Neuronal apoptosis is highly active during early neurodevelopment. It is estimated that, depending on the region, 20%–80% of all neurons formed in the CNS die by apoptosis in early life
(1). Apoptosis is regulated by several protein families, including the upstream Bcl-2 family (e.g., the antiapoptotic Bcl-2 and proapoptotic Bax) and the downstream caspase family (e.g., caspase-3). Pathological activation of neuronal apoptosis and abnormal expression of apoptotic regulatory proteins have been demonstrated in classic neurodegenerative disorders, including Alzheimer’s and Parkinson’s diseases
(2–
5). Apoptosis has also been hypothesized to contribute to the pathophysiology of schizophrenia
(6), a neurodevelopmental disorder that may encompass limited neurodegenerative features
(7). Postmortem evidence supports apoptotic involvement in schizophrenia; low levels of Bcl-2 have been demonstrated in Brodmann’s area 21 (middle temporal gyrus)
(8). It is interesting that this cortical area is also associated with low gray matter volume
(9), similar to low volumes in other temporal and frontal areas and in total cortical gray matter
(10,
11). Low levels of Bcl-2 in Brodmann’s area 21 may signal a role for apoptotic proteins in the mechanisms underlying the low gray matter volumes in schizophrenia.
The activation of apoptosis is controlled at multiple checkpoints within the cell. Upstream, the proapoptotic Bax and antiapoptotic Bcl-2 are membrane-bound pore-forming proteins that interact through heterodimerization. Together they regulate the mitochondrial transmembrane passage of cytochrome c, which in turn activates caspase proteins. The Bax/Bcl-2 ratio appears more important than the individual Bax or Bcl-2 level in determining a cell’s vulnerability to apoptosis; high Bax/Bcl-2 ratios lead to greater apoptotic activity
(12). Neuronal apoptosis in the CNS is significantly lower than normal in Bax-deleted mice and in Bcl-2-overexpressing mice, while in Bcl-2-deleted mice and in Bax-overexpressing mice, apoptosis is greater than normal
(13). Furthermore, in rat cortical development, the Bax/Bcl-2 ratio is highest during the first 2 postnatal weeks, which corresponds to the most active interval of neurodevelopmental apoptosis, while the Bax/Bcl-2 ratio decreases during normal aging, when neuronal apoptosis is rare
(13,
14). A high Bax/Bcl-2 ratio has been demonstrated in the cortex of brains from patients with Down’s syndrome
(15) and in human neuronal cultures treated with amyloid β, a model for Alzheimer’s disease
(16).
Caspase proteins are cysteine proteases that act downstream of the Bcl-2 family by initiating cellular breakdown during apoptosis
(17). Initiator caspases (e.g., caspase-8 and caspase-9) serve to activate effector caspases (e.g., caspase-3, caspase-6, and caspase-7). Among the effector caspases, caspase-3 is most frequently involved in neuronal apoptosis
(18). Cleavage of inactive procaspase-3 protein into active caspase-3 subunits is a marker of apoptotic activity
(19), and levels of activated caspase-3 subunits are significantly higher than normal in both the cortex of patients with Alzheimer’s disease
(4) and the substantia nigra of patients with Parkinson’s disease
(5). Thus, measurement of caspase-3 can yield information about ongoing neuronal apoptosis and provide a pathophysiological comparison to classic neurodegeneration.
This is a continuing investigation of apoptotic proteins in the temporal cortex of schizophrenia patients. It has been demonstrated that in these patients the temporal cortex, including Brodmann’s area 21, has a low volume of gray matter
(9,
20–22), experiences progressive volume loss in early stages of the illness
(23,
24), and is involved in certain clinical manifestations of schizophrenia, including auditory hallucinations
(20,
25). Western blot analysis was used to measure Bax, Bcl-2, procaspase-3, and activated caspase-3 subunit levels in blocks of Brodmann’s area 21. The primary hypothesis was that the Bax/Bcl-2 ratio would be high in patients with schizophrenia.
