Every year, more than $70 billion is spent on medical research in the United States alone, with a significant percentage of this funding coming from government agencies and charitable foundations
(1) . While these grants have contributed to impressive advances throughout medicine, quantifying the specific impact of this funding on the dual aims of disease amelioration and prevention represents an open challenge. How can we in the medical research community best report to taxpayers and philanthropists on the societal value produced by the monies entrusted to us? Or, to put it bluntly, what exactly is the return on investment for these research funds?
One approach to this question involves standard economic analyses
(2) . Indeed, using calculations to convert the impact of medical research on life expectancy and morbidity into dollars and cents, a strong argument can be made for the financial value of an investment in research
(3,
4) . In fact, some estimate that the economic benefit of medical research has been “exceptional,” with an estimated return to society of $70 trillion dollars since 1970
(5) . Economic analyses like these, when combined with assessments of the importance of scientific inquiry to society
(6 –
8), have been critical in justifying the expenditure of federal funds, particularly during tight fiscal periods. Yet these financial outcome measures seem removed from the core mission of academic medical research, which is by its nature a nonprofit enterprise. The potential financial payback to society does not seem to be a primary motivator to most medical scientists
(9) . Nor is it the type of outcome sought by the philanthropic agencies that increasingly fund our work
(10,
11) . In this sense, medical research entities are akin to not-for-profit “human services” agencies, where making dollars from dollars is not the primary goal.
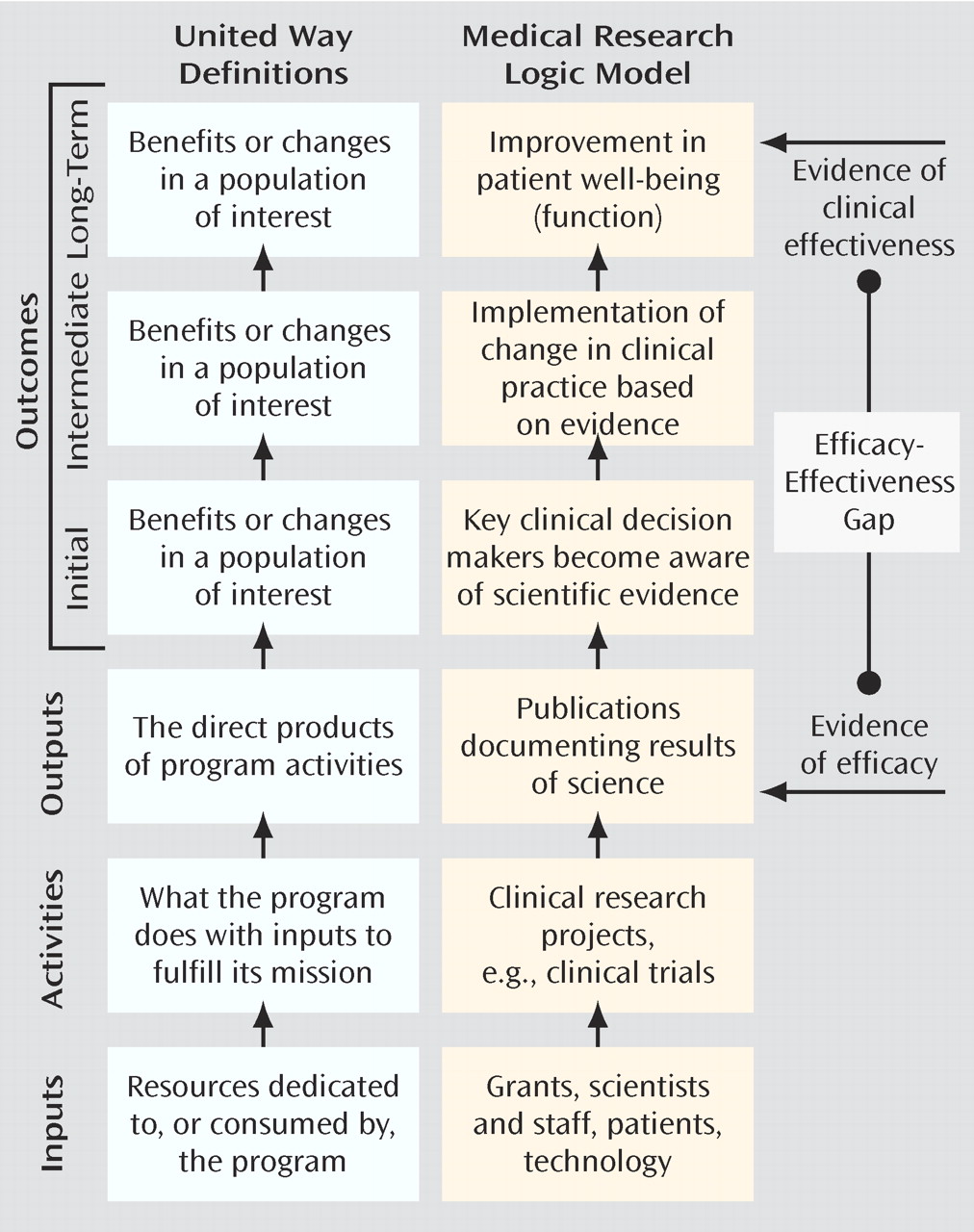
A second approach to return-on-investment reporting is provided by United Way in a helpful guide for nonprofit agencies
(12) . Their heuristic, which is based on a larger literature on performance-based management and program evaluation
(13), begins by recognizing that all organizations use various types of
inputs (personnel, funding, equipment, etc.) to carry out organizational
activities (
Figure 1 ). Most organizations then report on their
outputs, often in the form of a simple numerical tabulation of their activities. By measuring instead the
impact of these activities on the end-user, that is, how these activities contributed to the organization’s goals, one can better gauge the organization’s success. The measurement of
outcomes is the term applied to this impact assessment.
