Baseline
Subject characteristics
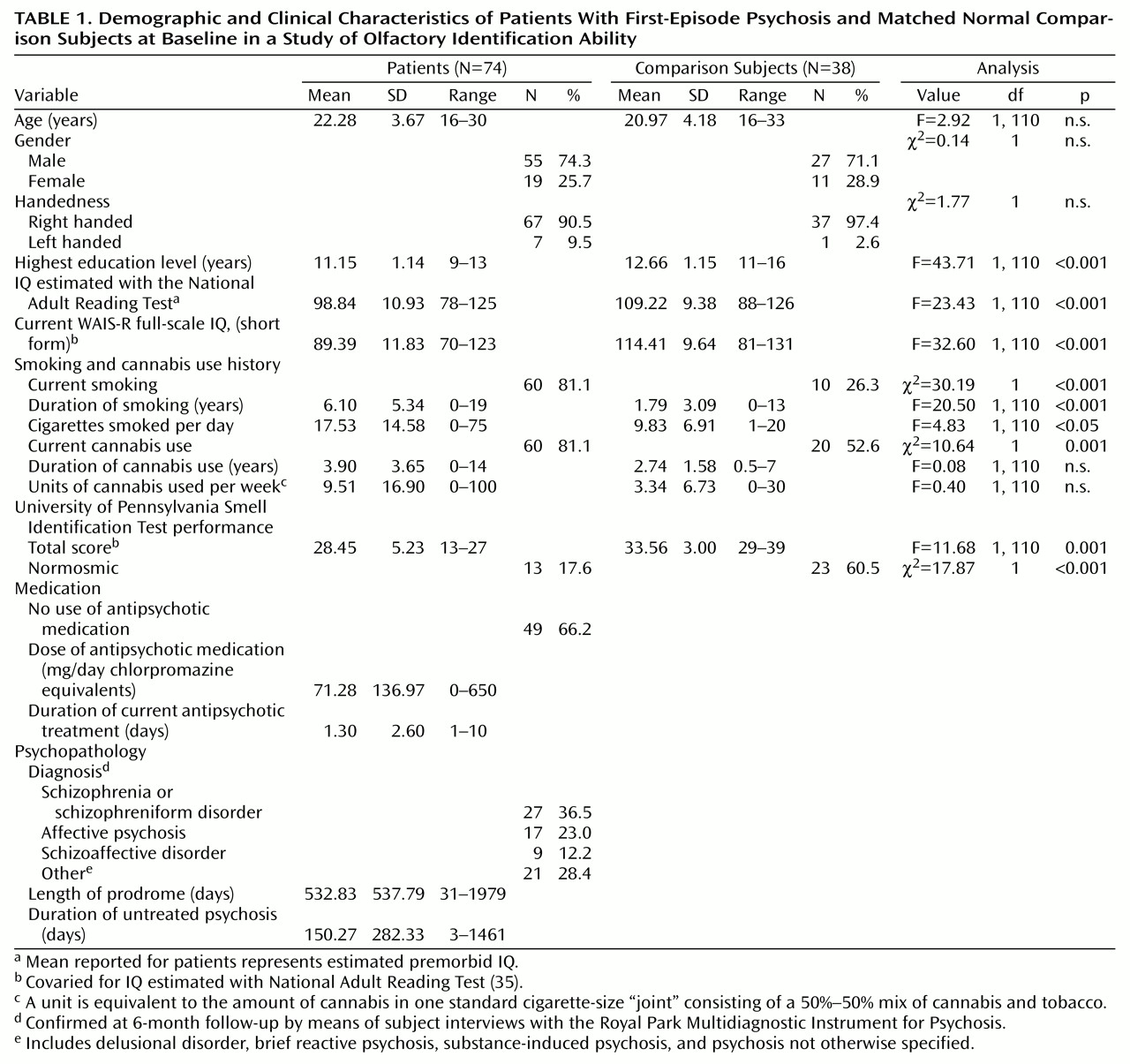
Table 1 presents the demographic, cognitive, and clinical characteristics of all subjects at baseline. Diagnoses for the first-episode patients were confirmed at 6 months postadmission by using the Royal Park Multidiagnostic Instrument for Psychosis and are reported in
Table 1. The proportion of patients with schizophrenia or schizophreniform disorder in this study (36.5%) was low compared to the proportion of patients with these disorders in the general Early Psychosis Prevention and Intervention Program (55%)
(30).
There was no significant difference between the patient and comparison groups in age and sex. However, the two groups differed in highest level of education and in IQ estimated with the National Adult Reading Test (
Table 1), with the comparison group having slightly higher scores on each of these measures. In the analyses that follow, IQ estimated with the National Adult Reading Test was used as a covariate where appropriate (unless otherwise stated).
