The implementation of Medicare Part D had the potential to significantly improve access to prescription medications for beneficiaries who previously did not have such coverage. Certain design features in Part D may have disproportionately altered care for patients with mental illness. Restrictive formularies, for example, limit treatment options for persons who use psychotropic medication, because these persons often need to try several medications before finding a product that works (
1 ). Because prescriptions are one of the largest components of mental health spending, second only to hospital care (
2 ), the introduction of Part D may have reduced access to mental health treatments compared with other, more generous, plans.
Persons dually eligible for Medicare and Medicaid were potentially vulnerable to Part D implementation because they were switched from their Medicaid drug benefits to a randomly assigned Medicare drug plan. The plan to which many dually eligible beneficiaries were assigned may not have been the best plan for their medication needs or their personal preferences, including their level of risk aversion (
3 ).
Although Part D plans are required to cover at least two drugs in each class of medications, "all or substantially all" antidepressant and antipsychotic medications are required to be included on formularies (
4 ). Despite these protections, an early report suggested that 53.4% of dually eligible beneficiaries using a protected class of mental health medications experienced problems accessing their medication after enrollment in Part D (
5 ). Of these patients, 27.3% reported a significant adverse event, such as increased suicidal ideation, emergency room visits, violent ideation, or psychiatric hospitalization or homelessness, as a result of their difficulty in attaining medication. In addition, simulation results using pre-Part D data indicated that greater medication switching among psychotropic users may have occurred in Part D plans, compared with before Part D, depending on medication used and plan characteristics (
6 ).
The purpose of our study was to examine the early experience of Medicare beneficiaries' medication use after the implementation of Part D in a nationally representative sample. We compared the use of two large categories of psychotropic medications with two large categories of nonpsychotropic medications to determine whether any of the changes observed were isolated among psychotropic medication users.
Study data and methods
We examined the rate of psychotropic use among Medicare beneficiaries using the 2004–2006 Medical Expenditure Panel Survey (MEPS) to estimate the relative impact of Part D and the differential impact among psychotropic medication users. MEPS is a two-year overlapping panel, and data are from a nationally representative sample of the U.S. noninstitutionalized population (
7 ). For Medicare beneficiaries and dually eligible beneficiaries we examined the rate of use of two large classes of psychotropic medications (antidepressant and antipsychotic medications) and two large classes of nonpsychotropic medications (lipid-lowering and antihypertensive agents). In addition, we examined whether Part D would influence the probability of refilling a prescription from one year to the next (repeat use) or the probability of initiating treatment (new starts).
We were particularly interested in the changes in rates of psychotropic use between 2005 and 2006, because Part D was implemented on January 1, 2006. We used data from survey respondents in 2005–2006 (panel 10) who were enrolled in Medicare at the end of 2005 to compare patterns of medication access between 2005 and 2006. We used data from Medicare beneficiaries in 2004–2005 (panel 9) to establish baseline trends before Part D.
Prescription medication use is captured in the MEPS through self-report and pharmacy follow-back (
8 ). We used the Multum Lexicon Therapeutic Class codes available in the MEPS files as a starting point to categorize medication classes, but we recoded a number of misidentified agents based on drug name. Individuals were described as medication users if they filled at least one prescription in the four targeted medication classes during the survey year. We took advantage of the complex survey design in MEPS in all estimates. Cell sizes of unweighted samples are reported; we urge caution in interpreting estimates because of small cell sizes (
9 ). All percentages were based on the weights and are reported as weighted. We performed statistical tests of means or proportions, retaining the estimates in cells generated on fewer than 100 observations for descriptive purposes only.
