Posttraumatic stress disorder (PTSD) is one of the most prevalent mental disorders
(1,
2), particularly among Veterans Administration (VA) health care enrollees
(3,
4). In the general population, PTSD is associated with high rates of smoking (45%
[5] versus the national average of 23%
[6]) and a fourfold increased risk for nicotine dependence
(7). The rates of smoking in veterans with PTSD (53%–66% [
8–
10]) are approximately double those of VA enrollees in general (30%
[11]). Moreover, a greater proportion of smokers with combat-related PTSD smoke heavily (>25 cigarettes per day) compared with veterans who smoke but do not have PTSD (48% versus 28%
[8]). PTSD is associated with a smoking quit rate of only 23%
(5), about half that of lifetime smokers without a mental disorder
(12), and falls third from the bottom in a ranking of quit rates for 13 mental disorders
(5). Smokers with PTSD also experience nicotine withdrawal symptoms in response to encounters with trauma-related stimuli
(13) and report smoking in order to relieve anxiety and tension
(8). Taken together, this research suggests a dynamic relationship between PTSD and tobacco use that argues for a coordinated approach to the treatment of both disorders.
Several pharmacological and behavioral treatments for nicotine dependence have shown efficacy in controlled clinical trials
(12,
14). However, these treatments are only as useful as the ability of health care organizations to deliver them effectively to individuals who need them most. Primary care providers only infrequently apply even brief, cost-effective tobacco-cessation interventions to smokers
(15–
18), despite the fact that 60%–70% of smokers want to quit
(6,
11). Nicotine-dependence treatment in patients with mental disorders may be particularly neglected since psychiatric patients receive cessation counseling at only 38% of primary care visits and 12% of visits with a psychiatrist
(16). Referral of patients to specialty smoking-cessation clinics is a commonly used alternative to primary care-based delivery of tobacco use treatment. However, the effectiveness of these clinics is compromised by poor patient compliance, with rates of attendance as low as 13%–14%
(19,
20) and limited capacity to provide repeated intervention for a chronic, relapsing disorder such as nicotine dependence. These limitations create a situation in which only 17% of smokers in the nation’s largest health care system (the VA) report receiving desired cessation treatment in the previous year
(11).
The simultaneous treatment of two or more interwoven disorders by a single provider team has shown promising clinical effectiveness for patients with severe mental illness and comorbid substance use disorders
(21,
22). This study accordingly tested an approach for improving the effectiveness of tobacco-cessation service delivery by integrating treatment for smoking into the routine mental health care of patients with PTSD. Specifically, this randomized, controlled clinical trial compared the effectiveness of two different methods for delivering guideline-based smoking-cessation treatment to patients receiving VA mental health care: 1) brief smoking-cessation interventions integrated with ongoing mental health care and delivered by mental health providers (integrated care) versus 2) smoking-cessation interventions delivered separately from mental health care by smoking-cessation specialists (usual standard of care). It was hypothesized that integrated care would result in higher rates of smoking cessation than would usual standard of care. A secondary objective of this study was to determine if smoking cessation was associated with worsening PTSD or depression symptoms. Anticipating this possibility is important, given the high co-occurrence of depression with PTSD
(23) and prior research showing that smoking abstinence can exacerbate depression in individuals with a history of this disorder
(24–
27).
Method
Subjects
Subjects (N=66) were recruited from the VA Puget Sound Health Care System PTSD clinic, which provides specialized outpatient treatment for chronic PTSD.
Table 1 reports demographic and smoking characteristics for the study group as well as psychometric data showing moderate to severe levels of symptoms on the PTSD Checklist
(28) and the Beck Depression Inventory
(29). Subjects in the integrated care and usual standard of care conditions did not differ significantly on baseline characteristics (all p values >0.05).
Subjects were included if they smoked ≥10 cigarettes per day, expressed a willingness to receive smoking-cessation treatment, and met DSM-IV criteria for PTSD. Exclusion criteria were 1) the use of smokeless tobacco, pipes, or cigars, 2) the presence of unstable axis I disorders, and 3) current substance dependence disorder other than tobacco use. After providing a complete description of the study, written informed consent was obtained. The subjects were then randomly assigned to integrated care (N=33) versus the usual standard of care (N=33) for smoking cessation. The rate of refusal to participate in this study was 3% for help-seeking smokers.
