Bedside neurological evaluations have been employed for several decades in studies addressing the question of schizophrenia’s “organic” basis (reviewed in references
1–3). Neurological examination abnormalities have been generally called “soft” neurological signs, in keeping with their reputed lack of specificity, validity, or localizing value
(3,
4). Several studies have shown that patients with schizophrenia have higher scores on measures of neurological abnormalities, compared with healthy persons. Many previous studies were limited by possible confounding factors, including prior use of neuroleptic medications and illness chronicity. Studies of first-episode, never-treated schizophrenia patients can address these confounds. Observations of generalized neuropsychological impairment in first-episode schizophrenia
(5–
8) have suggested a higher rate of global neurological examination abnormalities in such patients than in healthy comparison subjects. However, only a few studies
(9–
11) have explored neurological examination abnormalities in first-episode patients.
The diagnostic specificity of neurological examination abnormalities for schizophrenia remains unclear. As with many other aspects of schizophrenia neurobiology, comparisons of neurological examination abnormalities between schizophrenia patients and other psychiatric groups have yielded less consistent results than have comparisons of schizophrenia patients and healthy subjects. Comparisons of neurological examination abnormalities and overlapping neuropsychological deficits in schizophrenia and nonpsychotic psychiatric disorders have yielded mixed results, although a tendency for greater impairment in schizophrenia has been reported
(12–
15). Boks et al.
(16), reviewing this literature, found that only tactile extinction and four conventionally cerebellar signs were significantly more prevalent in schizophrenia than in mood disorders; insufficient data were available to assess the diagnostic specificity of motor sequencing tasks. Prior studies of diagnostic specificity
(2,
3) have not accounted for the possible effects of chronicity and prior treatments on neurological performance.
Comparisons between schizophrenia patients and patients with other psychotic disorders in particular domains of neurological performance can shed light on the specific functional deficits of schizophrenia and increase the clinical relevance of the neurological examination in the diagnosis and treatment of psychoses. Psychotic patients are well served by accurate pretreatment diagnosis, particularly because accurate diagnosis influences the likelihood of appropriate treatment
(17,
18). Although clinicians sometimes avoid a definitive diagnosis until they have observed the early course of the patient’s illness, the early course is itself influenced by the initial treatment. However, problems with communication and cooperation during initial evaluation of a psychotic person can make it difficult for a clinician to make a diagnosis by using standard phenomenology-based criteria. Supporting data can thus be valuable in the clinical assessment of psychoses, particularly at the patient’s first presentation.
The validity of neurological examination abnormalities in schizophrenia also remains uncertain. Structural brain abnormalities in schizophrenia, as measured by magnetic resonance imaging (MRI), include smaller volumes in the heteromodal association cortex
(19), basal ganglia
(20–
22), and cerebellum
(23). Although MRI has been used to examine relations between structure and cognitive deficits (e.g., references
24–26), few studies have examined relationships between neurological abnormalities and brain structure. Given the increasingly held view that schizophrenia is associated with structural changes in a widely distributed network of association cortical structures
(19,
27,
28), it is likely to be more fruitful to examine correlates of composite brain structures such as the heteromodal association cortex rather than single structures in isolation. Our first goal was to examine diagnostically specific differences between patients with first-episode schizophrenia, patients with nonschizophrenia psychoses, and healthy comparison subjects. The three groups were compared on motor and cognitive neurological examination abnormalities by using subscales of the Neurological Evaluation Scale
(29) derived from principal-components analysis
(30). Second, we examined the relationships between neurological examination abnormalities and structural brain abnormalities proposed to underlie the neurological functions that are impaired in schizophrenia. Specifically, we predicted 1) that scores for the more cognitively demanding perceptual tasks included in the neurological examination would be correlated with volumes of the association cortex and 2) that scores for the motor tasks would be correlated with volumes of the basal ganglia and the cerebellum. To address these questions, we examined a subset of first-episode schizophrenia patients with both MRI data and data on neurological examination abnormalities.
