Antipsychotic medications have long been a cornerstone of effective treatment for schizophrenia. Most second-generation antipsychotics have also been approved to treat bipolar disorder, and in late 2007 aripiprazole was approved for the adjunctive treatment of major depressive disorder. However, once a drug has been approved by the Food and Drug Administration (FDA), clinicians are free to prescribe it as they see fit. The benefits of such off-label use are usually unclear because there has not been a high level of published clinical research evaluating the safety and efficacy of these drugs for non-FDA-approved indications. Although some off-label use of second-generation antipsychotics may be appropriate, these drugs are expensive and have serious side effects (including weight gain, diabetes mellitus, tardive dyskinesia, and extrapyramidal symptoms), and their off-label use may therefore represent significant risk and cost with undemonstrated clinical benefit and potential harm.
Several studies have investigated the extent to which antipsychotic medications have been used off label. A previous Department of Veterans Affairs (VA) study found that 33.5% of patients who received a prescription for an antipsychotic medication in the VA health care system during a four-month period in 1999 did not have a diagnosis of either schizophrenia or bipolar disorder (
1 ). A more recent study by Domino and Swartz (
2 ), using data from the Medical Expenditure Panel Survey (MEPS), found rates of off-label use of 18%–19% in 1996–1997 and 2004–2005. A report by the Agency for Healthcare Research and Quality (AHRQ) found that the most common off-label uses of second-generation antipsychotics reported in the literature were treatment of agitation in dementia and treatment of depression, obsessive-compulsive disorder, posttraumatic stress disorder (PTSD), personality disorders, Tourette's syndrome, and autism; however, there was very little strong evidence in the literature that these drugs were effective in treating these disorders (
3 ).
The objective of this study was to determine the prevalence of off-label use of antipsychotic medications for mental illness in the VA health care system in fiscal year (FY) 2007 and to explore patient sociodemographic and clinical characteristics associated with off-label use of these drugs. This study contributes to the existing literature by providing more recent estimates than the previous VA study (
1 ). Also, because MEPS relies on patient self-report for diagnostic information (
2 ), this study offers more precise estimates because it is based on actual prescription records.
Methods
Sources of data
Data for the study were obtained from national administrative VA databases for FY 2007 (October 1, 2006, through September 30, 2007). Data on prescriptions for antipsychotic medications came from the Decision Support System pharmacy file. This file contains data on all prescriptions filled at VA pharmacies nationwide and includes the medication prescribed, strength, number of pills per prescription, and number of days supplied. Sociodemographic and clinical data came from the Outpatient Encounter file, which contains data on all VA outpatient clinic visits nationwide. Available sociodemographic data included age, gender, and race and ethnicity. Clinical data included diagnoses (based on International Classification of Diseases, 9th edition [ ICD-9 ] codes) and the degree to which the patient was service connected, which is a measure of disability. The study was approved by the institutional review boards of the West Haven VA Medical Center and the Yale School of Medicine.
Measures
First, all prescriptions for antipsychotic medications were identified among seven antipsychotic medication types: aripiprazole, clozapine, olanzapine, quetiapine, risperidone, and ziprasidone, plus first-generation antipsychotics considered as a class. Next, outpatient records were examined to determine whether patients had any visits that indicated a diagnosis of either schizophrenia ( ICD-9 codes beginning with 295) or bipolar disorder ( ICD-9 codes 296.0, 296.1, and 296.4–296.8).
Patients without a diagnosis of schizophrenia or bipolar disorder during FY 2007 who received an antipsychotic medication were considered to have received these medications off label. Among these patients, the average daily dose was computed for all prescriptions within seven days of the last prescription of the year by multiplying the number of pills by the strength per pill and dividing by the number of days supplied.
Doses of first-generation antipsychotics were converted to chlorpromazine equivalents.
