The rapidly escalating abuse of methamphetamine in the United States
(1) imposes a sense of urgency for understanding its effects on the human brain and its medical consequences. Methamphetamine is a particularly problematic drug in that it is not only highly addictive
(2) but also can be manufactured by small clandestine laboratories, making the control of methamphetamine supplies difficult
(3). As a result, epidemic pockets of methamphetamine abuse have recently developed in different areas of California and in sections of the southern and midwestern United States
(4). This rise in methamphetamine abuse has also been reported for other areas in the world
(5). As methamphetamine abuse rises, concern about its potential neurotoxic effects to the human brain increases, since methamphetamine administration in laboratory animals has resulted in profound and long-lasting toxicity to the brain. Particular damage has been documented in dopamine terminals
(6,
7). Since the doses of methamphetamine administered to laboratory animals differ from those used by human abusers of the drug, it has been unclear whether similar deficits occur in human methamphetamine abusers. To our knowledge, only two studies with human data have been published: a postmortem study of 12 methamphetamine abusers
(8) and an imaging study of six methamphetamine abusers
(9). These studies reported dopamine transporter reductions in the brain, which suggests that methamphetamine at the doses abused by humans also affects the dopamine terminals. However, there are no data on the functional significance of these changes. The purpose of this study was to assess if we could document the dopamine transporter changes and assess their functional significance in a larger group of detoxified methamphetamine abusers.
Methamphetamine abusers and comparison subjects without a history of drug abuse underwent positron emission tomography (PET) scans following administration of [
11C]
d-threo-methylphenidate (a dopamine transporter ligand
[10]) to measure dopamine transporters, which serve as a marker for dopamine cell terminals. We also administered a battery of neuropsychological tests to assess the effects of methamphetamine abuse on motor activity and cognition.
Method
Subjects
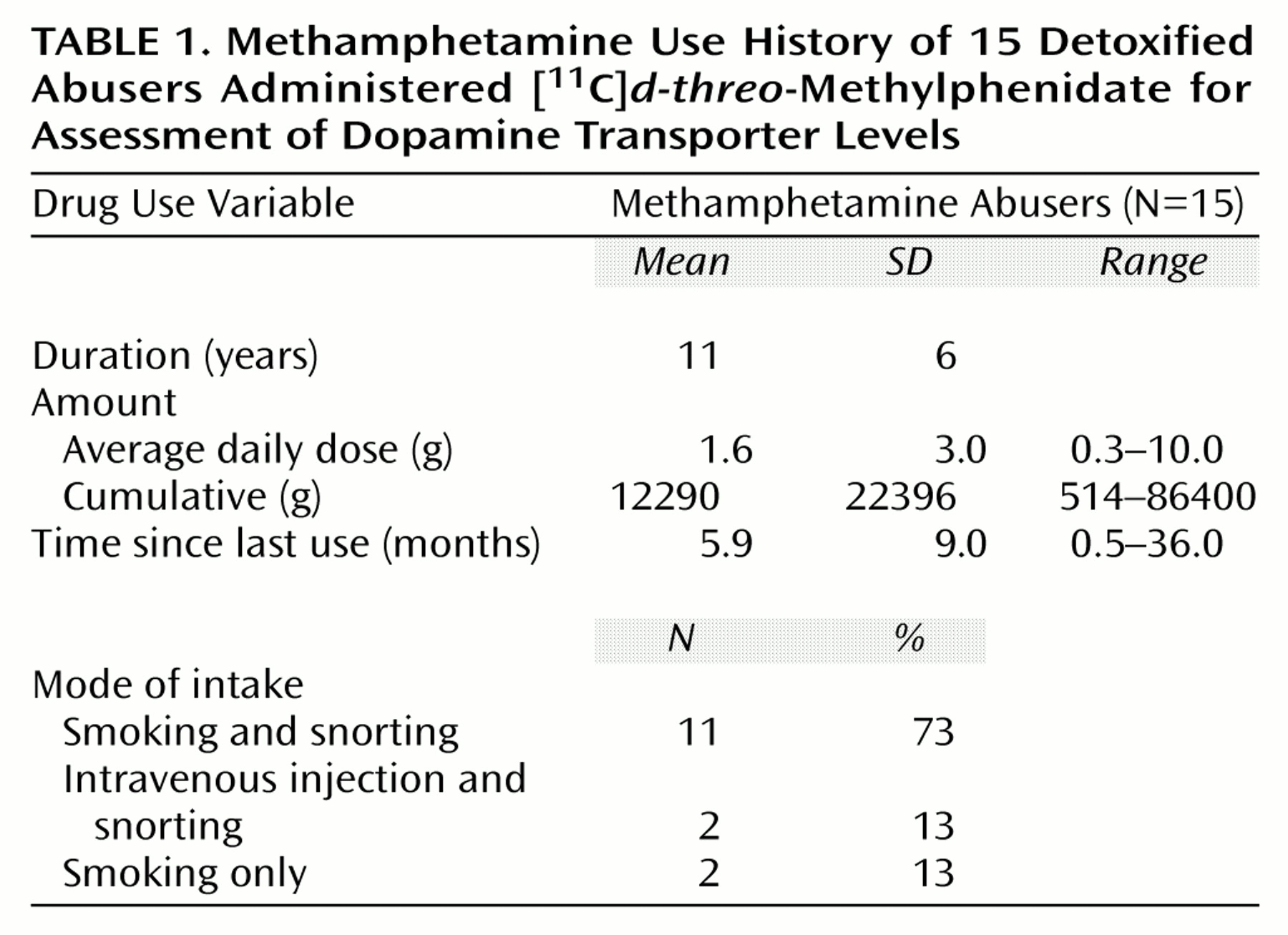