In this essay I argue that the current mode of reporting on the success of medical research is output based, whereas to optimally use the funds provided to us, an outcomes-based approach is needed. In addition to providing a clearer set of return-on-investment metrics to those who support our work, focusing on outcomes will force us to assess our current success (or lack thereof) in achieving our aims of cure or prevention of illness. While the general concept of outcomes assessment is applicable to all levels of research, from bench to bedside, examining the impact of basic medical research is a substantially more difficult task. In an effort to reduce this complexity, the focus of this article is on the area of medical research most proximal to clinicians: large-scale efficacy studies of therapeutic interventions. In addition, the principles presented here, though applicable to all fields of medicine, are of particular value to psychiatry, as funding for research in our field has historically lagged behind that provided for other disease states
(14,
15) . Framing the benefits of our work in terms of outcomes would provide a compelling argument for an expansion of resources for mental health research, helping to reverse this funding disparity. Thus, while I use the broad term “medical research” throughout, I provide examples specific to our work in psychiatry wherever possible.
Current State of the Art: The Output-Based Model
As we consider the application of the United Way model to medical research, it becomes apparent that the current mode of reporting on our activities is output-based (
Figure 1 ). The annual report to funding sources on research progress is largely a tallying of the quantity of our activities and our outputs. Typically it includes the number of research subjects enrolled (and their demographic profile), the number of projects completed, the number of abstracts presented at scientific meetings, and the number of manuscripts published in peer-reviewed journals. In some cases we might also document the additional “inputs” acquired with these funds, including the payment of research assistant salaries or the purchase of additional instrumentation. By measuring these inputs acquired and outputs achieved, the granting agency can presumably assess how the money is being used and how vigorously the scientist is pursuing his or her research objectives.
A similar model is used for internal evaluations of a scientist’s success in determining readiness for academic promotion. While attempts to integrate other criteria (e.g., devotion to clinical care or teaching) have received increasing attention
(16,
17), academic productivity remains the prime determinant of promotion
(16,
18 –
21) . Given the inclination to rely on objective, measurable factors, the number of dollars in grant funding (inputs) and the number of papers published (outputs) are most often at the center of this promotion evaluation.