Average daily doses were compared with the recommended dosing range for schizophrenia specified by the Schizophrenia Patient Outcomes Research Team (PORT) (
4 ), and patients with doses below this range were identified with a dichotomous variable. Patients without a diagnosis of schizophrenia or bipolar disorder were assigned to the following diagnostic groups on the basis of whether they had any visits that indicated the corresponding nonexclusive
ICD-9 codes: adjustment reaction (309 codes, excluding 309.81), alcohol abuse and dependence (codes 303 and 305.00), anxiety disorder (300 codes, excluding code 300.4), drug abuse and dependence (codes 292, 304, and 305.2–305.9), major depression (296.2 and 296.3), minor depression (296.9, 300.4, 301.1, and 311), organic brain syndrome and Alzheimer's disease (290, 293, 294, 310, and 331.00), other psychosis (297–299), and PTSD (309.81). Patients could belong to more than one diagnostic group.
Analysis
First, all patients who were given prescriptions for an antipsychotic medication during FY 2007 were identified, and the number and proportion of off-label users who received each drug were determined.
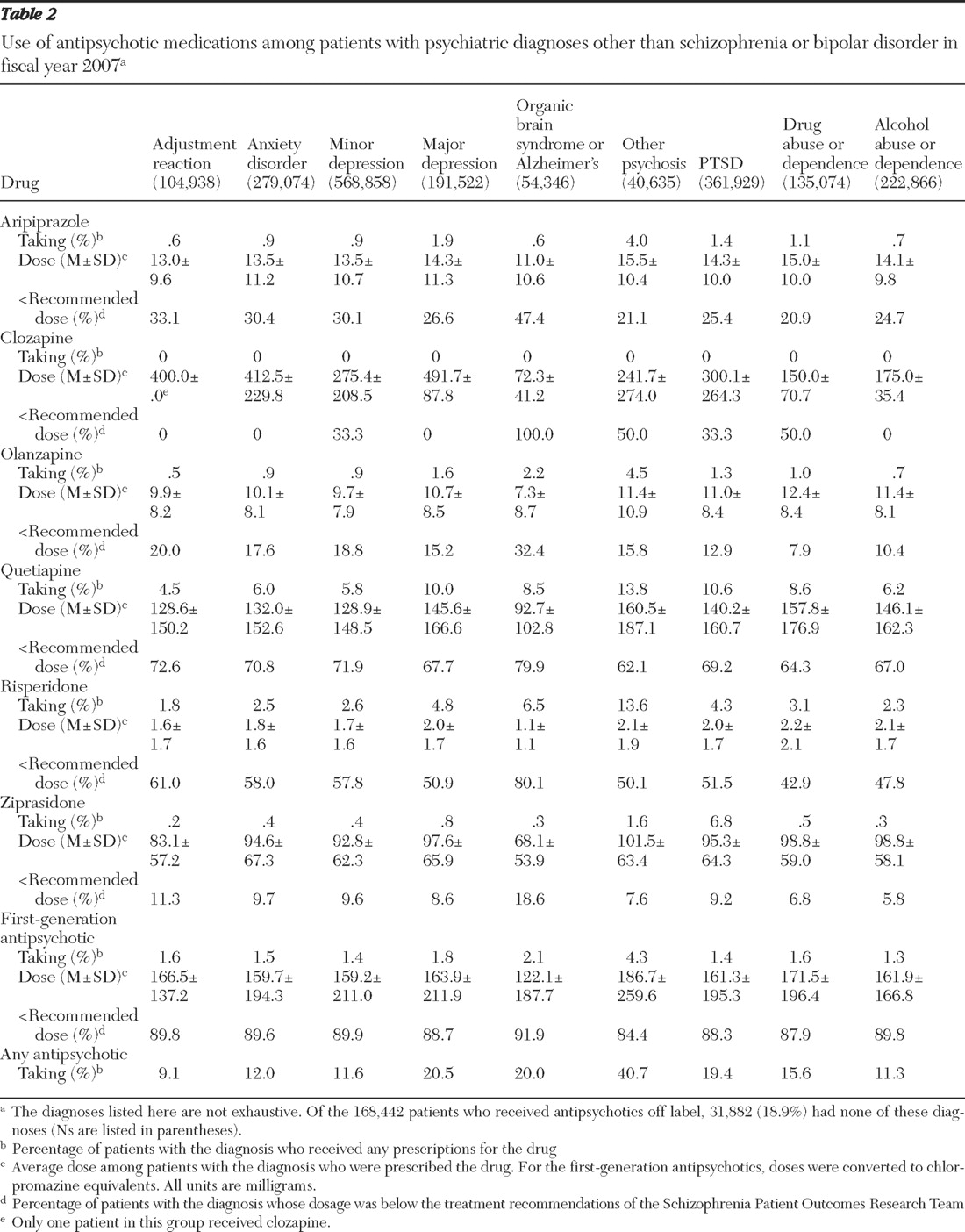
Next, all patients belonging to any of the nine diagnostic groups listed above (excluding those with any comorbid diagnoses of schizophrenia or bipolar disorder) were identified. The proportion of patients in each of these diagnostic groups who received a prescription for an antipsychotic medication was determined, along with the average daily dose of each antipsychotic among users of that medication and the percentage with doses below the PORT-recommended dosage range for schizophrenia.