Method
Subjects
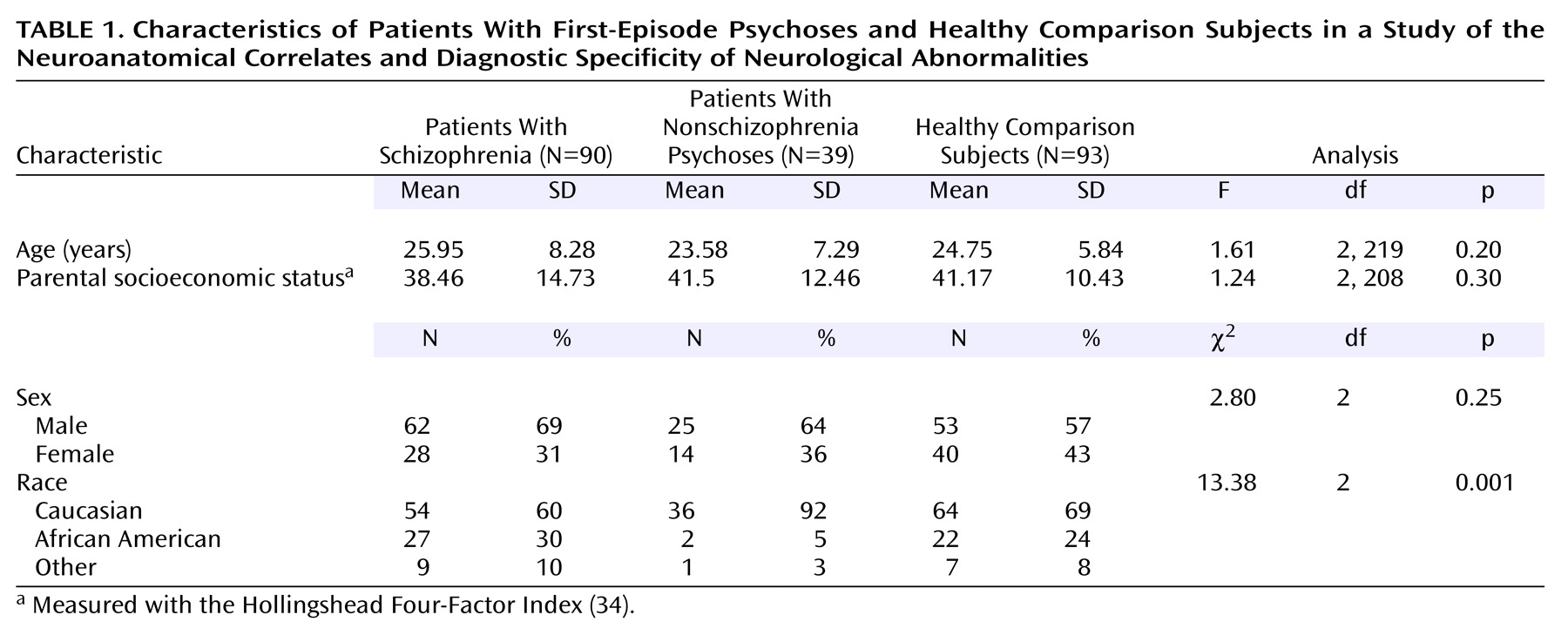
Patients were recruited from the inpatient and outpatient services of the Western Psychiatric Institute and Clinic, Pittsburgh, for a longitudinal study of first-episode psychosis
(31). Patients with previously untreated psychosis were evaluated by trained clinicians using the Structured Clinical Interview for DSM-IV, Patient Edition (SCID-P)
(32) and medical, neurological, and psychiatric assessments. Patients who were judged by the treating psychiatrist to be too ill to understand the study and competently provide informed consent were not approached to participate in the study. Patients were eligible to enter the study if they were aged 15–45 years, had an IQ >75, met the DSM-IV criteria for a psychotic disorder, and had no prior treatment with neuroleptics, no significant medical or neurological illness, no history of head injury with loss of consciousness temporally related to psychosis onset, and no current substance abuse or dependence. DSM-IV diagnoses were derived in consensus diagnostic evaluations by the raters and senior clinicians, including the first author, on the basis of all available clinical data and the SCID-P interview data. Ninety subjects were given a diagnosis of schizophrenia (N=74), schizophreniform disorder (N=3), or schizoaffective disorder (N=13), and 39 were given a diagnosis of another psychotic disorder, including psychotic depression (N=12), bipolar disorder with psychosis (N=11), delusional disorder (N=3), and psychosis not otherwise specified (N=13). All diagnoses were formally confirmed after at least 6 months of follow-up.