Rates of medication use
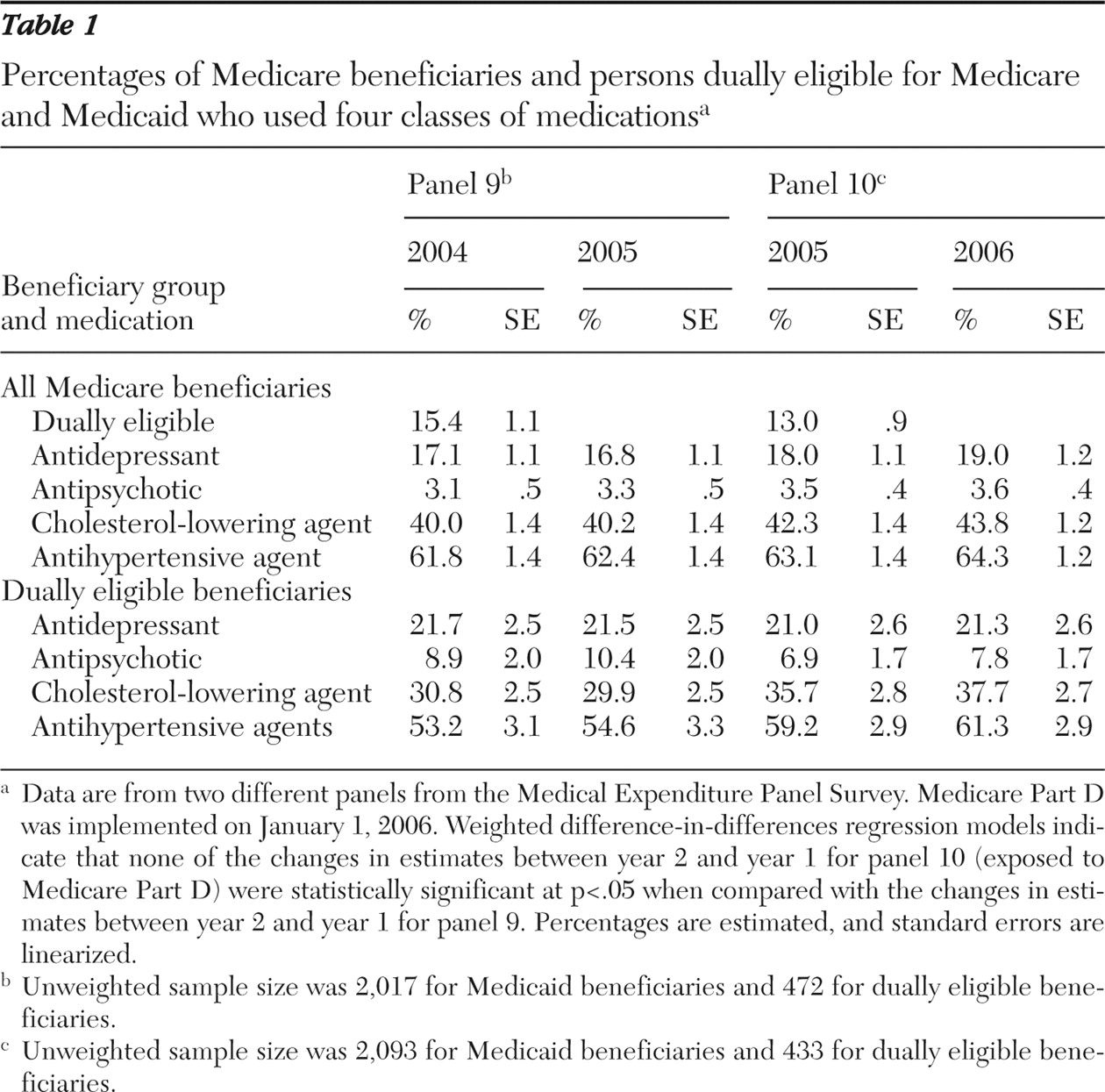
Between 17% and 19% of Medicare enrollees filled at least one prescription for antidepressant medications in each year during 2004 to 2006, and 3% to 4% filled a prescription for antipsychotic medications (
Table 1 ). A substantially greater number of beneficiaries (40% to 44%) used cholesterol-lowering agents, and 62% to 64% of beneficiaries filled prescriptions for antihypertensive agents each year. Compared with all beneficiaries, persons dually eligible for Medicare and Medicaid had considerably higher rates of psychotropic medication use and markedly lower rates of nonpsychotropic medication use. Antidepressant use by dually eligible beneficiaries ranged from 21% to 22%, and antipsychotic medication use ranged from 7% to 10%.