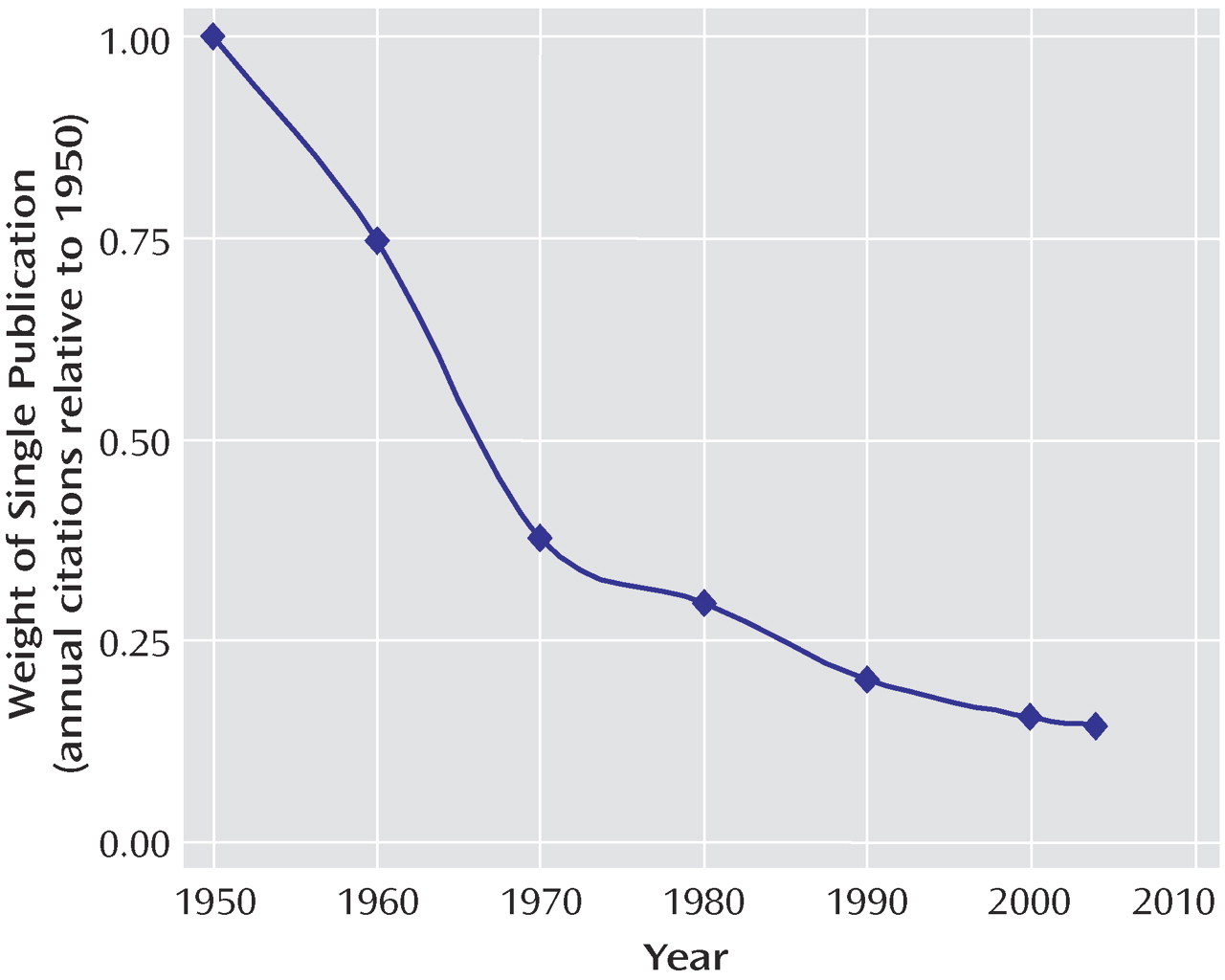
This output-based reward system has likely contributed to one well-documented phenomenon: an explosion in the number of medical manuscripts produced over the past several decades
(22,
23) . The expansion of science has been exponential, with a growth curve similar to “rabbits breeding among themselves”
(24) . Since 1950, for example, there have been approximately 15.3 million MEDLINE citations, with the annual number increasing every year. In an interesting form of academic inflation, if one paper published in 1950 were equal to a dollar, a single paper published today would be worth just over 14 cents (
Figure 2 ). While the sheer mass of this medical research output is a testament to the scientific efforts made over the past half century, it is also potentially overwhelming, even when broken down into today’s subspecialty niches. Busy clinicians, administrators, policy makers, and even full-time scientists simply do not have the capacity to keep up with the current literature
(25) . Perhaps this helps explain why as many as 46% of medical science manuscripts go completely uncited in the 5-year period after publication
(26,
27) .