Ninety-three healthy comparison subjects were recruited by means of advertisements in local neighborhoods and communities in which the patients resided. After participating in an initial telephone screening, these subjects were interviewed with the Structured Clinical Interview for DSM-IV–Non-Patient Edition (SCID-I/NP)
(33) by a psychiatrist (the first author) or a clinical psychologist. The comparison subjects had no current or past axis I disorder, no prior exposure to any psychotropic medication within 6 months of the baseline assessment, no history of neurological disorders or any other chronic medical problems with potential to influence neurological function, no mental retardation (IQ <75), and no reported history of schizophrenia or major mood disorder in first-degree relatives. The healthy comparison subjects and the patients did not differ in age, gender, or parental socioeconomic status as measured by the Hollingshead Four-Factor Index
(34). However, significantly more patients with nonschizophrenia psychoses were Caucasian, compared with the patients with schizophrenia and the healthy comparison subjects (
Table 1). After receiving a full explanation of the study, all patients and comparison subjects provided written informed consent.
Neurological Evaluation
Neurological evaluations were carried out by using a modified version of the Neurological Evaluation Scale
(29). The evaluations were done before patients began taking antipsychotic medication. The Neurological Evaluation Scale consists of 29 items, most of which are rated 0, 1, or 2 (strictly dichotomous measures are scored 0 or 2). The Neurological Evaluation Scale was administered in its entirety according to the original instructions of the instruments’ developers
(29) by a trained clinician who was unaware of the consensus diagnoses. The examination was supplemented by testing for the palmomental reflex
(35). The clinicians who conducted these assessments had established interrater reliability, as previously described
(36), and periodically conducted assessments together to avoid rater drift. The reliability studies involving these examiners had determined that 13 items from the supplemented scale showed adequate frequency of abnormality (>10%) and consistently adequate interrater reliability with several groups of psychotic patients and combinations of raters
(36). Only these 13 items were entered into the primary analyses reported here. For items measured bilaterally, only the higher of the two ratings was included in the analysis. Lateral dominance items (handedness, etc.) were not considered tests of performance and were therefore omitted from the analysis. Like a previous study conducted by our group
(30), the current study included a heterogeneous group of unmedicated patients with schizophrenia and with nonschizophrenia psychoses, and the 13 Neurological Evaluation Scale variables from all patients were entered into a principal-components analysis with varimax normalized rotation. Scores derived by summing the factor components were the primary dependent variables in the current analysis.
Neuroimaging Studies
MRI and Neurological Evaluation Scale data were available for 17 schizophrenia patients (12 male and five female patients; mean age=29.53 years, SD=8.46), nine patients with nonschizophrenia psychoses (five male and four female patients; mean age=22.44 years, SD=5.43), and 18 healthy comparison subjects (13 male and five female subjects; mean age=23.94 years, SD=5.37). MRI scans were conducted by using a 1.5-T Signa whole- body GE Scanner (General Electric Medical Systems, Milwaukee). Regional brain volumes were measured with three-dimensional spoiled gradient recall acquisition in the steady-state pulse sequence, which obtained 2.6-mm thick contiguous axial images (echo time=20 msec, repetition time=40 msec, acquisition matrix=256×192, field of view=20 cm, flip angle=10°). To facilitate image orientation, axial slices were obtained parallel to the anterior commissure–posterior commissure line. NIH Image software (version 1.55)
(37) was used to measure brain anatomy. None of the MRI scans in this data set showed motion and magnetic field inhomogeneity artifacts. All measurements were conducted by a trained and reliable rater who was blind to the clinical data and to the subjects’ groups. Interrater and intrarater reliabilities, measured on 10 scans completed about a month apart, were adequate for all the measured brain structures (intraclass r>0.90).