A significantly greater proportion of the patient group than of the comparison group smoked cigarettes. Among the smokers in the two groups, the patients smoked significantly more cigarettes per day and had smoked for a significantly longer period than the comparison subjects. In addition, significantly more patients than comparison subjects reported using cannabis. Of those using cannabis, patients and comparison subjects did not differ in the number of units of cannabis smoked per week or in how long they had used cannabis.
Smell identification
The results on the University of Pennsylvania Smell Identification Test for patient and comparison groups at baseline are shown in
Table 11. The first-episode psychosis patients achieved a significantly lower mean score than did comparison subjects. No group-by-sex interaction was observed (F=0.80, df=1, 109, n.s.).
Effects of IQ
The patient group and the comparison group differed on current full-scale IQ derived from the WAIS-R and on IQ estimated with the National Adult Reading Test, which represented premorbid IQ for the patients (
Table 1). The discrepancy between IQ estimated with the National Adult Reading Test and current IQ was significantly more pronounced in the patient group (difference=–9.45) than in the comparison group (difference=5.19) (F=46.20, df=1, 102, p<0.001, for current IQ covaried for IQ estimated with the National Adult Reading Test). Although the difference between groups in IQ estimated with the National Adult Reading Test was significantly associated with poorer performance on the University of Pennsylvania Smell Identification Test (F=7.70, df=1, 80, p<0.01), the difference in smell identification ability between the groups remained significant even after controlling for difference in IQ (F=12.46, df=1, 80, p=0.001).
Effects of cigarette smoking and cannabis use
To examine the effects of cigarette smoking and cannabis use on performance on the University of Pennsylvania Smell Identification Test, the patient and comparison groups were divided into users and nonusers. Smoking had no significant effect on smell identification ability in either study group (F=2.20, df=1, 77, p=0.14), and no interaction of study group and smoking was found (F=0.70, df=1, 77, p=0.41). Similarly, there was no effect of cannabis use on smell identification ability in either study group (F=0.13, df=1, 79, p=0.73) and no interaction of study group and cannabis use (F=0.18, df=1, 79, p=0.67, with IQ estimated with the National Adult Reading Test as covariate).
University of Pennsylvania Smell Identification Test score and age when THC use began were significantly correlated in the patient group (r=0.32, p<0.05) but not in the comparison group (r=–0.04, n.s.), indicating that poorer smell identification ability was associated with earlier age of initiating THC use in patients with psychosis. No relationships were found in either group for smoking history measures.
Effects of diagnosis, symptoms, and medication
Within the patient group, there was no significant difference between diagnostic subgroups in performance on the University of Pennsylvania Smell Identification Test (mean=29.86, SD=4.26, for patients with schizophrenia or schizophreniform disorder; mean=30.75, SD=5.19, for patients with affective psychosis; mean=28.67, SD=2.88, for patients with schizoaffective disorder; and mean=27.38, SD=4.86, for patients with other psychoses) (F=1.50, df=3, 54, n.s.). To examine the effects of medication, the patient group was subdivided into those who were taking antipsychotics (N=25) and those who were assessed to be neuroleptic naive (N=49). There was no difference in smell identification scores between the two subgroups (mean=28.95, SD=5.26, for those receiving antipsychotics and mean=29.33, SD=4.30, for those who were neuroleptic naive (F=0.09, df=1, 72, n.s.). There were no differences in smell identification ability between patients who were receiving anxiolytics (N=9) or anticholinergics (N=8) and those who were not receiving those drugs.
Higher scores on the Manchester Scale negative symptom item of “flattened affect” was associated with poorer performance on the University of Pennsylvania Smell Identification Test (r=–0.35, p<0.05). No associations were found between score on the smell identification test and any medication or other clinical variables, including duration of untreated psychosis or length of the prodrome.
Follow-Up
There were no significant differences at baseline in age, sex, or smell identification ability between subjects who were followed up (both patients and comparison subjects) and those who were not followed up. Patients who received a follow-up assessment had a significantly higher mean current full-scale IQ at baseline (mean=92.00, SD=13.29) than the patients who were not followed up (mean=84.06, SD=11.27) (F=7.39, df=1, 72, p=0.008). This difference was not found for the comparison subjects.
The 40 patients assessed at follow-up differed diagnostically from those who were not followed up. Fewer patients with other psychoses (N=5) were followed up, compared to those with schizophrenia or schizophreniform disorder (N=16), affective disorders (N=12), and schizoaffective disorder (N=7) (χ2=11.94, df=3, p<0.008). There was no difference in the dose of antipsychotics or the use of anticholinergics at baseline between those who were followed up and those who were not.