Why Measure Outcomes?
It is certainly easier to measure outputs than outcomes: data on dollars of funding and number of papers published are solid, quantifiable, and readily obtained, whereas outcome measures are often “fuzzy” and difficult to assess. As we will see, measuring the downstream impact of our work represents an extraordinary challenge, in large part because of our limited understanding of the typical paths from science to clinical practice. In order to justify this added difficulty, there must be some substantial value added.
There are three strong advantages of measuring outcomes in assessing the impact of our medical research efforts. First, measuring outcomes provides a clear and meaningful message regarding the return on investment to the major funders of our work. Framing the benefits of science not in terms of papers published but rather in terms of its impact on people gives the granting agency a greater, and more immediate, sense of contribution. An outcomes-based report, especially one that estimates improvements in overall well-being or functioning, also supplies these agencies with a common metric for comparison across disease states, allowing for informed decisions on allocation of resources.
Second, these outcome measures serve as a compass to keep our research efforts, individually and collectively, on track. Focusing on outcomes requires an honest assessment of our progress toward our long-term goals. If our aim is to develop a cure for Alzheimer’s disease, how close are we to this end? Are we any closer today than we were 10 years ago? How do we know for sure? Are we closer to developing cures for some diseases than for others? Why? This approach also forces us to reexamine our assumptions about the paths to future success. The degree to which the publication of scientific results in medical journals actually changes clinical practice is largely unknown. Perhaps there are additional avenues, besides peer-reviewed journal publication, that could augment the impact of science on clinical care. Only when considering the communication of our results as an initial outcome step, and not an end point, does this concept become apparent.
Finally, the description of research progress in outcomes-based language makes explicit the societal good embedded within medical research, an important step in improving the attitude of the general public toward medical science. There is currently a sense of doubt about the safety of the treatments we have to offer
(28), an open skepticism toward health proclamations (“yesterday X was good, today X is bad”
[29] ), and, more ominously, a rising level of distrust toward medical research
(30,
31) . These unfavorable attitudes, though particularly prominent in some communities
(32,
33), are increasingly present worldwide
(34) . In a recent report, 79% of African Americans and 52% of white Americans indicated a belief that they might be used as “guinea pigs” in medical research without their consent
(35) . A high percentage of each group (46% and 35%, respectively) also felt that they may sometimes be exposed to unnecessary risk by their doctor for the sake of science. The response to this crisis in confidence has largely come in the form of increased oversight and monitoring of our work by outside agencies. Engaging and informing the general public about the benefits of our work represents a proactive internal response to this issue.
Moving to an Outcomes-Based Approach
United Way’s approach to outcomes measurement involves the development of a “logic model”—a series of if-then statements describing the path to the desired outcome. The logic model can help us identify some near-term outcomes that can serve as leading indicators of long-term success. Given the estimated delay of 17 years between initial reports of efficacy and the successful implementation of a new therapeutic agent in clinical practice
(36,
37), it may be some time before the true impact of this research is actually known. Having some early measures of our impact is therefore critical.