Treatment Conditions
All study subjects received psychotropic medications for PTSD throughout their participation in the study, as prescribed by two PTSD clinic psychiatrists and a nurse practitioner. Each subject also received psychotherapy from an assigned case manager who coordinated their mental health care. Case managers included four psychologists, one social worker, an addictions therapist, and a technician.
Integrated Care: Experimental Condition
The subjects randomly assigned to integrated care received smoking-cessation interventions administered by their assigned PTSD clinic prescriber and case manager. Integrated care was modeled after the brief clinical interventions for primary care practitioners published in the U.S. Public Health Service’s clinical practice guideline titled
Treating Tobacco Use and Dependence (14). PTSD clinic staff received approximately 3 hours of training, plus as-needed consultation in smoking-cessation treatment from the clinic director (M.M.). They then delivered interventions using a treatment manual (available upon request from the first author) that operationalized interventions for each session.
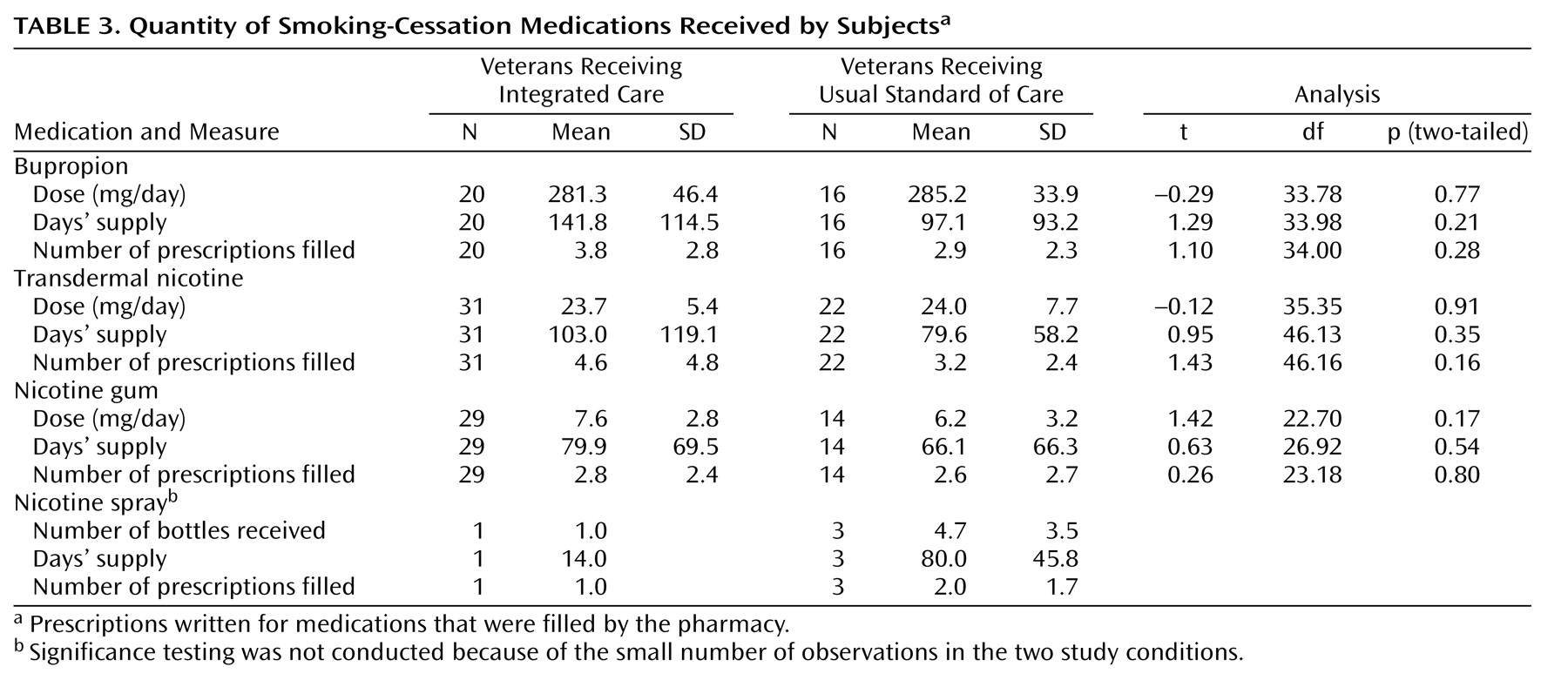
The subjects received smoking-cessation protocol medications (bupropion, transdermal nicotine, nicotine polacrilex gum, and nicotine spray) from the psychiatrist or nurse practitioner managing their pharmacological treatment of PTSD. Prescribers in the integrated care and the usual standard of care conditions agreed to adopt the practice of routinely prescribing bupropion, transdermal nicotine, and nicotine gum or spray in order to standardize the medication protocol across conditions. However, the prescribers were allowed to use their discretion to select only some of these medications, depending on patient preferences and medical contraindications.