Fifteen subjects (six men and nine women; mean age=32 years [SD=7], mean IQ=100 [SD=9]) who fulfilled DSM-IV criteria for methamphetamine dependence were studied. Characteristics of the drug use history for these subjects are presented in
Table 1. The subjects were included in the study if their average methamphetamine use involved at least 0.5 g/day, at least 5 days per week, for at least 2 years. Subjects were also required to have abstained from methamphetamine use for at least 2 weeks, which we confirmed by conducting a urine toxicology screening examination. Subjects were excluded from the study if they were seropositive for HIV or had a history of a comorbid psychiatric or neurological disorder, medical illness, current or past history of drug dependence (other than methamphetamine or nicotine), or a history of head trauma. Subjects were recruited from drug rehabilitation centers in the Los Angeles area. Detailed medical and drug use histories and results of physical, neurological, and psychiatric evaluations (conducted by L.C.) were independently corroborated at Brookhaven National Laboratory (by G.-J.W.). Blood test screenings, including HIV serology, were conducted to determine whether abnormalities were present. Twelve of the methamphetamine abusers had last used methamphetamine within 6 months of the study (range=2–24 weeks); the other three had not used it for at least 11 months (range=11–36).
Comparison subjects were 18 healthy volunteers (12 men and six women; mean age=31 years [SD=7], mean IQ=107 [SD=10]) who responded to a local advertisement. Exclusion criteria were the same as those for the methamphetamine abusers except the current or past history of drug problems included methamphetamine abuse. A complete medical and psychiatric examination was performed to ensure lack of medical, psychiatric, or neurological disease (G.-J.W., D.F.). The same screening laboratory tests as those given to the methamphetamine abusers (except for HIV serology) were used.
No subject was taking medications at the time of the study, and prescan urine tests were done to ensure the absence of psychoactive drug use in both the methamphetamine abusers and the comparison subjects.
Written informed consent was obtained from the subjects after the procedures had been fully explained. The study was approved by the institutional review boards at Brookhaven National Laboratory, the State University of New York at Stony Brook, and the Harbor-UCLA Medical Center.