While we might gain consensus on an appropriate long-term outcome for our work (e.g., improved mental and physical functioning, improved quality of life), determining reasonable near-term benchmarks is a more difficult proposition. Surprisingly, despite the current enthusiasm for translational research, the steps involved in the clinical implementation of a scientifically validated therapeutic intervention are not well worked out. The path described in this essay represents an initial attempt at describing this complex process, informed by the work of a few groups engaged in this type of program evaluation research
(11,
37 –
39) . It is important to emphasize that this model represents a rough estimate, and through an iterative process it will come to reflect real-world complexity. Also note that this approach does not exclude the use of qualitative methods, including the case study approach, to assess the impact of our work. In fact, these types of detailed analyses, with a focus on examining the dispersion and implementation of science at the single-project (or single-grant) level, have an important complementary role in further refining and improving our understanding of the gap between science and clinical care
(11,
39 –
41) .
Figure 1 outlines the core components of the process: research is published in journals (output); journals are read by physicians and other key clinical decision makers (initial outcome); these clinicians implement a change in practice (intermediate outcome); and a practice change leads to improvement in patients’ lives (long-term outcome). When viewed in this manner, it becomes clear that the relationship between outputs and outcomes is analogous to the relationship between the demonstration of research efficacy and the clinical effectiveness of a particular intervention. Substantial evidence suggests that this gap between efficacy and effectiveness is quite large, as many efficacious treatments are not implemented properly (and often not implemented at all) in the real-world setting of clinical medicine
(42 –
46) . From the standpoint of the clinical scientist following an output-based approach, the responsibility for this gap lies squarely on the shoulders of the treating physician. Once the data have been published, any inconsistencies between this evidence and clinical practice represent either a lack of awareness of the evidence or a lack of the clinical acumen necessary to appropriately apply the evidence. The output model essentially suggests that “if you build it (publish it), they will come,” perhaps with the additional proviso, “as long as it is in a decent journal.”
The outcome-based model, on the other hand, suggests that the successful implementation of a new intervention is a shared responsibility and, indeed, should serve as the basis by which we judge the quality of our research efforts. Any failures in the system are partially ours, and we should identify ways that we as scientists might help to fix them. This collaborative responsibility only becomes apparent once we begin to think beyond the outputs, when we begin to assess our success via the near- and long-term outcomes described below.
Initial Outcome: Key Clinical Decision Makers Become Aware of the Research Evidence
How can we best get research findings to those responsible for implementing clinical change? If considered at all, this step is likely viewed in terms of the journal’s “impact factor,” a value based on the number of other articles that cite articles published in the journal over a 2-year period
(47) . The impact factor has been shown to be highly correlated with both scientific selectivity (manuscript acceptance rate)
(48) and perceived journal quality among scientists
(49) . Because it is seen to be reflective of academic excellence, the impact factor of the journal in which one publishes is increasingly being used as an outcome measure, for both external reporting and internal advancement
(50 –
52) .
Impact factor alone, however, is unlikely to be predictive of subsequent clinical implementation. First, a journal’s impact factor measures the lateral spread of its output to other scientists, not the vertical spread of that information to the clinical domain. As most clinicians are not publishing papers as medical scientists, their reading habits are not necessarily captured by a citation-based measure
(53) . Studies of the reading habits of psychiatrists
(54) and surgeons
(55) show that the most widely read journals in these fields do not all have high impact factors. The correlation between the perceived quality of a journal and its impact factor is significantly higher among research physicians than among physicians in clinical practice
(49) . Similarly, the relationship between the perceived importance of a journal and its impact factor is tighter in “scientific” subfields than in clinical domains such as nursing
(56) . In fact, one study found an inverse correlation between a journal’s perceived clinical usefulness and its impact factor
(57) . Finally, Grant and colleagues
(37) found that nearly 75% of publications included in comprehensive clinical guidelines came from clinically focused journals, a disproportionate number when compared with the citation pattern of biomedical articles in general.