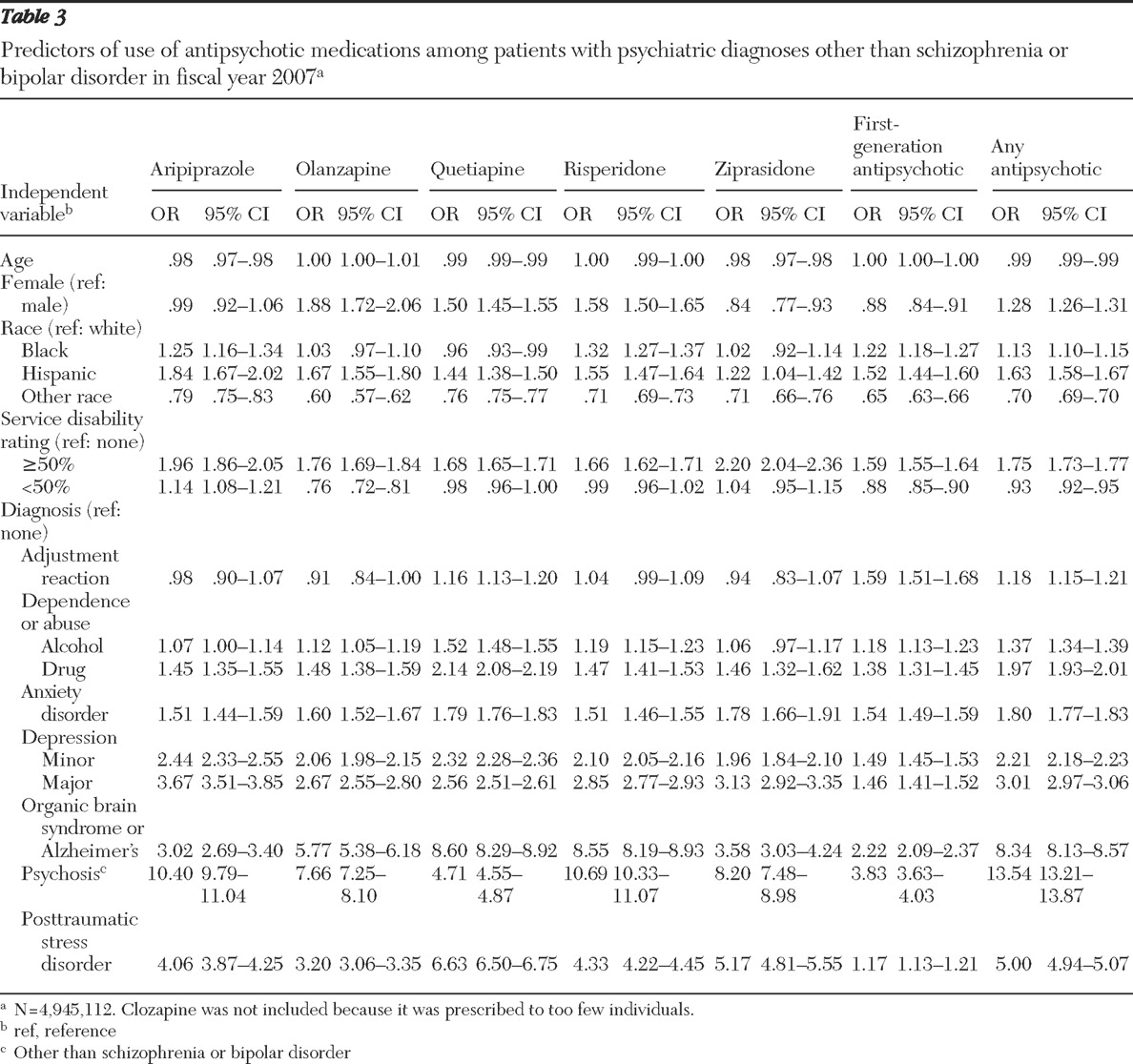
Finally, in order to identify patient-level factors associated with off-label use of these agents, logistic regression models were developed in which use of off-label antipsychotic agents was the dependent variable and the following were the independent variables: age, female gender, black race, Hispanic ethnicity, other race-ethnicity or race-ethnicity data missing, service-connected disability rating of at least 50%, service-connected disability rating of less than 50%, and diagnostic group. The service-connected disability rating is a measure of disability related to the veteran's military service. Because these models intended to predict off-label use, the sample was again restricted to individuals with no diagnosis of schizophrenia or bipolar disorder. Separate models were run with receipt of the following antipsychotic drugs as the dependent variable: aripiprazole, olanzapine, quetiapine, risperidone, ziprasidone, and first-generation antipsychotics. Because so few patients received clozapine off label, a separate model was not estimated for clozapine. A final model was estimated for receipt of any antipsychotic medication off label.
Discussion
In this study, we used national VA administrative data for FY 2007 to determine the extent to which antipsychotic medications are used off label and to identify factors associated with off-label use. We found a considerable degree of off-label use of these drugs; 60.2% of patients who were prescribed antipsychotic medications had no diagnosis of either schizophrenia or bipolar disorder during the fiscal year. The most frequently prescribed drugs for off-label use were quetiapine, risperidone, and first-generation antipsychotics. Patients diagnosed as having another psychosis, PTSD, or organic brain syndrome or Alzheimer's disease had the highest odds of being prescribed an antipsychotic off label.
