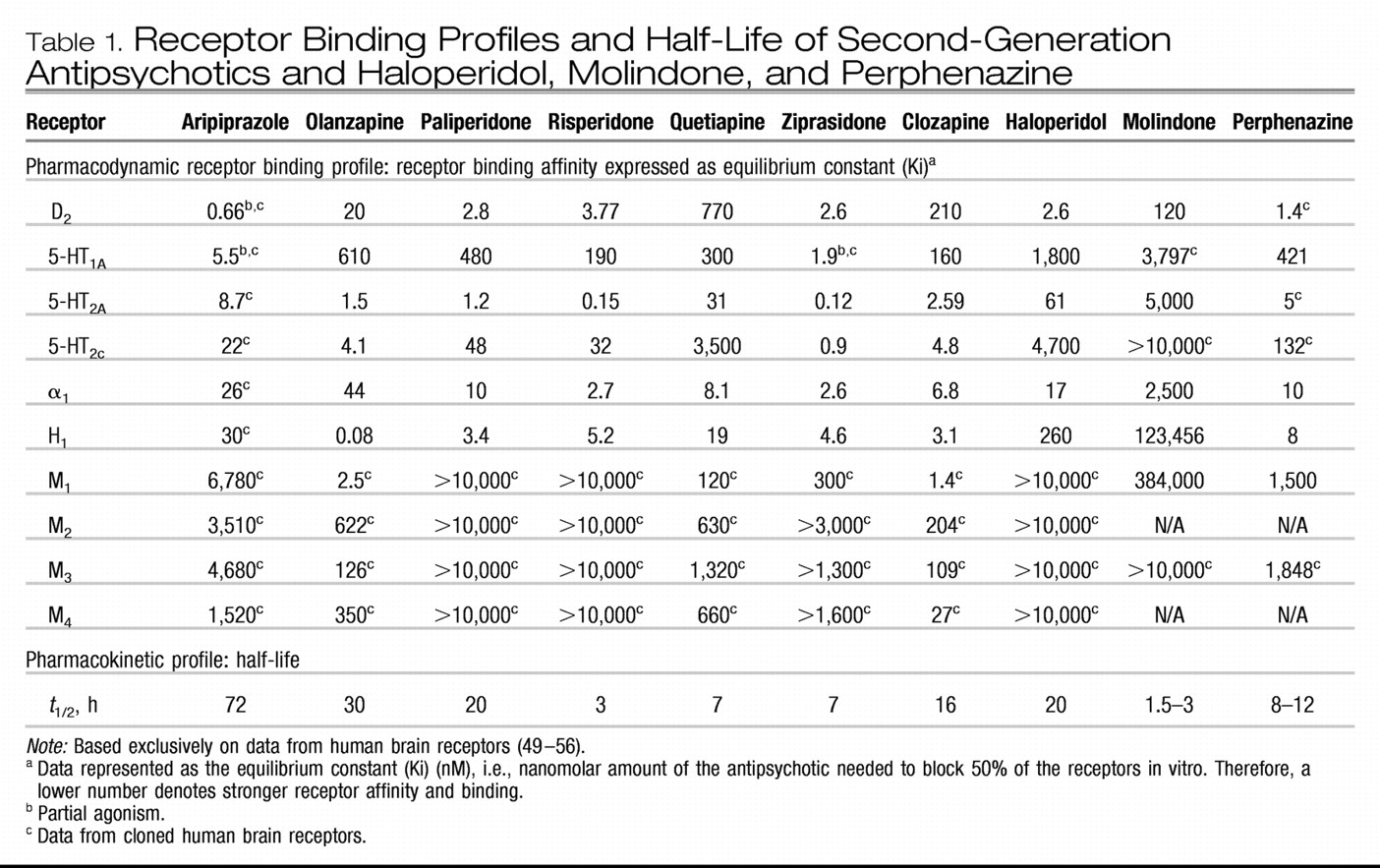
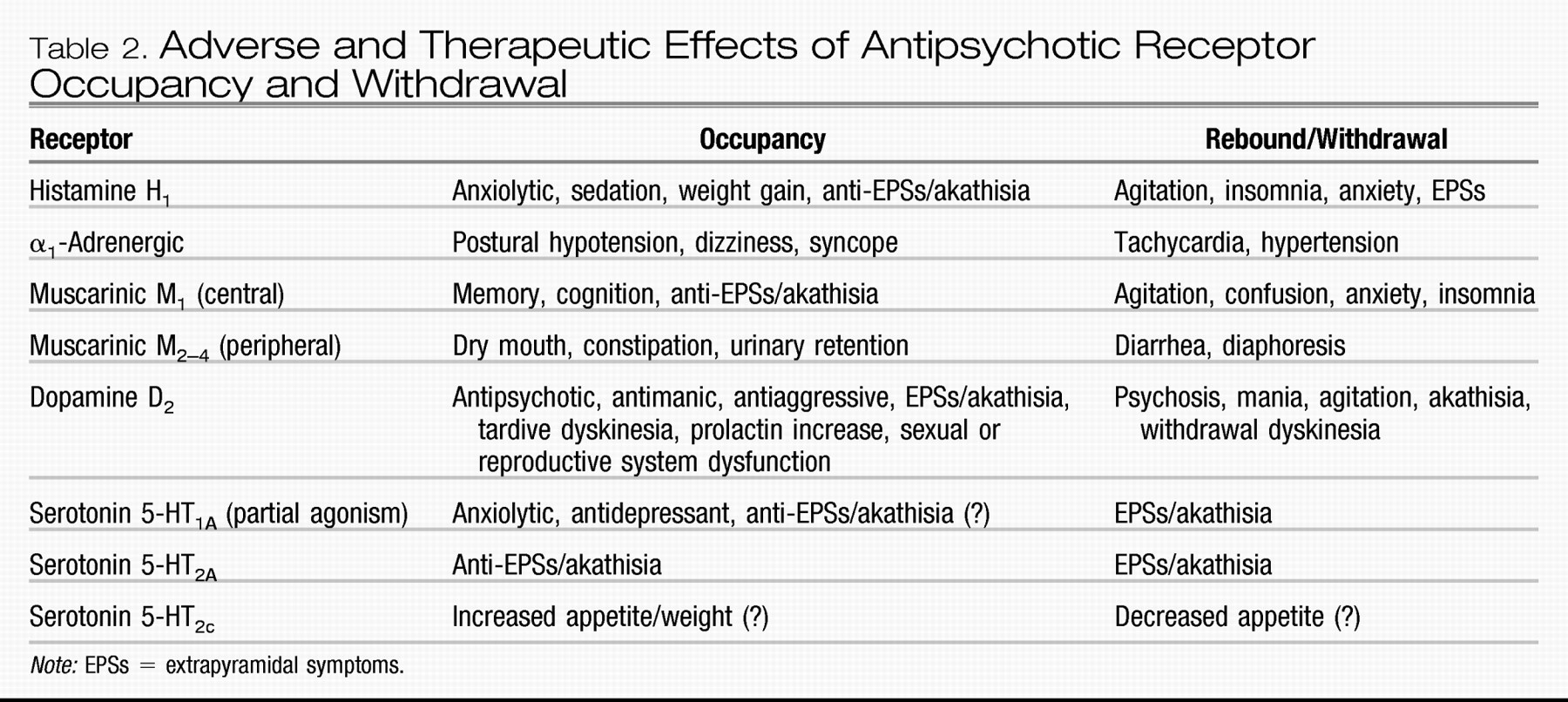
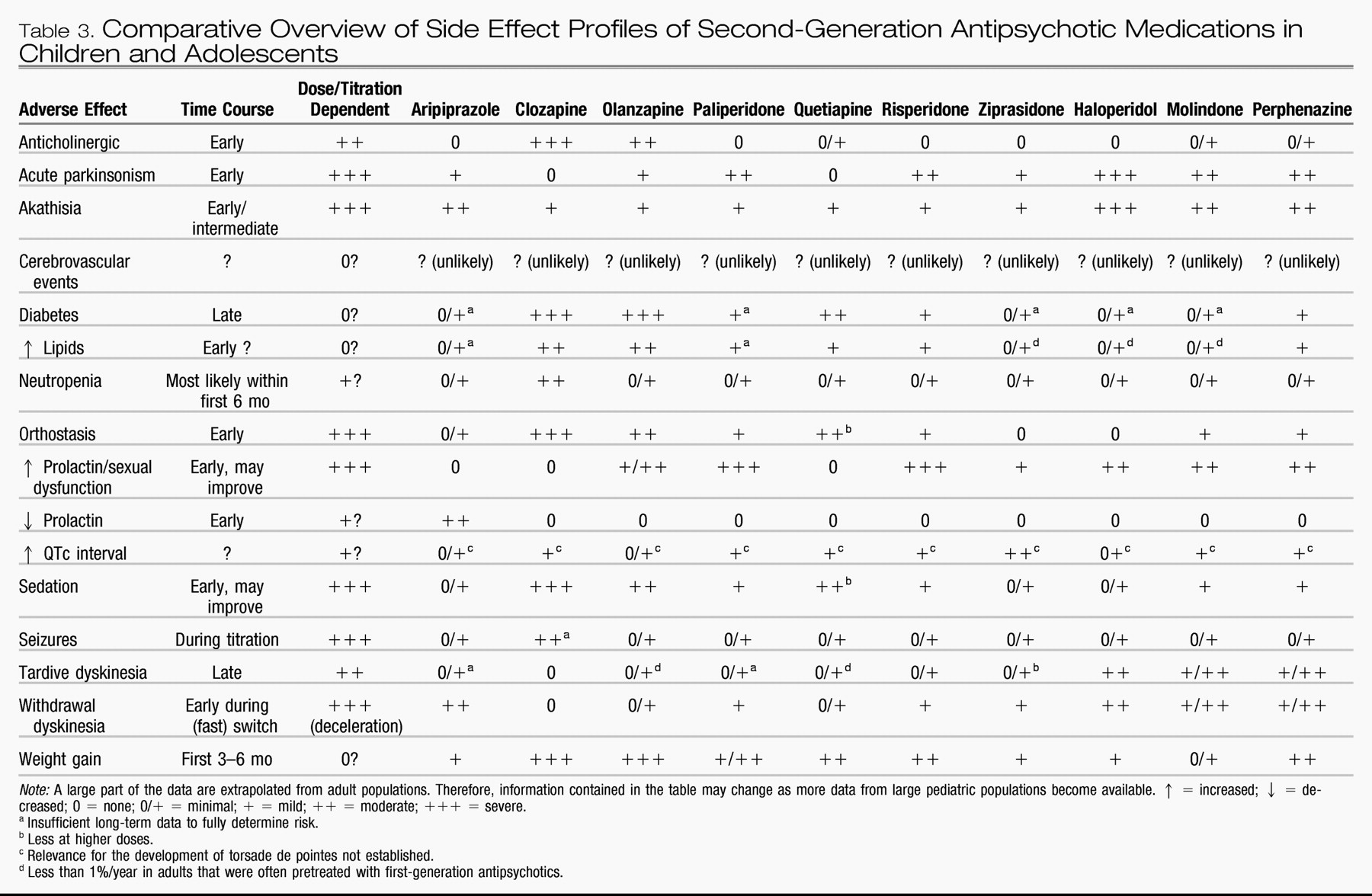
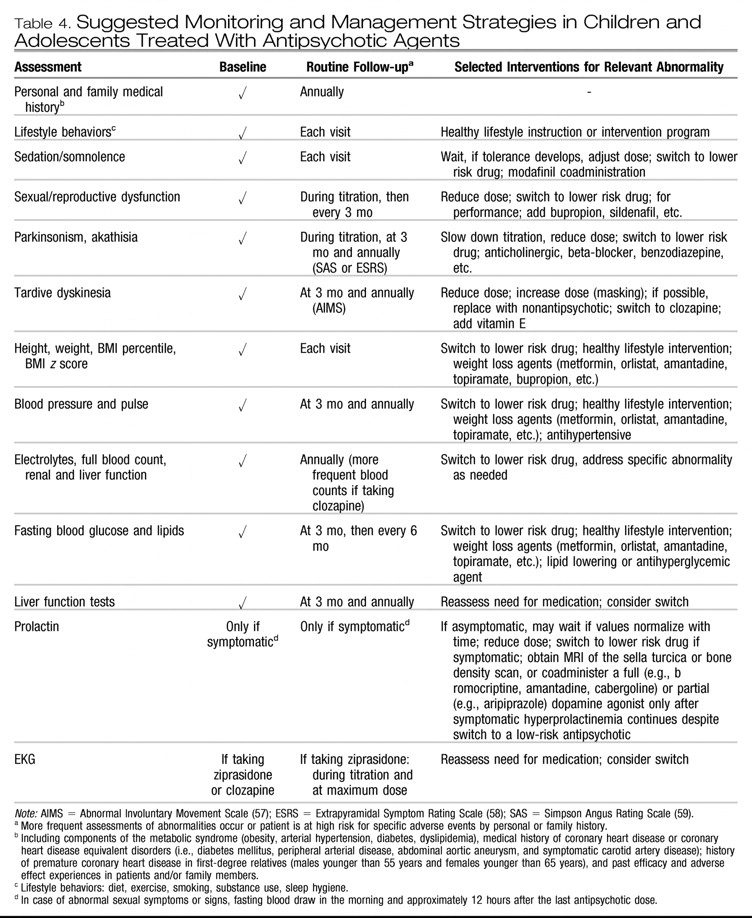
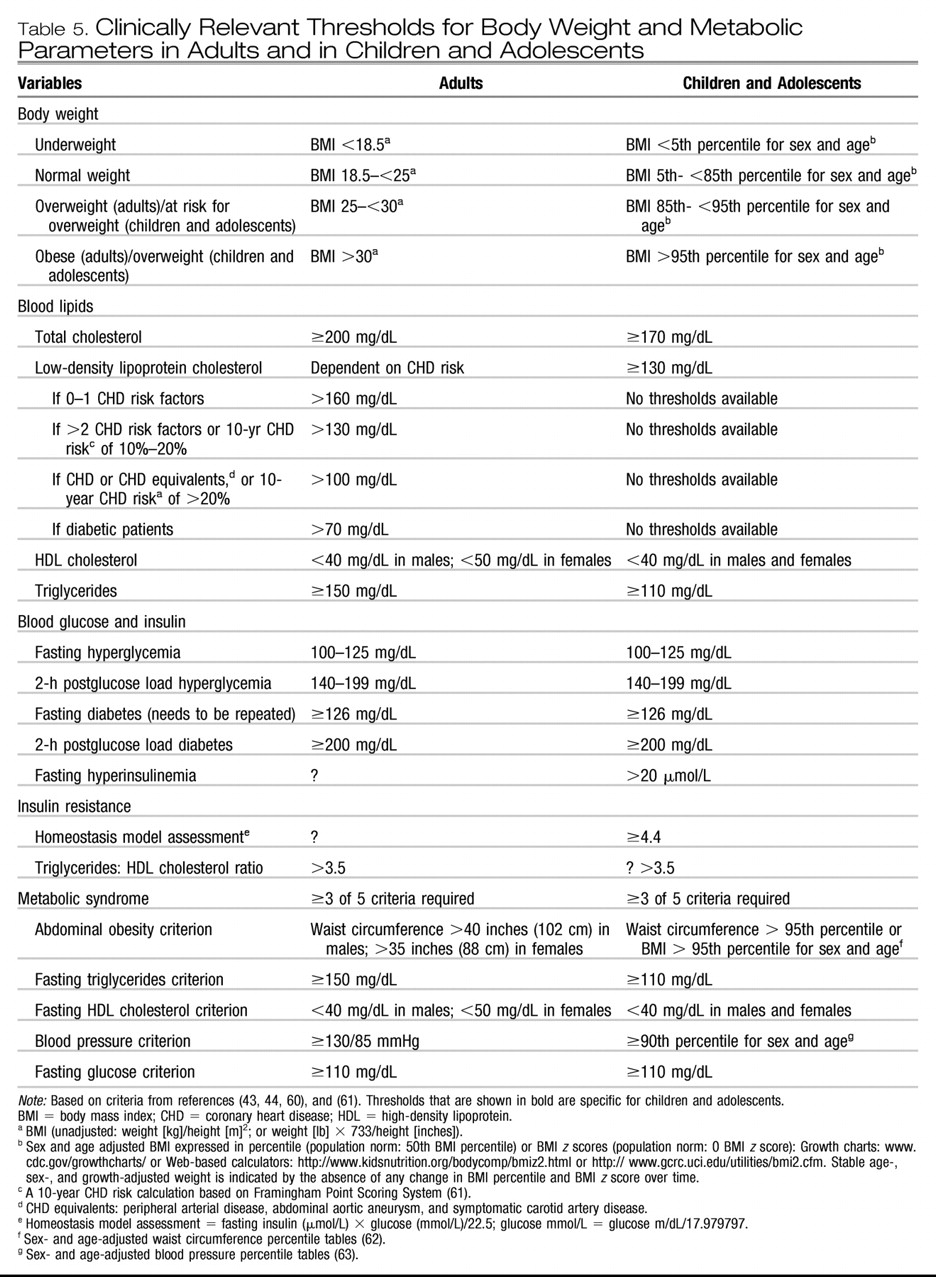
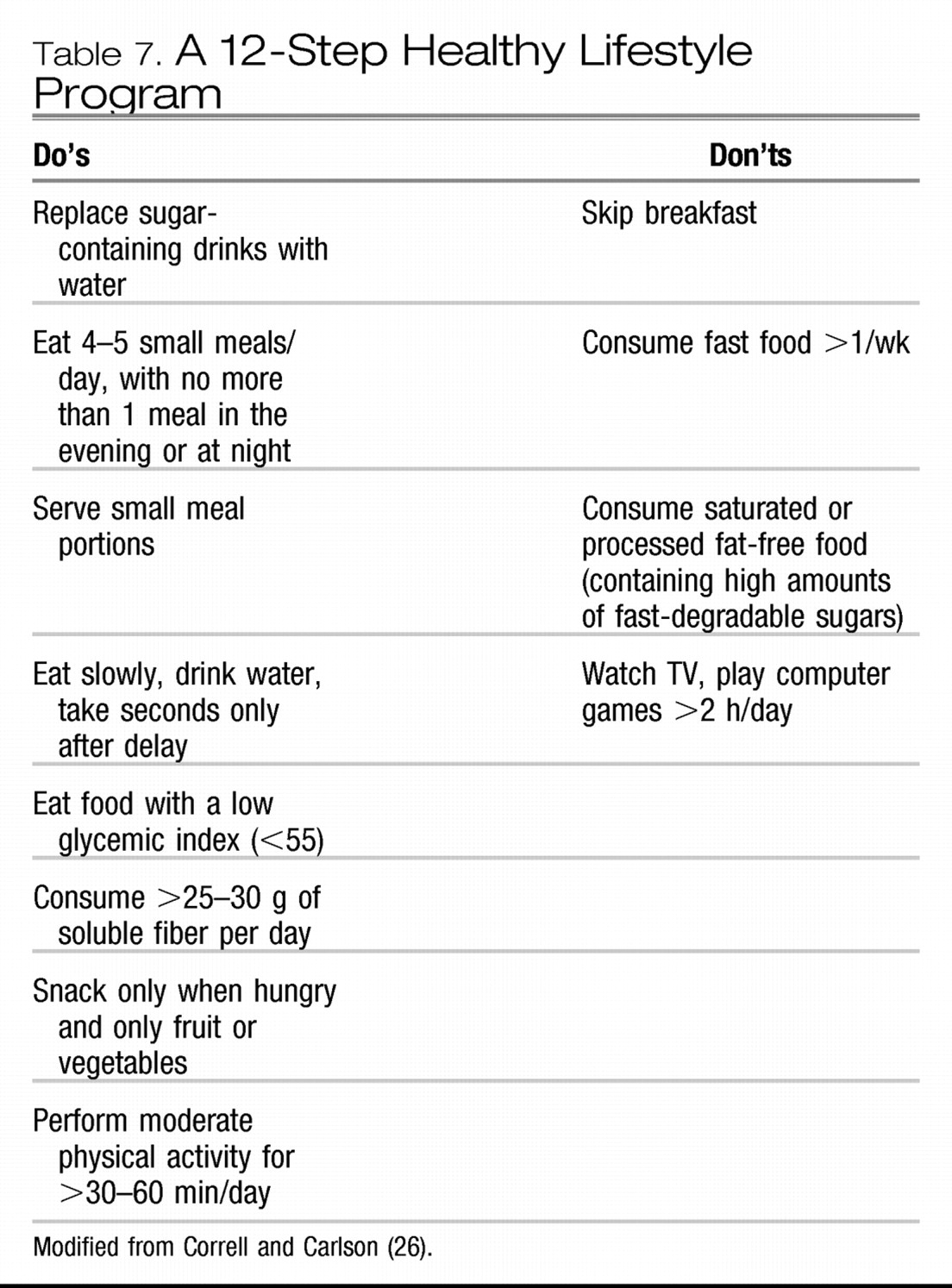
EXTRAPYRAMIDAL SIDE EFFECTS
In general, children and adolescents are more likely to experience extrapyramidal side effects (EPSs; i.e., parkinsonian side effects and dystonia) associated with FGAs and SGAs than adults (
6). To date, only one double-blind, randomized pediatric study directly compared EPS rates with an FGA (i.e., haloperidol; mean dose 5.0 mg/day) and SGAs (i.e., risperidone, mean dose 4.0 mg/day, and olanzapine, mean dose 12.3 mg/day) (
7). Results of substantial EPSs not only with haloperidol (67%) but also with olanzapine (56%) and risperidone (53%) suggest that children and adolescents are at risk for EPSs, even when treated with SGAs, at least at doses required to control psychosis. Although overall EPS rates were not significantly different in this study (