Our approach to measuring caudate volumes has been described earlier
(20). The heteromodal association cortex volume was computed by summing the volumes of the dorsolateral prefrontal cortex, the superior temporal gyrus, and the inferior parietal cortex. The dorsolateral prefrontal cortex and superior temporal gyrus were measured by using methods described previously
(38,
39). To measure the inferior parietal cortex, a ring was drawn in the subcortical white matter 2 cm from the cortical surface in axial slices
(40). The hemispheres were divided into four regions by using three perpendicular lines drawn across the interhemispheric fissure at the most posterior point of the genu, the most posterior point in the splenium of the corpus callosum, and the midpoint between these two points. The parietal cortex was defined as the third most anterior cortical region, with the uppermost slice showing the lateral ventricles as the upper limit and the lowermost slice showing the corpus callosum as the lower limit. To measure cerebellar volumes, consecutive axial slices showing cerebellar structures were measured. Total volumes were measured for the left and right cerebellar hemispheres, including the brainstem, midbrain, and cerebellar vermes.
Data Analyses
Data were analyzed by using analysis of variance (ANOVA), with diagnosis as the grouping variable and the neurological examination abnormalities measures (the factor scores and the total score for the 13 reliable items) as dependent variables. When the ANOVA yielded significant results, Newman-Keuls post hoc tests were used to examine the specific group differences. Correlational analyses were done by using Spearman correlations and partial correlations. Two-tailed significance tests were used throughout, and alpha was set at p<0.05. Bonferroni corrections were applied to address multiple comparisons.