Although the rate of off-label use of antipsychotic medications was comparable with the 63.6% rate seen in the Georgia Medicaid population in 2000–2001 (
5 ), we found a higher rate of off-label use than was found in many previous studies. A previous VA study reported that 42.8% of VA patients who were prescribed second-generation antipsychotics during a four-month period in 1999 received these drugs for an off-label use (
1 ). At the time, second-generation antipsychotics had not been FDA approved for treating bipolar disorder, however. If patients with a diagnosis of bipolar disorder in that study are removed, the rate of off-label use falls to 33.4%. The VA study did not include first-generation antipsychotics, which explains at least some of the difference in rates of off-label use. However, first-generation antipsychotics accounted for only 20.5% of off-label use in our study, so the exclusion of these drugs is not likely to explain all of the difference.
A more recent study based on national MEPS survey data reported that the percentages of antipsychotic medication users with an affective disorder or anxiety spectrum disorder without comorbid schizophrenia in 2004–2005 were 22% and 19%, respectively (
2 ). Although these results are similar to those of our study (in which 20.0% of off-label users had an anxiety disorder), the MEPS study did not report rates of off-label use more generally or for other mental conditions. In addition, because MEPS contains only self-reported diagnoses, it is likely that disease prevalence was underestimated, especially for mental illnesses, which can lead to bias in the estimate of off-label use. An advantage of our study is that diagnoses were based on administrative records from actual VA clinic visits and hence may be more accurate than patient self-reports.