n = 40), the severity was greater with haloperidol, indicating that reporting global incidence rates are insufficient to guide clinical treatment. In several short-term, double-blind, placebo-controlled trials, risperidone was associated with EPS rates between 8% and 26% (
8–
11). In four open-label extension trials lasting 11 to 36 months, EPS rates ranged from 8.6% to 26.0% (mean 15.7%) (
12). Although in these trials, rating scale-measured EPS severity generally did not increase, EPS rates could be underestimates because they are based on spontaneous reports of mostly prepubertal individuals with autism spectrum disorder treated with relatively modest mean risperidone doses (1–2 mg/day). As in adults, clozapine (
13,
14) and quetiapine (
15) appear to be associated with relatively low EPS rates in pediatric patients, whereas more data are needed for ziprasidone and aripiprazole. In one recently presented double-blind, placebo-controlled study of aripiprazole in adolescents with schizophrenia, EPSs occurred in 18% of patients (
16). Of note, due to complementary actions of dopamine and serotonin regarding EPSs, concurrent treatment with serotonin reuptake inhibitors may trigger or aggravate EPSs. Treatment options are listed in
Table 4.
Akathisia. In children and adolescents, less is known regarding the risk for akathisia, which has been substantial with FGAs across age groups. In general, it can be difficult to properly diagnose akathisia because the presentation overlaps with psychomotor agitation due to psychosis, mania, and anxiety. In children and adolescents, akathisia may present as difficulty falling asleep and can be mistaken for attention-deficit/ hyperactivity disorder. In one study (