Perhaps more important, the information used by clinicians to implement change in practice may not come from a journal publication at all
(38) . The medical journal is only one node in a complex network of information sources available to clinicians in practice, which now includes professional meetings, continuing medical education (CME) seminars, discussions with colleagues, visits from pharmaceutical representatives, textbooks, newspapers, Web sites, and even feedback from patients
(55,
58,
59) . Surveys indicate that clinicians have only a few hours per week to read the literature and that they tend to read only a handful of the 15,000 biomedical journals currently in publication
(53 –
55,
59 –
64) . It is therefore not surprising that clinicians may turn to secondary and tertiary channels of information to learn about recent scientific evidence
(59,
65) . The journal impact factor alone is unlikely to capture the degree to which these multiple channels transmit scientific information to clinicians and decision makers.
Measuring the penetration of research into the clinical domain is thus a complex but important first step in assessing the impact of science on patient health. As we begin to look beyond the impact factor for better measures of information awareness, three broad approaches hold the most promise:
We will continue to rely on scientometrics, the discipline involved with measuring the communication of scientific information, to provide us with tools to track our outcomes. This approach is most appealing to the individual scientist, as scientometric values could be made available online, allowing for easy, low-cost outcomes reporting. While there are no perfect measures at present, one novel option uses a combination of the journal impact factor and a number of other measures, including the citation rates of our research in nonjournal media, such as newspapers, patents, and clinical guidelines
(56,
58,
66) .
Ideally, we would like to know the actual number of people reading (and understanding) our work. With paper-only journals, actual readership is difficult to assess, especially on an article-by-article basis. As more and more clinicians access journal articles via the Internet, our ability to track readership will increase. Although currently this information is held by the publishers, the increasing use of open-source scientific journals, as well as new regulations requiring public access to research funded by the National Institutes of Health, may soon provide the individual scientist with ready access to this Web tracking data.
Surveys could be used to gauge penetrance of information. These could range from very small scale (tracking poster readership at a conference) to very large scale (tracking nationwide awareness of a specific set of research findings or reports). This type of assessment might be particularly relevant as a follow-up to the publication of large multicenter trials, treatment recommendations, and algorithms.
Intermediate Outcome: Change in Clinical Practice Is Implemented
Measuring awareness of a particular research finding is insufficient, for one simple reason: there is a tremendous difference between knowing and doing. As Wilson and colleagues noted, “The naive assumption that when research evidence is made available it is routinely accessed by practitioners, appraised, and then applied in practice is now largely discredited. While awareness of a practice guideline or a research-based recommendation is important, it is rarely, by itself, sufficient to change practice”
(67) . Measuring the degree of implementation—the actual change in practice stimulated by a particular scientific finding—is therefore a critical intermediate indicator of success.
The rate of implementation of our existing scientific successes is suboptimal. Despite the ubiquity of published guidelines and treatment algorithms, examples of the underutilization of efficacious treatments are abundant across many medical disciplines, including psychiatry. Less than half of patients diagnosed with depression
(68) or panic disorder
(69) in the primary care setting receive any pharmacological treatment. Similar figures have been reported for patients treated primarily by psychiatrists, including those with psychotic disorders
(70) . Even when patients receive medication, they often receive inadequate doses for inadequate durations. Less than a third of all patients with schizophrenia, for example, are treated with a recommended dose of an antipsychotic (71). The implementation rates of scientifically vetted psychosocial interventions are even more abysmal. In one study, only 17.6% of patients with a depressive or anxiety disorder received appropriate counseling
(72) . Among patients with schizophrenia who had regular contact with their families, family psychoeducation was provided to as few as 10% of patients and their families
(71) . A comparably low percentage of patients receive other valuable interventions, including vocational rehabilitation and social skills training
(73) .
There are several causes of this implementation gap, including many beyond the direct control of clinicians or scientists. Economic and regulatory factors can have a tremendous role in the usage rates of specific treatments or technologies
(74) . Even consumer demand can shape practice patterns, as evidenced by the increased rate of third-party reimbursement for complementary and alternative therapies
(75) . These factors notwithstanding, there are a number of ways in which medicine as a field can act to improve concordance between evidence and clinical practice. Most of the focus thus far has been on the clinical end of the process, including the use of feedback to encourage performance at or above established benchmarks and enforcement of practice guidelines through financial incentives or penalties
(76) . By better understanding the processes by which change is accepted or rejected by the clinical end user, we in the research community can also play a role in these programs of process improvement.