Results
Bax and Bcl-2 Immunoblots
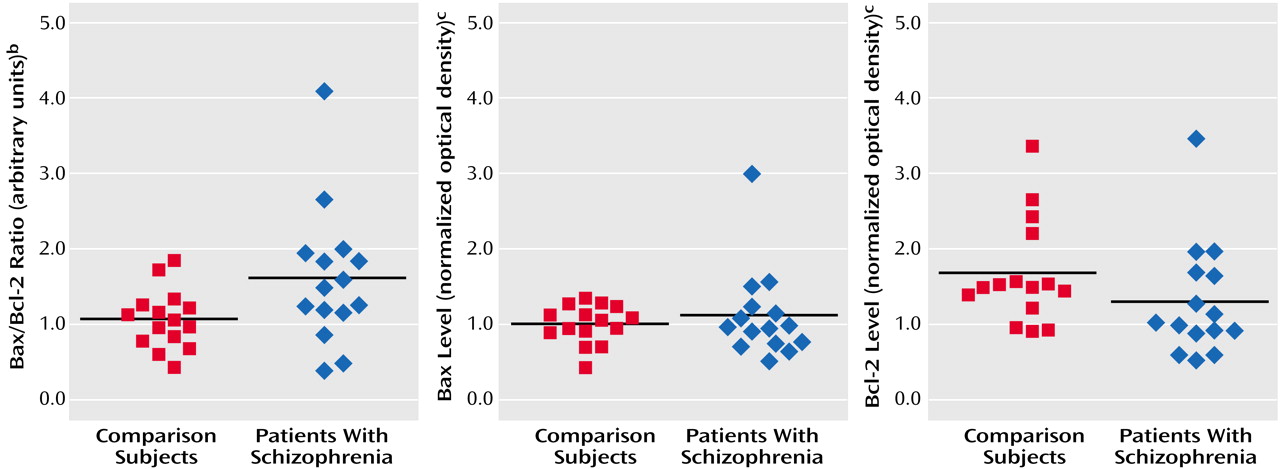
The mean Bax/Bcl-2 ratio was 50% higher in the schizophrenia group (mean=1.65, SD=0.90) than in the nonpsychiatric comparison group (mean=1.10, SD=0.37) (
Figure 1 and
Figure 2). Individually, the mean Bax level in the patients with schizophrenia (mean=1.18 normalized optical density, SD=0.59) did not differ from that of the comparison subjects (mean=1.06, SD=0.25) (t=0.72, df=28, p=0.48) (
Figure 2), and the mean Bcl-2 level of the schizophrenia group (mean=1.39, SD=0.75) did not differ from that of the comparison group (mean=1.75, SD=0.69) (t=1.35, df=28, p=0.19) (
Figure 2). It was the effect of differences in the Bax and Bcl-2 levels and the resulting Bax/Bcl-2 ratio for each individual subject that produced a substantial difference in Bax/Bcl-2 ratio between the two groups.
There was a nearly significant group effect on the mean Bax/Bcl-2 ratio across all psychiatric groups according to ANOVA (F=2.36, df=3, 59, p=0.09). The mean ratios for the patients with bipolar disorder (mean=1.18, SD=0.71) and major depression (mean=1.12, SD=0.52) were close to that of the nonpsychiatric comparison subjects (mean=1.10, SD=0.37). The ratio did not correlate with age across all groups (r2=0.01, N=60, p=0.53) or with lifetime fluphenazine-equivalent dose in the schizophrenia group (r2=0.16, N=15, p=0.14). The Bax/Bcl-2 ratio also did not differ between women (mean=1.11, SD=0.53) and men (mean=1.37, SD=0.75) (t=1.46, df=58, p=0.15) or between the left (mean=1.34, SD=0.57) and right (mean=1.20, SD=0.76) (t=0.80, df=58, p=0.43) sides of the brain across all samples.