7), olanzapine was associated with akathisia in 12.5% of pediatric patients. Aripiprazole, the only available antipsychotic with partial dopamine D
2 agonism, has been associated with akathisia in adults. In one retrospective chart review of 30 children and adolescents (mean age 13.3 years, range 5–19), akathisia was recorded in as many as 23% of patients taking aripiprazole (
17). However, the mean starting dose of 9 ± 4 mg in this sample was relatively high and almost identical to the mean final dose of 10 ± 3 mg. Furthermore, it is unclear whether abrupt switches could also have increased the akathisia rate via withdrawal/rebound phenomena (
Table 2). In a large randomized placebo-controlled study of more than 300 patients, aripiprazole dosed at 10 or 30 mg/day was associated with akathisia in <10% of adolescents with schizophrenia, but patients were titrated from a starting dose of 2 mg/day, reaching the maximum dose at day 5 or 13, respectively (
16). This suggests that slower titration of a partial agonist may reduce the rate of akathisia in some patients. As for parkinsonian adverse effects, concurrent use of serotonin reuptake inhibitors can also trigger or aggravate akathisia. Treatment options are listed in
Table 4.
Withdrawal Dyskinesia. During treatment with FGAs, children and adolescents seem to be at risk for developing withdrawal dyskinesias, yet, different from adults, these are frequently reversible (
18). Rates of withdrawal dyskinesia appear to be lower with SGAs compared with FGAs (
19), although a switch from an antipsychotic with strong D
2 affinity (e.g., risperidone, aripiprazole) to one with less potent affinity (e.g., quetiapine) may predispose to withdrawal dyskinesia. In one study (
20), 2 of 13 children (15.4%) developed mild, reversible withdrawal dyskinesia after 7 months of risperidone treatment. In another, more recent study, however, 0 of 38 patients developed withdrawal dyskinesia after risperidone (mean dose 2 mg/day) was discontinued after 4 months of treatment (
21). The risk for withdrawal dyskinesia can be reduced with slow cross-titration and overlapping cross-titration (
5).
Tardive Dyskinesia (TD). A recent meta-analysis of 10 studies lasting at least 11 months reported on TD rates in 783 pediatric patients ages 4 to 18 (weighted mean 9.8) years old (
12). Most patients were prepubertal (79.2%), male (81.7%), and white (78.4%). Across these studies, only three cases of TD were reported, resulting in an annualized incidence rate of 0.4%. Although this pediatric rate is approximately half the risk found in another meta-analysis of 1,964 nonelderly adults, (
22) firm conclusions are precluded by the facts that none of the pediatric studies were designed specifically to detect TD, antipsychotic doses were low, and lifetime exposure was relatively short.