Neuropsychological Evaluation
Within 2 weeks of the PET scans, we administered a neuropsychological battery to the methamphetamine abusers that was designed to include measures sensitive to functional deficits of the frontal lobe and the striatum
(11). Five areas were assessed. First, motor function was rated by performance on two measures: the Timed Gait task, in which gross motor function is assessed by having the subject walk as fast as possible in a straight line, and the Grooved Pegboard task, in which fine motor coordination is assessed by having the subject insert pegs into small holes angled in different directions as quickly as possible. Second, attention was rated by performance on four tasks: the California Computerized Assessment Package
(12), in which the subject responds as fast as possible to numbers and letters on a computer screen; the Symbol Digit Modalities Test, in which the subject matches numbers with symbols; the Trail Making Test, in which the subject draws lines connecting consecutive numbers or numbers that alternate with letters; and the Stroop Interference Test, in which the subject reads color names printed in incongruent ink colors and has to suppress the tendency to say the word instead of the color. Third, memory was rated by performance on the Rey Auditory Verbal Learning Test, in which the subject has to learn and recall lists of unrelated words immediately, after a time delay, and after a distractor. Fourth, depressive symptoms were measured with the Center for Epidemiologic Studies Depression Scale
(13). Last, general intelligence was rated with the New Adult Reading Test Revised, which gives an estimate of verbal intelligence.
Scans
PET scans were performed by using a CTI 931 scanner (Siemens, Knoxville, Tenn.) (spatial resolution: 6 × 6 × 6.5-mm full width at half maximum). Dynamic scans that followed previously described procedures
(11) were started after intravenous injection of 4–8 mCi of [
11C]
d-threo-methylphenidate (specific activity >0.4 Ci/μmol at time of injection); the scans lasted a total of 84 minutes.
Image Analysis
Regions in the striatum (caudate, putamen) were obtained from three sequential planes and in the cerebellum from two sequential planes and were drawn on the averaged emission scans (activity between 10 and 84 minutes). Regions in the striatum were selected in multiple planes to increase the reproducibility of the measures