Caspase-3 Immunoblots
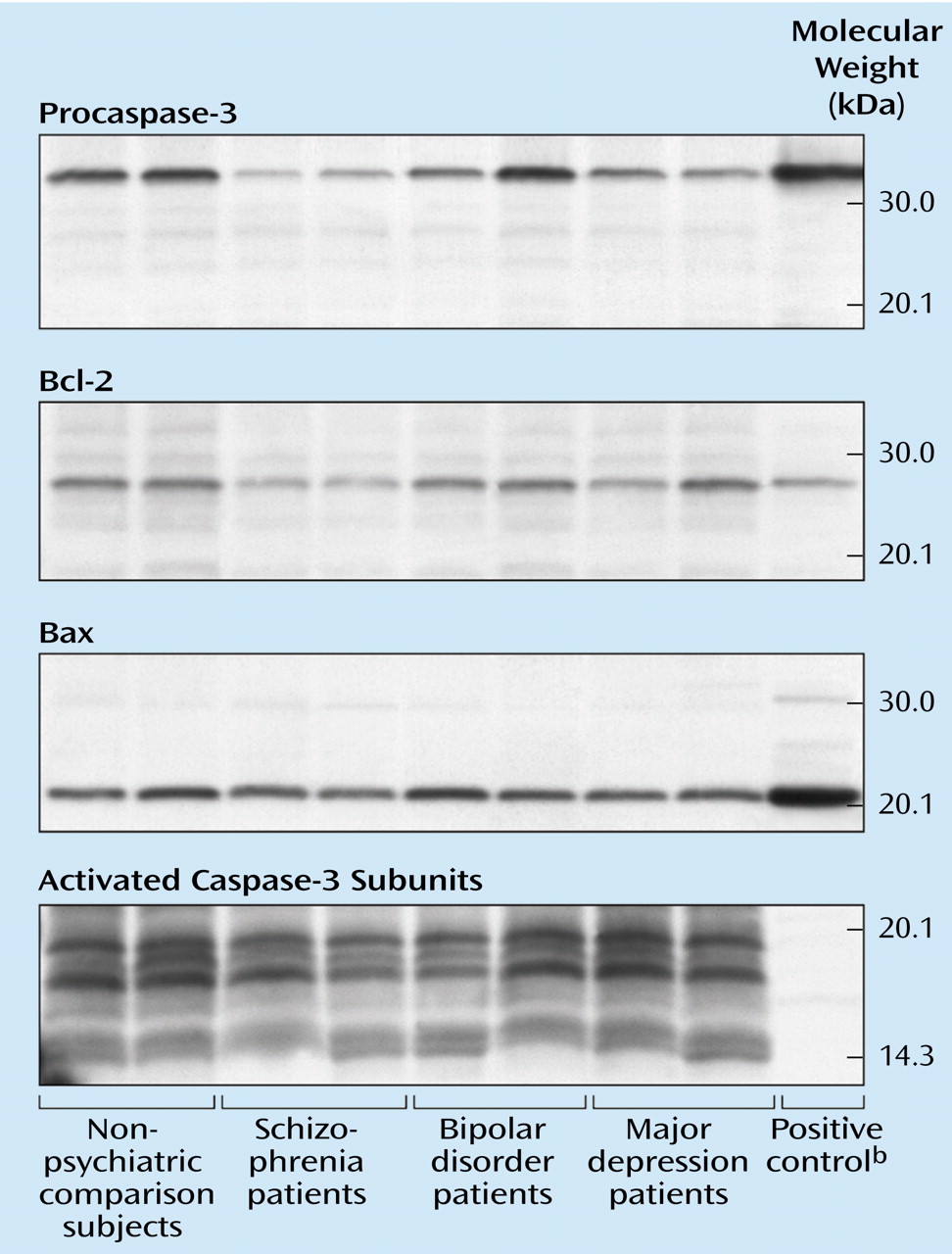
The immunoblots exposed to activated caspase-3 antibodies revealed two large-fragment bands migrating to 20 and 17 kDa (
Figure 1). Three isoforms of the large caspase-3 subunit have previously been described at 20, 19, and 17 kDa
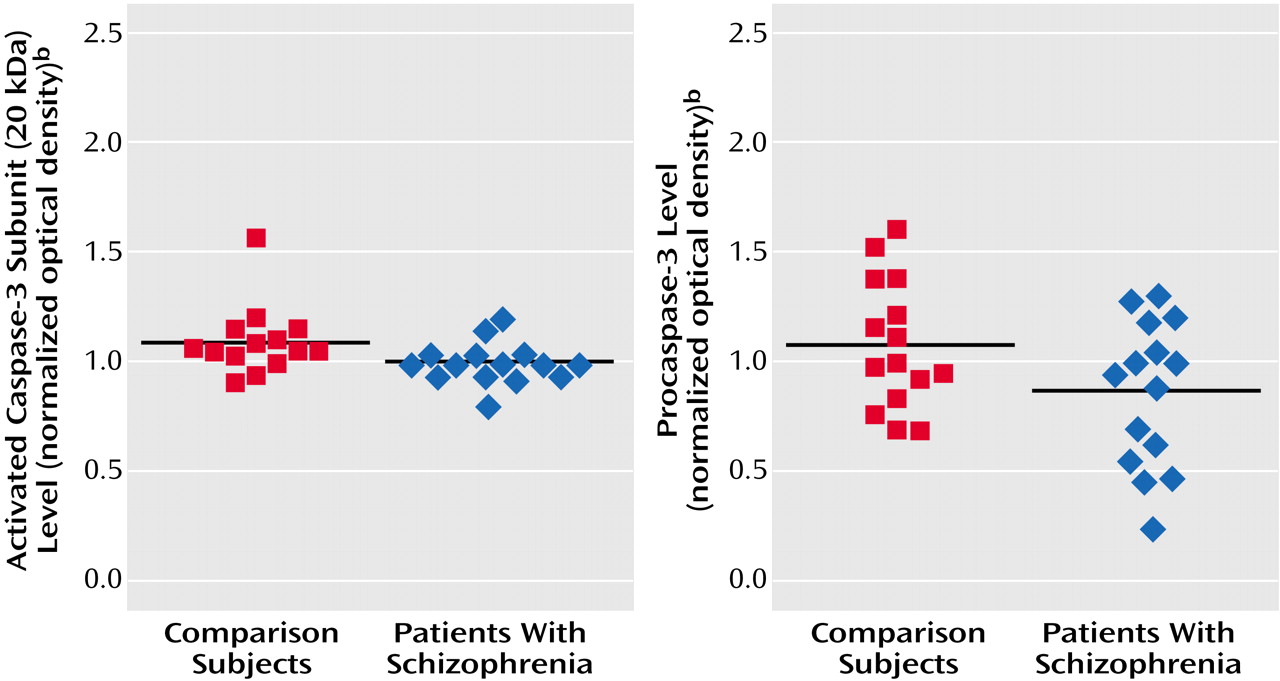
(28). Among these bands, the 20-kDa isoform was most readily quantifiable on our gels, and it was 8% lower in the schizophrenia group (mean=1.01 normalized optical density, SD=0.09) than in the nonpsychiatric comparison group (mean=1.10, SD=0.16) (t=2.00, df=27, p=0.06) (
Figure 3). The small caspase-3 subunit migrating to 12–14 kDa could not be measured reliably across all samples owing to faint expression and blot artifacts, but no qualitative group differences were evident (
Figure 1). Although the difference was not quite significant, the procaspase-3 levels also appeared somewhat lower in the schizophrenia group (mean=0.87 normalized optical density, SD=0.33) than in the nonpsychiatric comparison group (mean=1.08, SD=0.29) (t=1.87, df=28, p=0.08) (
Figure 1 and
Figure 3).
