have demonstrated more impairment among male patients in several functional domains
(45,
46), whereas others have indicated either no gender differences
(47,
48) or even greater abnormalities among women
(46,
49,
50). Further studies are needed to elucidate the effect of gender on memory performance and brain activation in schizophrenia. Second, the patients in the study were stable outpatients with schizophrenia in remission who were treated with novel antipsychotics. These drugs have been associated with improved performance on cognitive tasks
(51). Consequently, our study group may have already profited from such a therapeutic effect, and therefore they would not have the dysfunctions of untreated schizophrenia when they joined the study. However, episodic memory impairments in schizophrenia have also been found in studies that have reduced the confounding effects of medication
(2), and there is no evidence that long-term neuroleptic exposure in schizophrenia affects CBF in the prefrontal and/or temporal lobe regions
(52). Third, since all subjects had completed the words subtest of the Recognition Memory Test before scanning, training effects must be considered. The activation patterns observed in the study group thus cannot be equated with those that would have emerged without this test run. On the other hand, having subjects complete a prescan test may eliminate artificial findings resulting from mere differences between participants in ability to follow test procedures. Last, we chose to contrast encoding and recognition with a resting baseline reference condition rather than performing serial subtractions of increasingly complex reference tasks
(53). We were therefore not able to control for nonmemory functions. However, the use of active reference conditions has the potential limitation of confounding the interpretation of activation effects if cognitive components interact between one or more reference tasks
(54). Indeed, this study was not designed to look for cognitive differences between the patients and the healthy comparison subjects but to look for differences of functional networks employed to process the tasks in question. Nevertheless, our findings would have been strengthened by including a motor baseline task with the resting baseline task to control for motor activation during the acquisition and recognition conditions.
Our findings are highly clinically relevant insofar as encoding and recognition are needed both for everyday life and for participation in the workforce. Therefore, deficiencies in this area may render psychosocial rehabilitation efforts more difficult. The study findings add to our understanding of neuropsychological dysfunction in patients with schizophrenia and support the need to take such specific impairments into account when planning treatment.