Results
Factor Analysis
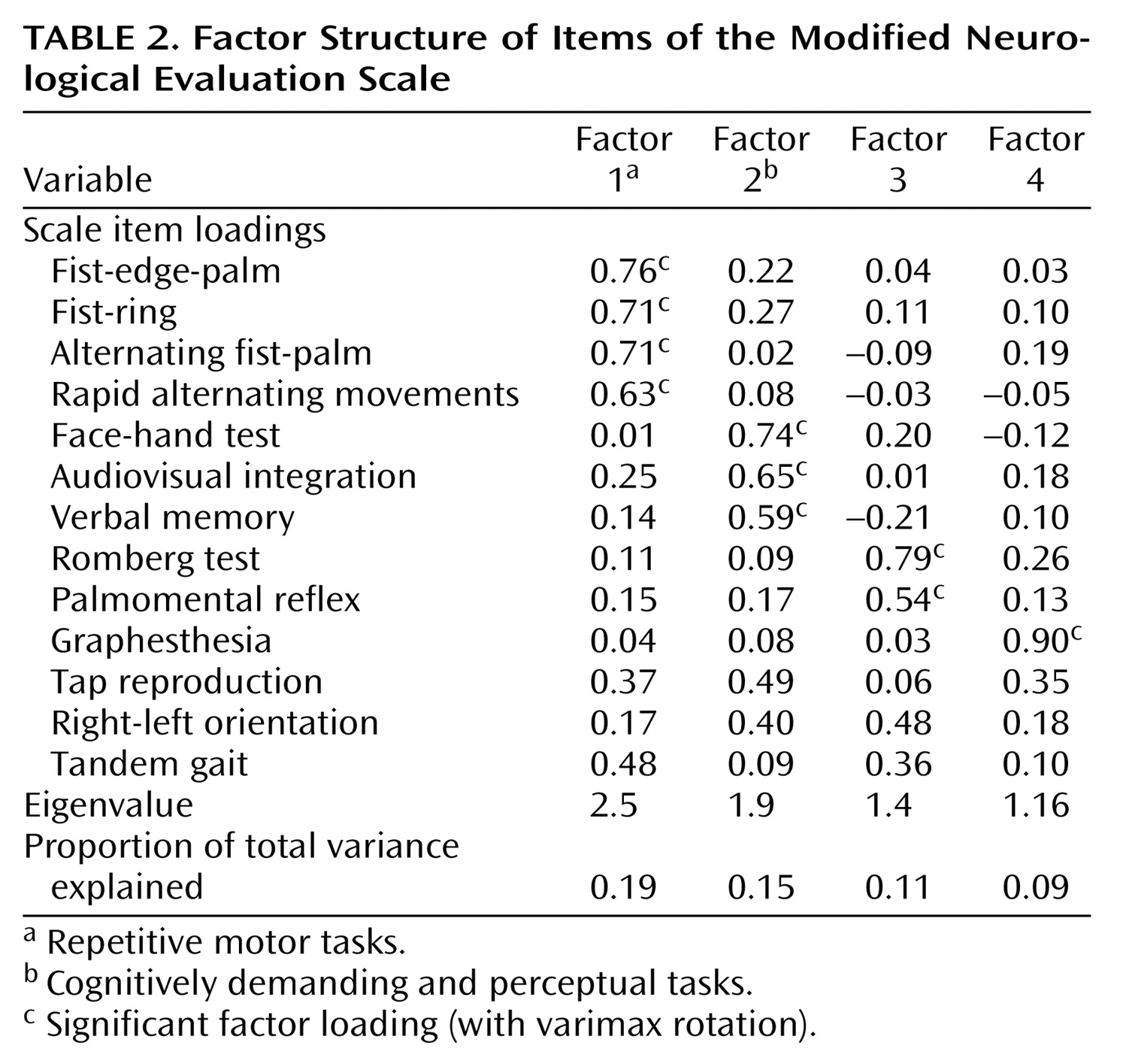
The principal-components analysis revealed four factors with eigenvalues >1 (
Table 2). The first consisted of tests requiring repetitive hand movements (fist-ring, fist-edge-palm, alternating fist-palm, and rapid alternating movements). The second factor consisted of tasks that were more complex and more cognitively demanding and that involved sensory processing (verbal memory, audiovisual integration, and face-hand test). The Romberg test and the palmomental reflex test loaded onto a third factor, and the graphesthesia test loaded onto a fourth. Tandem gait, tap reproduction, and right-left orientation did not load strongly or selectively on any factor. Together, the four factors accounted for 54% of the total variance.
Neurological Evaluation
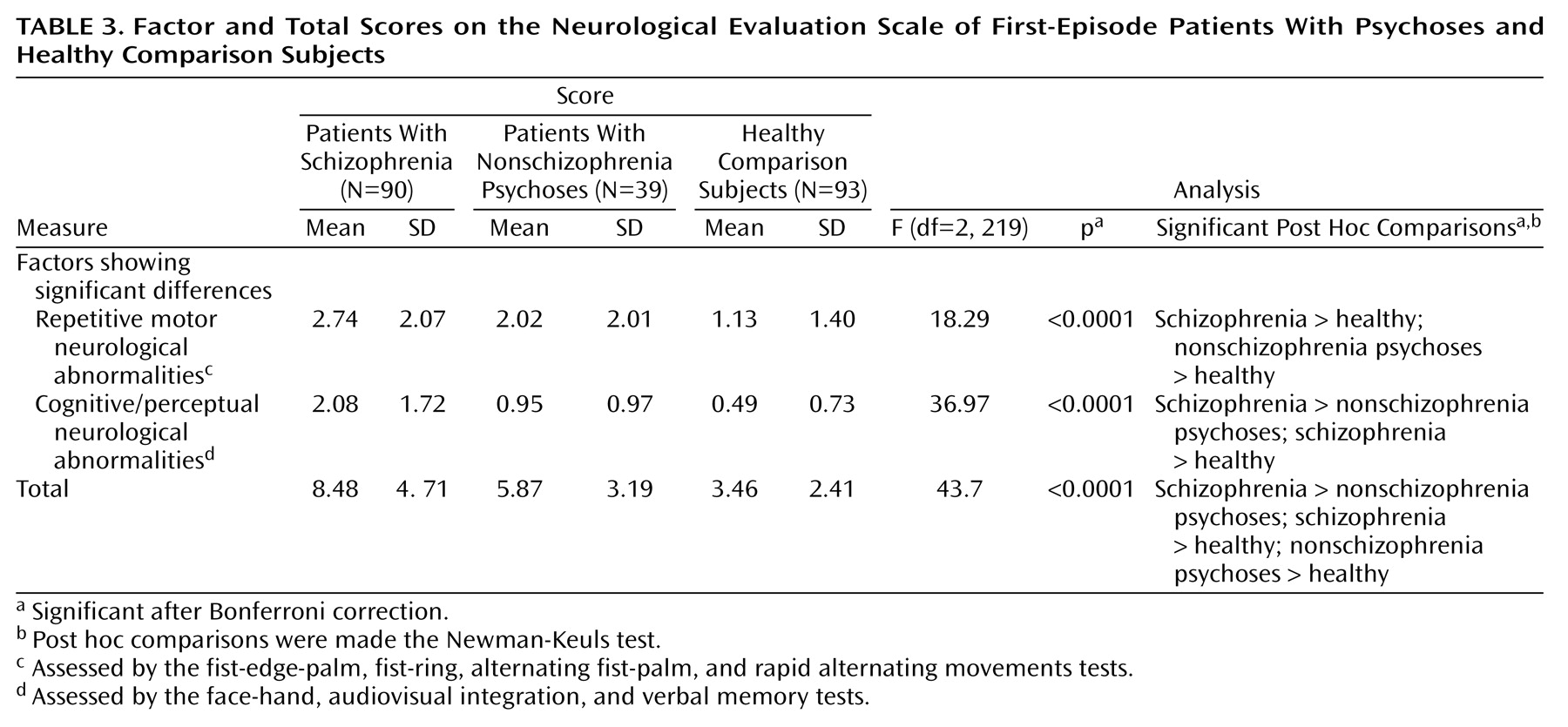
Group comparisons by ANOVA (
Table 3) were highly significant for the two primary factors but not statistically significant for the third and fourth factors (p>0.10). Post hoc comparisons of groups (
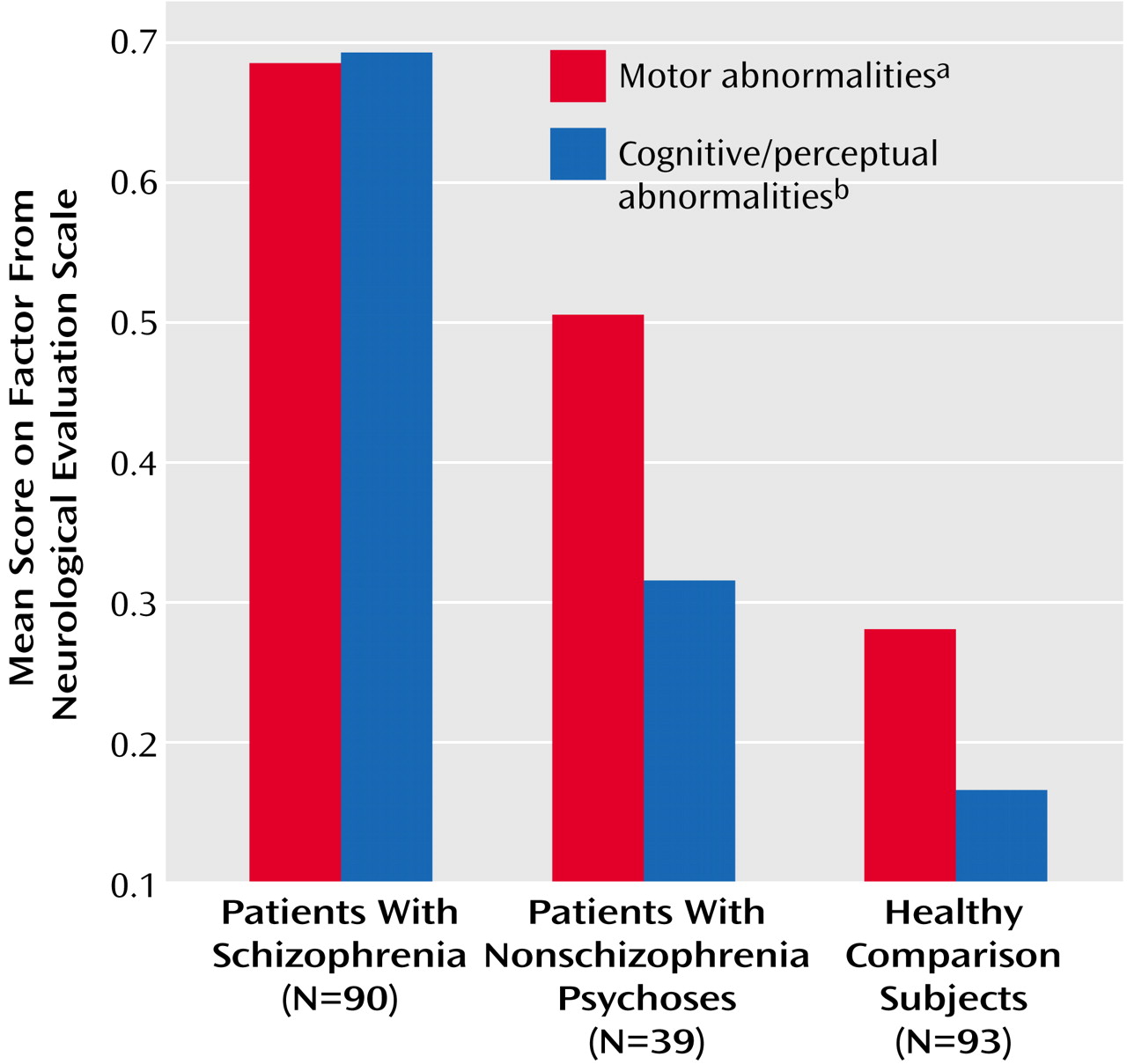
Table 3) showed significant relative impairment of motor tasks (tests for repetitive motor neurological abnormalities) in both patient groups and impairment in the cognitively demanding perceptual tasks in only the schizophrenia group. A repeat of the analysis using average Neurological Evaluation Scale factor scores instead of total scores showed the same findings (
Figure 1). Inclusion of tap reproduction and right-left orientation in the motor abnormalities factor, as suggested by a previously described factor solution for data from a larger, heterogeneous group of schizophrenia patients
(30), also yielded the same results. The conceptually derived sensory integration subscale (face-hand test, audiovisual integration, right-left orientation, stereognosis test, and graphesthesia test)