The core behavioral counseling components of integrated care, administered by case managers, consisted of 1) the assessment of tobacco use status, abstinence history, and individualized reasons for quitting; 2) education about the health risks of smoking and the benefits of quitting; 3) advice to quit smoking; 4) application of motivational interventions for ambivalent smokers; 5) setting a date to quit; 6) behavioral counseling to help subjects prepare to quit smoking, coping with smoking urges and barriers to abstinence, and developing problem-solving skills; 7) self-help reading materials; 8) intrasession support and assistance in identifying extrasession social support; and 9) self-directed behavioral methods for reducing anxiety, consisting of a relaxation training tape and written materials on stress management. The protocol required case managers to administer five individual behavioral counseling sessions on a once-weekly basis, plus one follow-up contact (the sessions averaged 20 minutes each). These interventions were rolled into regularly scheduled visits addressing PTSD and comorbid mental disorders for subjects whose treatment plan ordinarily included individual sessions with a case manager. For subjects receiving only group therapy for PTSD, case managers scheduled separate individual smoking-cessation sessions. After delivering the six core behavioral counseling sessions, clinicians used their discretion to periodically assess smoking status and reinstate cessation treatment for subjects who relapsed.
Usual Standard of Care: Comparison Condition
The subjects randomly assigned to the usual standard of care were referred to the VA Puget Sound Health Care System’s Smoking Cessation Clinic. Nursing personnel who staffed this clinic were trained at Mayo Clinic’s Nicotine Dependence Treatment Center and had extensive experience in cessation treatment. This clinic delivered U.S. Public Health Service guideline-adherent cessation treatment
(14). Clinic providers used the same algorithm as integrated care providers for prescribing smoking-cessation medications. The subjects attended one group orientation class, followed by individual sessions in which they received medications and behavioral counseling. Unrestricted access to usual standard of care treatment sessions was provided to all subjects. That is, the number of treatment contacts received was determined by clinical recommendations of usual standard of care providers and the preferences and initiative of subjects. The subjects assigned to the usual standard of care condition received absolutely no tobacco-cessation interventions from their PTSD clinic providers.
Measures
Delivery of study treatments
The Consumer Health Information and Performances Sets, an administrative database accounting for services delivered by the VA, was used to track smoking-cessation medications actually received by the subjects. This database provided information about the proportion of subjects who filled prescriptions for study medications as well as doses for these medications. The number of smoking cessation behavioral counseling sessions delivered to the subjects was extracted from the VA’s computerized patient record system, where clinicians are required to document all patient visits.
Adherence of integrated care case managers to the treatment manual was assessed by independent review of the computerized patient record system. These records contained provider documentation of interventions delivered during each visit on specially prepared charting templates that listed a total of 31 specific protocol interventions prescribed by the manual. These interventions constituted the core behavioral counseling components of integrated care (e.g., “set and recorded a quit date,” “identified strategies for coping with withdrawal symptoms”). After each session, case managers checked off interventions delivered on the charting template and provided a narrative summary of smoking status and treatment progress. The study coordinator (D.Y.) independently reviewed chart notes for all providers and treatment sessions in order to compute clinicians’ self-recorded adherence to protocol interventions as a percentage of the total recommended. This review showed that 80% of behavioral counseling interventions prescribed by the treatment manual were reportedly administered.