The mean values for the 20-kDa caspase-3 subunit for the patients with major depression (mean=1.03 normalized optical density, SD=0.09) and bipolar disorder (mean=1.01, SD=0.21) did not produce a group effect by ANOVA (F=1.29, df=3, 58, p=0.29). The 20-kDa caspase-3 values did not correlate with age across all groups (r2=0.00, N=59, p=0.89) or with lifetime fluphenazine-equivalent dose in the schizophrenia group (r2=0.00, N=14, p=0.84). They also did not differ between women (mean=1.00, SD=0.12) and men (mean=1.06, SD=0.16) (t=1.43, df=57, p=0.16) or between the left (mean=1.05, SD=0.17) and right (mean=1.03, SD=0.13) (t=0.57, df=57, p=0.58) sides of the brain across all groups. Finally, the procaspase-3 levels of the patients with major depression (mean=0.90 normalized optical density, SD=0.34) and bipolar disorder (mean=0.94, SD=0.46) did not produce a group effect by ANOVA (F=1.01, df=3, 59, p=0.40).
Postmortem Stability of Bax and Caspase-3
The Western blots of rat cortex revealed high postmortem stability of the Bax and caspase-3 proteins over 24 hours. The mean Bax level in the rat cortex 24 hours postmortem (mean=0.20 optical density, SD=0.02) was not significantly different from that at 0 hours postmortem (mean=0.22, SD=0.02) (t=1.79, df=6, p=0.13). Likewise, the mean value for the caspase-3 20-kDa subunit at 24 hours postmortem (mean=0.22 optical density, SD=0.01) was unchanged from that at 0 hours (mean=0.22, SD=0.00) (t=1.10, df=5, p=0.33).