As expected, the mean daily dose of antipsychotic medication (in chlorpromazine equivalents) at follow-up (mean=88.3 mg, SD=129.09) was higher than at baseline (mean=31.62, SD=73.16) (Wilcoxon z=–2.6, p=0.009, N=32). For patients who were assessed at follow-up, the mean Manchester Scale score for positive psychotic symptoms decreased significantly from baseline to follow-up, indicating that the patients’ clinical state had improved (scores decreased from mean=2.16, SD=1.13, to mean=0.80, SD=0.90 for delusions, Wilcoxon z=–4.08, p<0.001, N=40; from mean=1.32, SD=1.28, to mean=0.62, SD=0.88 for hallucinations, Wilcoxon z=–3.3, p=0.001, N=40; and from mean=1.45, SD=1.09, to mean=1.03, SD=0.90 for incoherence of speech, Wilcoxon z=–2.00, p<0.05, N=40). No significant improvement in negative symptoms was found.
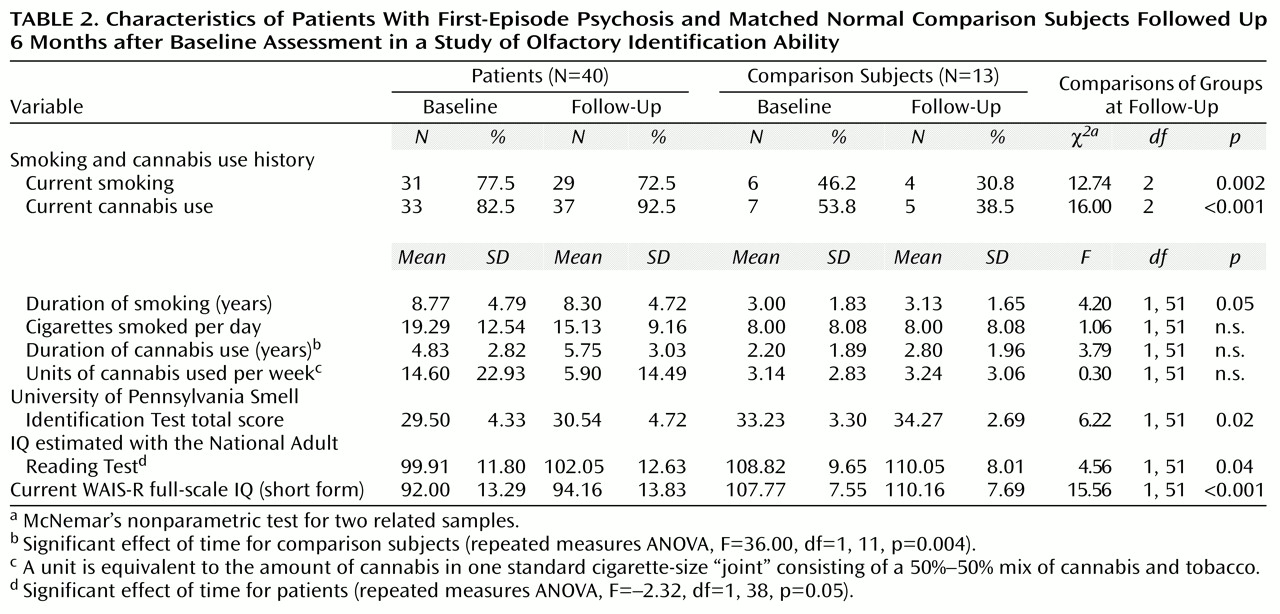
Data on substance use and cognitive measures for the 40 patients and 13 comparison subjects reassessed at follow-up are presented in
Table 2. Differences between the patient and comparison groups in smell identification ability, IQ, and substance use at 6-month follow-up were similar to those at baseline. There was no significant change between baseline and follow-up in the proportion of patients or comparison subjects who smoked cigarettes or used cannabis. For the patient group, IQ estimated with the National Adult Reading Test showed a modest but significant increase at follow-up.
Although the difference in smell identification scores between groups was maintained between the two time points (F=9.25, df=1, 41, p<0.01), there was no significant change in smell identification scores between assessments in either the patient group or the comparison group (effect of time: F=1.86, df=1, 41, n.s.; group-by-time interaction: F=0.01, df=1, 41, n.s.). No significant association was found between smell identification ability and medication dose at follow-up assessment. As at baseline, there were significant negative associations between the University of Pennsylvania Smell Identification Test score and the negative symptom items of flattened affect and poverty of speech (r=–0.39, p<0.05, and r=–0.34, p<0.05, respectively). Further, change in the University of Pennsylvania Smell Identification Test score between the two assessments was significantly associated with change in score for flattened affect (r=–0.43, p=0.03), indicating that improved performance on the smell identification test was associated with decreased severity of flattened affect. The association between change in the University of Pennsylvania Smell Identification Test score and change in poverty of speech did not reach significance (r=–0.36, p=0.07).