Smoking outcomes
Seven-day point prevalence abstinence, verified by expired carbon monoxide (≤10 ppm), was the primary outcome measure used to compare integrated care and the usual standard of care at 2, 4, 6, and 9 months after random assignment. The period after randomization provided a consistent marker across subjects of the initiation of the treatment process. The convention of using an end-of-treatment phase or an initial quit date as a starting point for abstinence measurements was not followed because this study aimed to compare tobacco cessation treatments as they are actually practiced in a clinical setting. Specifically, guideline-adherent treatment for a chronic relapsing condition, such as tobacco use disorder, is a continuous process requiring multiple quit attempts and reapplication of interventions aimed at “recycling” patients who relapse
(14,
30,
31). Consistent with expert recommendations
(12,
14,
31), study treatments were not defined by a discrete episode of care marked by a fixed endpoint, number of sessions, or single quit date.
Repeated 7-day point prevalence abstinence was used as a proxy for prolonged abstinence, consistent with prior research
(32–
36). Repeated 7-day point prevalence abstinence was based on three consecutive follow-up assessment intervals (4, 6, and 9 months after random assignment). The 2-month assessment was not included in this computation, allowing subjects a stabilization period to recover from lapses after initial intervention
(37). Although repeated 7-day point prevalence abstinence is a less conservative measure than prolonged abstinence, the two measures nevertheless correlate highly (r=0.85–0.94 [
38,
39]).
Mental health quality assurance outcomes
The PTSD Checklist
(28) and the Beck Depression Inventory
(29) were completed by subjects at baseline and 6- and 9-month follow-ups. The subjects rated symptoms that occurred during the prior week. The PTSD Checklist and the Beck Depression Inventory were used to assess changes in mental health symptoms over time in order to determine if smoking cessation was associated with worsening symptoms.
Patient satisfaction outcomes
The subjects in each study condition rated their satisfaction with the amount and quality of smoking-cessation treatment received by using a 4-point Likert scale (1=“very dissatisfied” and 4=“very satisfied”). The ratings were gathered at the terminal (9-month) study assessment.
Data Analysis
Abstinence data were analyzed in an intent-to-treat analysis by using marginal logistic regression with the method of generalized estimating equations. This statistical approach to analyzing smoking-cessation outcome data conforms to recommendations by an expert panel from the Society for Research on Nicotine and Tobacco
(39). The generalized estimating equation method has several advantages in that it accounts for the correlation of repeated measurement, uses all available data points, and is sensitive to the pattern of change over time
(40). Covariates in the analysis were baseline measures of severity of nicotine dependence (the Fagerstrom Test for Nicotine Dependence
[41]) and depression (the Beck Depression Inventory). Depression was included as a covariate because of evidence showing that it may adversely affect smoking-cessation outcomes
(42–
44).
The effect of smoking cessation on mental health symptoms at 6- and 9-month follow-up assessments was analyzed by computing change from baseline scores for the PTSD Checklist and the Beck Depression Inventory at these assessment intervals. Separate Mann-Whitney U tests were then used to compare these change scores for continuing smokers versus subjects who were abstinent at each assessment interval.
All categorical variables (other than abstinence status) were analyzed with chi-square tests. Normally distributed continuous variables were analyzed with Student’s t tests. Nonparametric tests (Mann-Whitney U and Spearman’s correlation) were performed on nonnormally distributed variables.
Discussion
This study demonstrated the feasibility of training mental health providers to integrate guideline-based smoking-cessation treatment into mental health care for veterans with PTSD. PTSD clinic prescribers readily incorporated the delivery of tobacco-cessation medications into their clinical practice. Case managers also documented a high rate of adherence to prescribed behavioral counseling interventions, although the absence of verification through independent ratings of session content limits the validity of this finding. The integrated model of smoking-cessation treatment tested here was more effective for PTSD patients than for care provided by VA smoking-cessation specialists, as measured by point-prevalence abstinence. The difference between study conditions on the repeated 7-day point prevalence abstinence measure (12% for integrated care, 3% for the usual standard of care) was not significant, owing to the limited statistical power of the study. Since completing this randomized trial, PTSD clinic staff have delivered integrated care to 107 additional patients, indicating sustainability of the intervention in routine clinical practice. (For these patients, carbon monoxide-verified [≤10 ppm], repeated 7-day point prevalence abstinence quit rates measured at 4, 6, and 9 months after treatment onset were 15%.)