As described by Harvey Fineberg in a 1985 Institute of Medicine report, there are 10 key factors that influence the diffusion of new technology (or therapeutics) into the clinical domain
(77) . Half of them relate specifically to the innovation itself. Understanding these five technology-related factors can help scientists increase the implementation of their findings:
The channel of communication. Some sources of information simply have more influence on decision making than others. The degree to which physicians are influenced by information from one journal as compared with another, presumed to be related to journal impact factor, is actually largely unknown. Other forms of information delivery, including direct personal contact, may have a greater impact than academic publications. Indeed, although physicians indicate that they are swayed more by published guidelines and CME events than by advertisements, industry literature, and pharmaceutical representatives
(59), there is some evidence to suggest that the latter sources are as influential as the former
(78 –
81) . The use of “opinion leaders” does not need to be confined to the pharmaceutical industry. In fact, a recent report highlights the role of word-of-mouth communication (or “buzz marketing”) in reducing the inappropriate use of antibiotics
(82) . For clinical scientists who are interested in seeing their research findings implemented in the clinical domain, these data suggest that a more active approach to information dissemination may be necessary
(53) .
The coherence of the findings with prevailing clinical beliefs. The driving factor in determining the perceived scientific validity of a research article is its coherence with the physician’s own experience, cited as being “always” or “often” important by 88% of those physicians surveyed, followed by discussion with colleagues (73%) and reliance on the peer review process (48%)
(59) . Preconceived beliefs, based on either personal experience or prevailing paradigms, can therefore serve as stern gatekeepers to the entry of new ideas. The limited use of cognitive behavior therapy for the positive symptoms of schizophrenia, for example, may partially be accounted for by the belief among some psychiatrists that brain-based symptoms require biologically based treatments
(83) . Thus, if a finding requires thinking that is “outside the box,” preparatory information may be necessary before the evidence is accepted and implemented.
The quality and quantity of the evidence. Scientists will be reassured to know that the quality of the evidence is still an important factor in determining whether a change in practice is implemented. The publication of a well-conducted, large-scale, randomized, controlled trial in a respected journal has been shown to have an impact on subsequent prescribing practice
(84,
85) . This may be truer for the implementation of a new therapy than the abandonment of an old one, where other factors, including sensationalistic media coverage, can play a role
(86) . Somewhat surprisingly, the data on the impact of evidence quality are sparse. Several important questions remain largely unexamined, including the relative impact of a number of smaller studies reporting similar results as compared with a single large-scale trial.
The nature of the change. If the change in practice is easy to learn, fits well within the existing clinical structure, and is relatively inexpensive, it is more likely to be instituted. If any of these qualities are not met, the intervention either will not be used at all or will be modified (simplified) by end users to fit their means
(87) . In the latter case, the revised intervention may or may not retain the efficacy of the original innovation. For example, faithful adherence to a manualized treatment model has been shown to be related to better clinical outcomes for a variety of mental health interventions, including individual psychotherapy
(88,
89), assertive community treatment
(90), and family-based therapeutic approaches
(91) . These studies suggest that interventions designed to be congruent with current clinical realities may see better translation from laboratory to clinic
(92) .
The impact of the change on key clinical branch points. Physicians are more likely to implement a novel therapy (or use a novel technology) if it helps at an important clinical branch point or improves a symptom that is unaided by existing treatments. There are few (if any) examples of the use of technology in this manner within current psychiatric practice. Moving forward, we should consider these areas of unmet clinical need, as they serve as important guides in the development of novel technologies. The use of deep brain stimulation in treatment-refractory patients with obsessive-compulsive disorder
(93) and the potential use of neuroimaging and genetics in predicting medication response
(94) are two exciting examples of this approach that hold promise in the near future.