The rate of off-label use of antipsychotics is high compared with off-label use of other medications. Using data from a survey of office-based physicians, Radley and colleagues (
6 ) found that 21% of all prescriptions were for an off-label use, and less than one-third of these (27%) were supported by strong scientific evidence of clinical efficacy. Rates of off-label use were highest for cardiac medications (46%), anticonvulsants (46%), and antiasthmatics (42%). Antipsychotic medications were not well represented in the study (only two were included), but this is not surprising given that the population surveyed was not limited to mental health providers.
A high rate of off-label antipsychotic use would not necessarily be of concern if there were scientific evidence supporting the effectiveness of these medications for conditions other than schizophrenia and bipolar disorder. However, such evidence is generally lacking or weak. Although a meta-analysis by Schneider and colleagues (
7 ) found a small but statistically significant benefit of aripiprazole and risperidone for treating agitation in dementia, the Alzheimer's disease arm of the Clinical Antipsychotic Trials of Intervention Effectiveness (known as the CATIE study) found that second-generation antipsychotics were no more effective than placebo in treating Alzheimer's disease (
8,
9 ). In addition, according to a recent AHRQ report (
3 ), the scientific evidence supporting the effectiveness of off-label antipsychotic medication use is weak for most conditions, including depression and PTSD—conditions with relatively high rates of off-label use in our study.
There may be several reasons why prescription drugs are used off label. For example, the characteristics of patients enrolled in clinical trials may bear little resemblance to those of patients typically seen in clinical practice, leading clinicians to question the generalizability of the results. Furthermore, efficacy data may not exist for some conditions or populations (such as children), although pharmaceutical manufacturers have a powerful incentive to conduct clinical trials to show efficacy in conditions other than those for which their drug has been approved for use. In fact, given that aripiprazole is now approved for the adjunctive treatment of major depressive disorder, this previous off-label use of this drug appears justified. Another possible explanation for off-label use is that clinicians try these medications as a last resort with patients who have not responded to other treatments. However, journalistic accounts suggest that some pharmaceuticals manufacturers have illegally marketed antipsychotic medications for off-label use (
10,
11,
12 ).
The average dose prescribed when antipsychotic medications were used off label varied considerably across drugs. For patients with prescriptions for quetiapine, risperidone, or first-generation antipsychotics, the average dose was generally below the PORT-recommended range for schizophrenia, and a relatively large percentage of patients were given doses below the PORT recommendations. The average aripiprazole dose across all diagnostic groups was 13.8 mg per day, which is at the lower end of the PORT-recommended range of 10–30 mg per day. However, the average doses of olanzapine and ziprasidone were well within the PORT-recommended ranges for these medications, ranging from 7.3–11.4 mg per day and 68.1–101.5 mg per day, respectively, depending on the diagnosis. Further research is needed to understand why off-label dosing of some antipsychotic medications is higher than that of others.
There are a number of limitations associated with these administrative data. First, although detailed information is available on the medication and dose prescribed and number of days the medication was supplied, we could not determine what condition a medication was intended to treat because many patients received multiple psychiatric diagnoses. Hence, although we could determine whether a patient given a prescription for an antipsychotic medication had a diagnosis of PTSD, we could not say that the diagnosis of PTSD was the reason for the antipsychotic prescription because comorbid conditions were often present. Second, diagnoses were based on ICD-9 codes, which may have been miscoded or incomplete.
These data represent prescriptions written in FY 2007. At the time, no antipsychotic had been approved by the FDA for treatment of any mental disorder other than schizophrenia or bipolar disorder. Aripiprazole was approved for adjunctive treatment of major depressive disorder in late 2007, and more up-to-date data will be needed to evaluate the decision of this new approved usage.
Finally, our data were limited to prescriptions filled within the VA system. To the extent that patients received outpatient care or prescription medications outside of the VA, our estimates of off-label use may be biased conservatively. In addition, the VA database does not include children, a particularly vulnerable patient population that has experienced significant growth in antipsychotic use (
2 ).