Subjects in integrated care participated in a greater number of smoking-cessation counseling sessions than subjects in usual standard of care and were significantly more likely to receive transdermal nicotine and nicotine gum. They were also more likely to receive combination tobacco-cessation pharmacotherapy, which has been shown to improve smoking quit rates over use of monotherapy
(14). These findings suggest that integrated care was a more effective vehicle than the usual standard of care for delivering cessation treatments of sufficient intensity. Integrated care may have been more successful at engaging subjects in cessation treatment, as indicated by higher satisfaction ratings for integrated care than usual standard of care and encouraging medication compliance. Additionally, the continuous therapeutic relationship afforded by integrated care, as opposed to the episodic care provided by the usual standard of care, may have improved access to ongoing monitoring and provider-initiated relapse management. Other research has shown a “dose-response” relationship between the number of smoking-cessation contacts and smoking outcomes
(14,
33,
35), as well as higher quit rates for extended versus brief tobacco use treatments
(45). In fact, some treatment trials suggest that cessation may be related more to the number of counseling sessions received than specific therapeutic methods used for smokers with a positive history of depression
(32–
35). Successful smoking-cessation treatment for veterans with PTSD may similarly require a greater number of contacts over an extended time.
Guideline-recommended smoking-cessation treatments yielded point prevalence quit rates of 15% to 25% in U.S. Public Health Service meta-analyses of 6,000 studies involving follow-up assessments of at least 5 months
(12). Individuals with mental disorders were typically excluded from smoking-cessation clinical trials reviewed by the U.S. Public Health Service, as active psychiatric comorbidities complicate cessation efforts and are associated with significantly reduced quit rates
(5,
46–49). The fact that point prevalence quit rates observed in this study fell within the range of those reported in meta-analyses of treatment trials is encouraging, given that our subjects were chronically mentally ill and experienced moderate to severe symptoms of PTSD and depression. Research is clearly needed that improves upon the repeated 7-day point prevalence abstinence quit rate (12%) of the integrated-care condition. Reapplication of integrated care over a longer period in order to promote “recycling” is one possible strategy to test. Another is to increase the intensity of initial integrated care interventions in an effort to prevent early relapse to smoking. Application of integrated care on a systemwide basis, even with a prolonged smoking quit rate of 12%, still has a potential for producing 10,000 quitters from within the population of PTSD patients seen at the VA alone (estimates derived from VA databases [
11,
50,
51]).
There exist remarkably few smoking-cessation clinical trials involving individuals with current mental disorders that provide a standard of comparison for the present study. A number of studies have been conducted with smokers having a history of past major depression
(32–
36,
52,
53). However, these studies do not provide the appropriate standard of comparison for the present study, and the preponderance of evidence from these trials does not substantiate the hypothesis that history-positive smokers are less likely to quit than history-negative smokers
(54). Clinical trials involving smokers with current mental disorders who meet the minimum recommended standard for abstinence at follow-up assessments (≥6 months [
37,
55]) have focused on schizophrenia and alcohol dependence. (Single point prevalence abstinence rates at the terminal 6-month follow-up assessment for studies of smokers with schizophrenia: 11%
[56], 12%
[57], 7%–17%
[58], and 19%
[59]; single point prevalence abstinence rates at the terminal 12-month assessment for studies of alcohol-dependent patients: 12%
[60] and 13%–19%
[61].) Quit rates from these studies of clinical samples are roughly equivalent to, if not slightly lower than, the single-point prevalence quit rates for integrated care reported here. The only study
(61) reporting prolonged abstinence (repeated 7-day point prevalence abstinence at follow-up months 1, 3, 6, and 12) found quit rates of 10% and 12% for two active tobacco-cessation treatments involving recovering alcoholics. These results are comparable to the repeated 7-day point prevalence abstinence quit rate of 12% in the present study.