(34) also showed a significant group effect (F=18.57, df=2, 219, p<0.0001); post hoc comparisons of patient groups showed significant differences, with the schizophrenia patients having more neurological examination abnormalities than the patients with nonschizophrenia psychoses (p=0.003, Newman-Keuls).
Because there were fewer Caucasian subjects among the schizophrenia patients than among the patients with nonschizophrenia psychoses, we compared factor scores for Caucasian, African American, and other ethnic group subjects. The cognitive/perceptual neurological abnormalities factor score was significantly higher in the non-Caucasian patients (F=4.07, df=2, 219, p=0.01). Restricting the comparison of diagnostic groups to Caucasian subjects (the number of non-Caucasian patients was insufficient for diagnostic group comparisons) yielded the same results as described earlier. Excluding the schizoaffective disorder group also yielded the same results. Analysis of covariance, with age as a covariate, also had no effect on group differences in neurological examination abnormalities.
Neuroanatomical Correlates of Neurological Examination Abnormalities
In the schizophrenia group, scores for the repetitive motor neurological examination abnormalities factor correlated significantly with left caudate (rs=–0.54, N=17, p=0.03) and cerebellum (rs=–0.6, N=16, p=0.01) volumes. Cognitive/perceptual neurological abnormalities factor scores correlated with left caudate (rs=–0.61, N=15, p=0.02), cerebellum (rs=–0.67, N=16, p=0.005), and left heteromodal association cortex (rs=–0.67, N=16, p=0.005) volumes. The left heteromodal association cortex and cerebellum volumes were significantly correlated with cognitive/perceptual neurological abnormalities factor scores after