Haloperidol Effects on Bax, Bcl-2, and Caspase-3
The mean Bax/Bcl-2 ratio in the frontal cortex of the haloperidol-treated rats (mean=0.88, SD=0.12) was not significantly different from that of the rats given the saline control (mean=0.99, SD=0.18) (t=1.47, df=11, p=0.17). There was also no significant difference in the mean Bax level between haloperidol (mean=0.99 normalized optical density, SD=0.10) and saline (mean=1.10, SD=0.23) (t=1.14, df=11, p=0.28) or in the mean Bcl-2 level between haloperidol (mean=0.89, SD=0.06) and saline (mean=0.86, SD=0.06) (t=1.01, df=11, p=0.34). The mean level for the 20-kDa caspase-3 subunit also did not differ between haloperidol (mean=1.01 normalized optical density, SD=0.28) and saline (mean=1.04, SD=0.31) (t=0.21, df=11, p=0.85).
Discussion
In this study, the mean Bax/Bcl-2 ratio in Brodmann’s area 21 was 50% higher in patients with schizophrenia than in nonpsychiatric comparison subjects. This finding provides new evidence that apoptotic proteins contribute to the underlying pathophysiology of schizophrenia. A high Bax/Bcl-2 ratio has been found to increase the vulnerability of neurons to apoptosis
(12), and a high ratio occurs in several classic neurodegenerative disorders characterized by progressive neuronal loss
(15,
16). In schizophrenia, this finding may be particularly relevant to the evidence for progressive gray matter loss, as seen most consistently in early stages of the disorder
(22–
24,
29,
30).
The individual mean levels of Bax and Bcl-2 in the schizophrenia patients did not differ from those of the comparison subjects. Using ELISA, we previously detected 25% less Bcl-2 in schizophrenia patients than comparison subjects in Brodmann’s area 21
(8). Numerically, the Bcl-2 level was 21% lower in the schizophrenia patients in the current study, but the lack of significance probably reflects the lower sensitivity of Western blot compared to ELISA. It was necessary to reanalyze Bcl-2 with a different method—Western blot, in triplicate—in the current study in order to derive the Bax/Bcl-2 ratio since a Bax ELISA was unavailable. Furthermore, Western blot has the advantage of quantification and protein identification within one technique, while ELISA does not allow direct visualization of protein bands.
In contrast to the high Bax/Bcl-2 ratio, neither the activated caspase-3 nor procaspase-3 level was statistically different from that of the nonpsychiatric comparison subjects, although they both were modestly lower. This result may indicate that apoptosis is not active in Brodmann’s area 21 in chronic schizophrenia. However, this interpretation is qualified by several considerations. First, apoptosis may be active only during distinct phases of illness. For example, if apoptotic activity is limited to prodromal and new-onset schizophrenia, when progressive gray matter loss has been identified, then the current study using tissue from patients with chronic illness would miss such activity. Second, a high Bax/Bcl-2 ratio may reduce neuronal viability in the absence of caspase-3 activity and/or involve other effector caspases. It is interesting that the somewhat lower caspase-3 levels may have relevance to a recent study demonstrating a subset of neurons with fewer single-stranded DNA breaks in the cingulate cortex of patients with schizophrenia
(31). In contrast, consistently higher procaspase-3 and activated caspase-3 levels in Alzheimer’s and Parkinson’s diseases
(4,
5) suggest that the pathophysiology of schizophrenia is distinct from classic neurodegeneration.
The current findings raise several other considerations for the pathophysiology of schizophrenia. First, schizophrenia is increasingly conceptualized as a disorder of the synapse, given converging lines of evidence from microarray, neuropathological, and neuroimaging techniques
(32,
33). Furthermore, several studies suggest that synaptic remodeling and elimination may be influenced by apoptotic protein activity in a process termed “synaptic apoptosis”
(34,
35). Although this process is speculative, if synaptic loss in schizophrenia is mediated by apoptotic protein activity and is episodic rather than continuous (e.g., during adolescent synaptic pruning or in response to discrete episodes of excitotoxic stress), then the caspase-3 level may be increased only transiently and then return to baseline. Second, the data do not rule out an early neurodevelopmental role for apoptosis in schizophrenia. A fetal or perinatal insult could transiently alter normal developmental apoptosis to yield enduring cytoarchitectural deficits. It is interesting that multiple proapoptotic stimuli, such as ischemia, hypoxia, and pro-inflammatory cytokines
(36,
37), have also been implicated as insults during early life that increase the risk of developing schizophrenia
(38–
40). Studies of the normal developmental pattern of apoptotic protein expression in the human frontal cortex have revealed that infancy is the period of highest Bax levels and lowest Bcl-2 levels across the lifespan
(27,
41). This suggests that neuronal apoptosis during the perinatal period may be especially vulnerable to dysregulation from neurodevelopmental insults.