The rate of smoking abstinence for integrated care subjects at study termination was less than half that observed at the 2-month assessment. Several possible mechanisms that may contribute to smoking relapse in PTSD patients deserve further exploration. Specifically, pre- and postcessation negative affective states
(24,
32,
33,
44,
46,
62) and increased symptoms of depression immediately following a quit attempt
(63) increase relapse susceptibility and may have suppressed sustained quit rates in this study. Heightened withdrawal sensitivity has predicted relapse in smokers with a history of depression in some studies
(64,
65) and may possibly explain continued smoking in PTSD patients as well.
Stopping smoking in this treatment trial was not associated with worsening symptoms of PTSD or depression measured at 6 and 9 months after treatment onset. Our results are more consistent with studies showing that mood disturbances do not result from stopping smoking
(32,
46,
66) than with studies showing that they do
(25–
27,
67) among individuals with a history of depression. However, comparing our results with those of others is attenuated because of the marked differences in study groups (i.e., current PTSD versus a history of major depression). Three other possible explanations for this finding also deserve consideration. First, transient symptom exacerbations related to postcessation nicotine withdrawal may have occurred shortly after subjects’ cessation attempts but were undetected because outcomes were only measured at four discrete time points in our study. Second, the fact that 86% of the subjects in the present research were receiving antidepressant medications (other than bupropion for smoking cessation) for PTSD may have prevented potential deterioration effects. This possibility is supported by evidence showing that sertraline reduces nicotine withdrawal symptoms
(53) and that nortriptyline alleviates postcessation negative affect
(35) in smokers with a history of major depression. Third, subjects who experienced worsening psychiatric symptoms upon trying to quit smoking may have simply resumed smoking in order to manage these symptoms. Our data do not support this latter explanation since symptoms of PTSD and depression did not change from baseline to the 6- and 9-month assessment for subjects who continued to smoke compared to those who quit. However, the nonsignificant difference between subjects who quit and those who did not on symptom measures may reflect low statistical power owing to the small number of quitters involved in comparisons at months 6 and 9.
Interpretation of this study’s findings may be limited by several methodological shortcomings. This investigation did not follow the convention of measuring outcomes from a clearly demarcated quit date or the end of the intervention period because smoking-cessation treatment was conceptualized as a continuous process involving reapplication of treatment and multiple quit attempts
(31). The comparability of findings with other research that measures outcomes from an initial quit date may therefore be limited. Biomarkers of longer-term nonsmoking status were not used but should ideally have supplemented expired carbon monoxide in order to verify subject self-reports. Continuous abstinence was also not assessed because it is not verifiable with standard biological assays. However, continuous abstinence data based on subject self-report alone would have been informative because subject reports of smoking status are generally valid
(68). Proportions of subjects who received prescriptions filled by the pharmacy were reported, but a specific measure of medication adherence was not included. Thus, it is impossible to determine to what degree, if any, medication adherence may have affected outcomes. PTSD patients with a current substance abuse disorder were included in the study, but those with current substance dependence were not. Thus, our findings may not generalize to smokers with PTSD and comorbid substance dependence disorders. Finally, a much larger study group is clearly required in order to conclude definitively that integrated care is more effective than the usual standard of care for smoking cessation in other clinical settings.
This service delivery study was conducted in the spirit of a clinical effectiveness trial in order to promote generalization to “real world” health care settings
(69). To this end, treatments were represented as they are likely to be actually practiced, and broad subject inclusion criteria were used to ensure that the study group reflected the population of VA PTSD clinic patients. Despite threats to internal validity inherent in such trials
(69), this study provided a favorable test of the feasibility and outcome of APA recommendations
(49) to implement smoking-cessation treatment in practice-based settings for patients with current mental disorders. Additional studies of integrated models of smoking-cessation treatment for mentally ill patients are warranted, given the extraordinarily high prevalence of heavy smoking in these individuals (41%
[5] to 50%
[70]).