Bonferroni correction. None of these correlations was significant in the healthy comparison group or the group with nonschizophrenia psychoses. The correlation of cognitive/perceptual neurological abnormalities factor scores with left heteromodal association cortex volumes persisted after correction for intracranial volume.
We also examined the relationships between the components of heteromodal association cortex volume (dorsolateral prefrontal cortex, superior temporal gyrus, and inferior parietal cortex volumes) and the Neurological Evaluation Scale measures. Repetitive motor neurological abnormalities factor scores did not correlate with the volumes of any of the heteromodal association cortex components. On the other hand, cognitive/perceptual neurological abnormalities factor scores correlated significantly with left dorsolateral prefrontal cortex volumes (rs=–0.51, N=16, p=0.04) and with right superior temporal gyrus volumes (rs=–0.55, N=16, p=0.03) in the schizophrenia group. Neither of these correlations was significant in the nonschizophrenia psychoses group or the comparison group.
Discussion
Our main observations are that repetitive motor task performance was impaired in both schizophrenia patients and patients with nonschizophrenia psychoses and that repetitive motor task performance did not distinguish between the two patient groups. On the other hand, cognitively demanding and perceptual tasks were markedly more impaired in schizophrenia patients than in either the patients with nonschizophrenia psychoses or the healthy comparison subjects. Thus, neurological examination abnormalities may have some measure of diagnostic specificity among the idiopathic psychoses. This observation is of clinical relevance and may help clarify the pathophysiological basis of schizophrenia, as contrasted with the other psychoses.