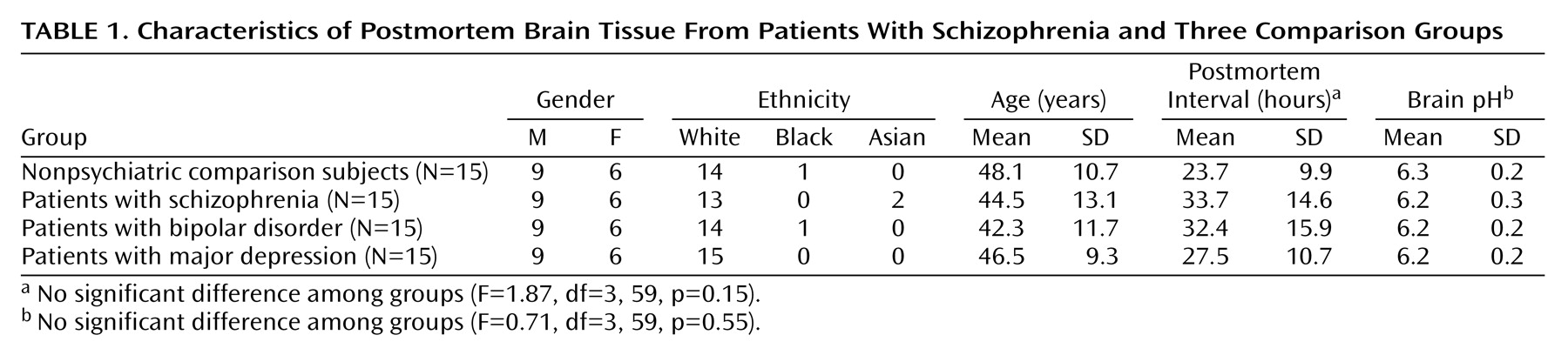
All of the subjects in this study were group matched for age, gender, postmortem interval, side of brain, and brain pH, limiting the potential confounding effects of these variables. Although chronic antipsychotic treatment may alter the expression of apoptotic proteins, one week of haloperidol treatment in animals did not produce changes. Furthermore, cumulative antipsychotic medication exposure did not correlate with the Bax/Bcl-2 ratio or caspase-3 level. Postmortem interval could affect results if apoptotic proteins demonstrated poor postmortem stability. However, the study of postmortem stability showed no change in Bax, procaspase-3, or activated caspase-3 (20-kDa subunit) over 24 hours. Similarly, high postmortem stability for Bcl-2 was previously determined
(27). Finally, patient-related factors, including diagnostic heterogeneity, substance abuse, and medical illness, could introduce unintended variability, but efforts were made to limit these factors during subject selection
(26).
This study revealed limited associations with other psychiatric diagnoses. The Bax/Bcl-2 ratio and caspase-3 level were essentially no different in the patients with bipolar disorder or major depression from those in the nonpsychiatric comparison subjects. Since substantially lower cortical volume and neuronal and glial density have been documented in affective disorders
(42,
43), this finding suggests that divergent pathophysiological mechanisms may underlie the neuropathology of schizophrenia and primary affective disorders.
In conclusion, this study indicates that apoptotic proteins may reduce neuronal viability in adult schizophrenia without inducing large-scale apoptosis. An intriguing possibility that merits further inquiry is the extent to which apoptotic proteins influence synaptic elimination and thereby contribute to neuropil loss in schizophrenia. The differential regulation of the expression of Bax, Bcl-2, and caspase-3 proteins demonstrates that the pathophysiology of schizophrenia is distinct from that of classic neurodegenerative disorders. Ultimately, if clinically relevant roles for apoptotic proteins can be demonstrated, their pharmacological manipulation may permit novel approaches to schizophrenia treatment.