The rates of medication use were slightly higher in all medication categories in 2006 than in 2005. However, the rate of change in levels of use from year 1 to year 2 was not statistically different between the two panels for any of the four medication classes, indicating no strong evidence of a difference in access resulting from Part D, either among all Medicare beneficiaries or among those dually eligible for Medicare and Medicaid.
The rate of repeat use across years was consistently high for all medication categories, ranging from 85% to 97% across categories. The prevalence of repeat use of both psychotropic medications among dually eligible beneficiaries was somewhat lower in 2006 than in 2005, and the repeat use of cholesterol-lowering and antihypertensive agents increased. Differences between panels in repeat use were not statistically significant. New starts of medications in year 2 among beneficiaries who did not report taking the medication in year 1 ranged from 1.3% for antipsychotics to 16.3% for antihypertensives. New starts for all medications except antipsychotics were greater among panel 10 respondents (measured in 2006) than among panel 9 respondents (measured in 2005). Again, the differences between the panels were not statistically significant.
Discussion
Even though the large investment in Medicare Part D was intended to increase access to medications (
10 ), we found no strong evidence that Medicare Part D was associated with large changes in access to medications in the four classes of chronic medications examined here. This result is somewhat different from the recent finding by Donohue and colleagues (
11 ); their study also used MEPS data and found a statistically significant increase in the rate of antidepressant use from 2005 to 2006 among Medicare beneficiaries. Their analysis pooled the two panels in each year and therefore averaged the differences in the level of use between panel 9 and panel 10. In contrast, we treated each panel individually in our analysis, and we found that (but were unable to explain why) beneficiaries in panel 10 had a slightly greater level of use than beneficiaries in panel 9. This may explain in part differences in our estimate.
Prior studies have found that overall use of medications was estimated to increase by 13% among elderly persons as a result of Part D (
12 ). Increased utilization following Part D could come from increases in the intensive margin (levels of use among users) as well as increases in the extensive margins (number of users). Combined with the findings from the analysis presented here, these results seem to indicate that Part D was not associated with large increases in access to medications but potentially associated with increases in the level of use among medication users. Because each of the medication classes examined in this study represents a chronic health care treatment, the lack of a difference in filling rates is perhaps not surprising and may indicate that access to these medications among persons with appropriate diagnoses was already high before Part D (
13 ). The modest decrease in antidepressant continuation among dually eligible beneficiaries should be more closely monitored to determine whether Part D plans may have been more successful at deterring inappropriate use or whether any barriers discouraged appropriate use of psychotropics.
The fact that little evidence was found of differences in the rates of use across years of both protected psychotropic classes and nonpsychotropic classes with lower levels of protection suggests that the special status for psychotropics worked or, more cynically, that these protections were not necessary.
Conclusions
Access to medications is only one piece of quality of care and is a rather crude indicator of medication use. Important differences in the quantity and adequacy of medications used, the types of medications received, medication switching because of formulary restrictions, or changes in medication adherence may still be unearthed by future research using Part D claims data. Although this analysis used MEPS data from a nationally representative sample, these data are not ideal to monitor changes in adherence or to detect small differences in the rate of medication access. These results should be verified in a larger sample with greater power to detect differences. Concerns remain about the amount of medication received among medication users, and the concerns about of psychotropic access in Medicare are not likely to subside until Part D claims data have been analyzed.
Acknowledgments and disclosures
Dr. Farley has received consulting fees from Takeda Pharmaceuticals. Dr. Domino reports no competing interests.