By taking a more active stance toward the development and promotion of technology, we raise the likelihood that the technology will actually be used. Measuring the implementation rate is admittedly a challenge. At a local level, physician surveys or even electronic medical record searches may provide some information on the use of a particular treatment. More accurate information would likely require access to a database of physician prescribing practices or medication insurance claims. The impact of a specific large-scale trial or a set of clinical practice guidelines could be assessed by comparing prescribing practices before and after publication. An even better approach, one that would provide information on the relationship between awareness and implementation, would involve an assessment of educational activities and prescribing practices in a closed system, such as a large health maintenance organization (HMO). By having a subsample of physicians within the HMO access all journals and CME materials via monitored Web-based portals, one could quantify actual reading patterns and then cross-reference this with actual changes in prescribing or orders for diagnostic tests.
Long-Term Outcome: Implementation of Change Leads to Patient Benefit
Even when scientifically supported interventions are implemented in the community, their effectiveness often falls short of the efficacy demonstrated in clinical trials
(42) . There are a number of reasons for this phenomenon, including lower rates of medication adherence, a higher prevalence of adverse events due to drug-drug interactions, and the impact of multiple comorbid conditions
(89) . Thus, the real-world performance may not replicate that demonstrated in a “clean” and controlled research environment, further contributing to the efficacy-effectiveness gap.
Developing clinical trials with greater real-world validity (i.e., effectiveness trials) represents another way in which the medical research community can play a greater role in narrowing this gap
(95) . This research strategy aims to maintain scientific rigor while incorporating all of the “confounds” associated with clinical reality. The recent paper by Roy-Byrne and colleagues
(96) demonstrating the feasibility and effectiveness of a primary care-based intervention for panic disorder is a nice example of this approach. Other large-scale studies using similar methods for bipolar disorder
(97) and schizophrenia
(98) are currently under way. As these trials are completed, we may begin to develop measures of clinical efficacy that better reflect the eventual therapeutic effectiveness of a particular intervention.
In measuring patient benefit, we should increasingly consider the use of general measures of functioning or well-being, such as the disability-adjusted life-year (DALY)
(99), the quality-adjusted life-year (QALY), and the Medical Outcomes Study 36-item Short-Form Health Survey (SF-36)
(100) . Although these measures may seem vague and unsatisfying when compared with disease-specific scores, they contain tremendous comparative value for reporting purposes. They serve as a common metric to allow funders to assess the value of an investment across disease states. How else could they determine whether the development of a drug providing a 12% decrease in patients’ Beck Depression Inventory scores is a better return on their investment than a psychosocial intervention yielding an 18% improvement in patients’ Yale-Brown Obsessive Compulsive Scale scores? Furthermore, given the significant impact of major psychiatric illness on overall functioning, framing our outcomes in this manner highlights the potential economic benefit of psychiatric treatment in general, providing a persuasive message not only to the funders of our research but also to third-party payers of clinical care.
Conclusions
It is by measuring three outcomes—awareness, implementation, and patient benefit—that we can provide a real sense of the clinical return on an investment in medical research. Admittedly, our ability to provide this information is limited at present. There are many areas where interesting questions outnumber practical answers, in part because of our surprisingly limited understanding of the path from scientific innovation to clinical use. In addition, the costs involved with developing better measures, such as the use of “market research” to measure the impact of publications on awareness and prescribing practices, would likely be nontrivial. The solutions to these limitations will require creative thought and a new level of cross-disciplinary collaboration.
This should not dissuade us from taking the initial steps, however imperfect, toward examining the real outcomes of the work we are doing. The shift to this mode of reporting will have substantial benefits for our ability to track progress, raise funds, and improve the overall standing of medical research in the public eye. Furthermore, this change from an output-based to an outcomes-based perspective is a critical step in examining the role that clinical scientists can play in decreasing the well-documented efficacy-effectiveness gap. Without this shift, there will be little incentive for researchers to look beyond the outputs of their work and little understanding of the specific areas where they can help in the implementation of this work.
We are entering a new era of accountability. Several broad institutions, from business to education to medicine, are being called upon to quantify their quality. We in medical science can also begin this type of self-evaluation, with the goal of improving not only the quality of our efforts but also our capacity to report on what we’ve done.