Our findings of a higher rate of cognitive/perceptual neurological examination abnormalities in schizophrenia and of a higher rate of motor abnormalities in both groups of patients with psychoses are somewhat consistent with previous findings. Schizophrenia patients have been found to perform similarly to patients with psychotic depression on motor
(41) and pursuit eye movement tasks
(42). Patients with schizophrenia and patients with schizoaffective disorder have been found to be more impaired in “parietal” tasks (those measured by the stereognosis, double-simultaneous stimulation, construction, and logical-grammatical relationships tests) but not in motor tasks, compared to patients with psychotic mood disorders
(43–
45). Not all studies have found such diagnosis effects
(8,
46). Nevertheless, our observations raise the possibility that cognitively demanding perceptual components of the neurological examination have neuropsychological parallels in schizophrenia and may be related to neurobiological impairments in specific brain circuits that mediate these functions. Validating this point, we found that MRI measures had a distinct pattern of relationships to the two predominant neurological examination abnormalities factors.
Few studies have examined the neuroanatomical correlates of neurological examination abnormalities in schizophrenia by looking at specific structural-neurological relationships. Two groups
(47,
48) found smaller basal ganglia volumes to be associated with motor deficits. In our data, cognitive/perceptual neurological examination abnormalities were significantly and strongly correlated with smaller volumes in the left heteromodal association cortex and the cerebellum and were less strongly correlated with caudate volumes. On the other hand, motor abnormalities tended to be correlated with smaller right and left caudate and cerebellar volumes but not with heteromodal association cortex volumes. Thus, our MRI findings suggest that specific domains of impaired neurological functions observed in first-episode schizophrenia patients may stem from somewhat distinct neuroanatomical alterations in schizophrenia. This view is consistent with our observations that the cognitive/perceptual neurological abnormalities factor correlated more strongly than the motor abnormalities factor with neuropsychological assessments that tap into executive function and memory
(49). On the other hand, the motor abnormalities seen in the patients with nonschizophrenia psychoses may be associated with cerebellar and striatal involvement and a relative preservation of the association cortex. The lack of such correlations in the data for the patients with nonschizophrenia psychoses in our study suggests that the observed neurological-anatomical relations are specific to schizophrenia.
The strengths of this study included the relatively large number of well-characterized, neuroleptic-naive psychotic subjects; the use of only reliable items from the Neurological Evaluation Scale in the evaluations; and the use of factor analyses to reduce the number of neurological tests and the number of pathophysiologically meaningful dimensions derived from the tests. In addition, both a well-matched healthy comparison group and a group of first-episode patients with nonschizophrenia psychoses were examined. However, all patients in the study were well enough to provide informed consent and to tolerate extensive testing and interviews before treatment; thus, the patients in the study may not be representative of patients with never-treated psychoses. As stated earlier, examiners were often aware of subjects’ symptoms and histories and therefore may not have been blind to clinical data. However, they were not aware of the consensus research diagnoses that were confirmed weeks after their evaluations, and the schizophrenia-specific finding was not anticipated. The psychotic mood disorder group was restricted to patients with mood-incongruent psychotic features. This limitation may not have weakened the finding of diagnostic specificity but may have diminished the representativeness of the group with nonschizophrenic psychoses. Finally, the significant intersubject variability in the anatomical landmarks made precise partition of the heteromodal association cortex difficult; the geometrical partitioning approach we used may not be neuroanatomically precise, but it has good reliability.
Although replication of the study findings with larger, more heterogeneous groups of patients is needed, our findings provide new evidence suggesting that the so-called “soft” neurological signs have some measure of diagnostic specificity and neuroanatomical validity within the broad group of idiopathic psychoses. These observations have implications for further etiological and pathophysiological research in schizophrenia, as some evidence suggests that “integrative” neurological examination abnormalities may be useful in defining genetic risk for schizophrenia
(50). Because these bedside measures are relatively inexpensive and convenient to use, they may be valuable as part of neuropsychiatric assessments in both developed and developing countries.