(14). The regions were then projected to the dynamic emission scans to obtain tissue time activity curves. By means of a graphical analysis technique for reversible systems
(15), these tissue time activity curves along with the time activity curves for unchanged tracer in plasma were used to calculate in the striatum and cerebellum the transfer constant (K
1) from plasma to brain of [
11C]
d-threo-methylphenidate and its distribution volume, which corresponds to the equilibrium measurement of the ratio of tissue concentration to plasma concentration. The ratio of distribution volume in the striatum to that in the cerebellum, which corresponds to (B
max/K
d) + 1 and is insensitive to changes in cerebral blood flow
(16), was used as model parameter of dopamine transporter availability.
Statistics
Differences between comparison subjects and methamphetamine abusers in K1, distribution volume, and Bmax/Kd were tested with unpaired Student’s t tests (two-tailed). For the methamphetamine abusers, Pearson product-moment correlation analyses were performed between dopamine transporter measures and neuropsychological test scores, the years and doses of methamphetamine used, and days since last methamphetamine use. We hypothesized a priori an association between dopamine transporter levels and performance on the two motor and the three auditory verbal learning tasks, since abnormal results on these tests had been found in patients with Parkinson’s disease in proportion to dopamine damage. A significance threshold was set at p<0.05. For exploratory analyses of the correlation between dopamine transporter levels and performance on the other seven neuropsychological tests, we set the significance threshold at p<0.007.
Results
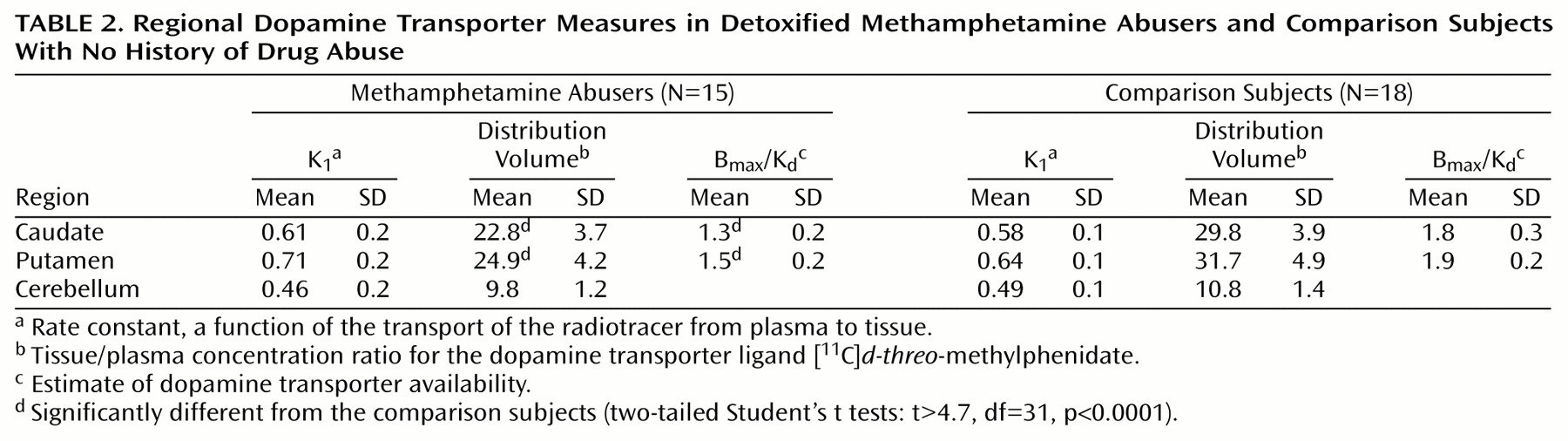
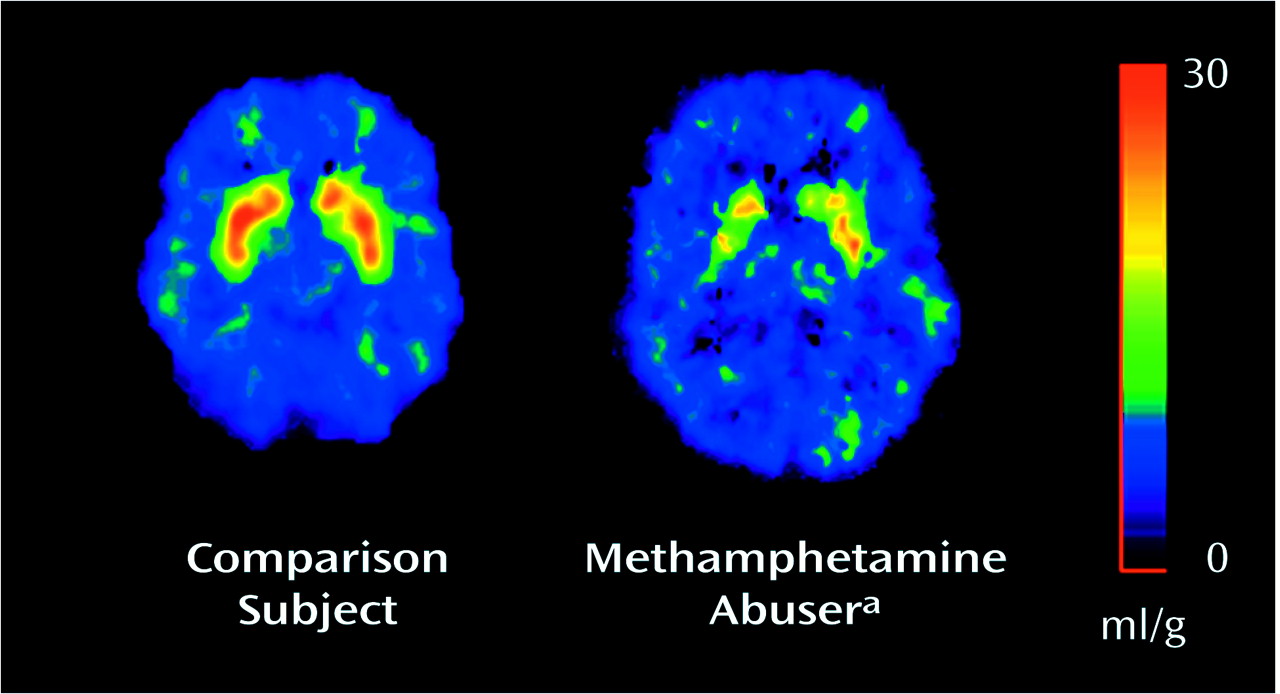
The K
1 measures did not differ between the comparison subjects and the methamphetamine abusers, whereas the distribution volumes in the caudate and putamen, but not in the cerebellum, were significantly lower in the abusers (
Figure 1,
Table 2). Dopamine transporter availability (B
max/K
d) was significantly lower in the methamphetamine abusers than in the comparison subjects both in the caudate (27.8% difference) and in the putamen (21.1% difference) (
Table 2).
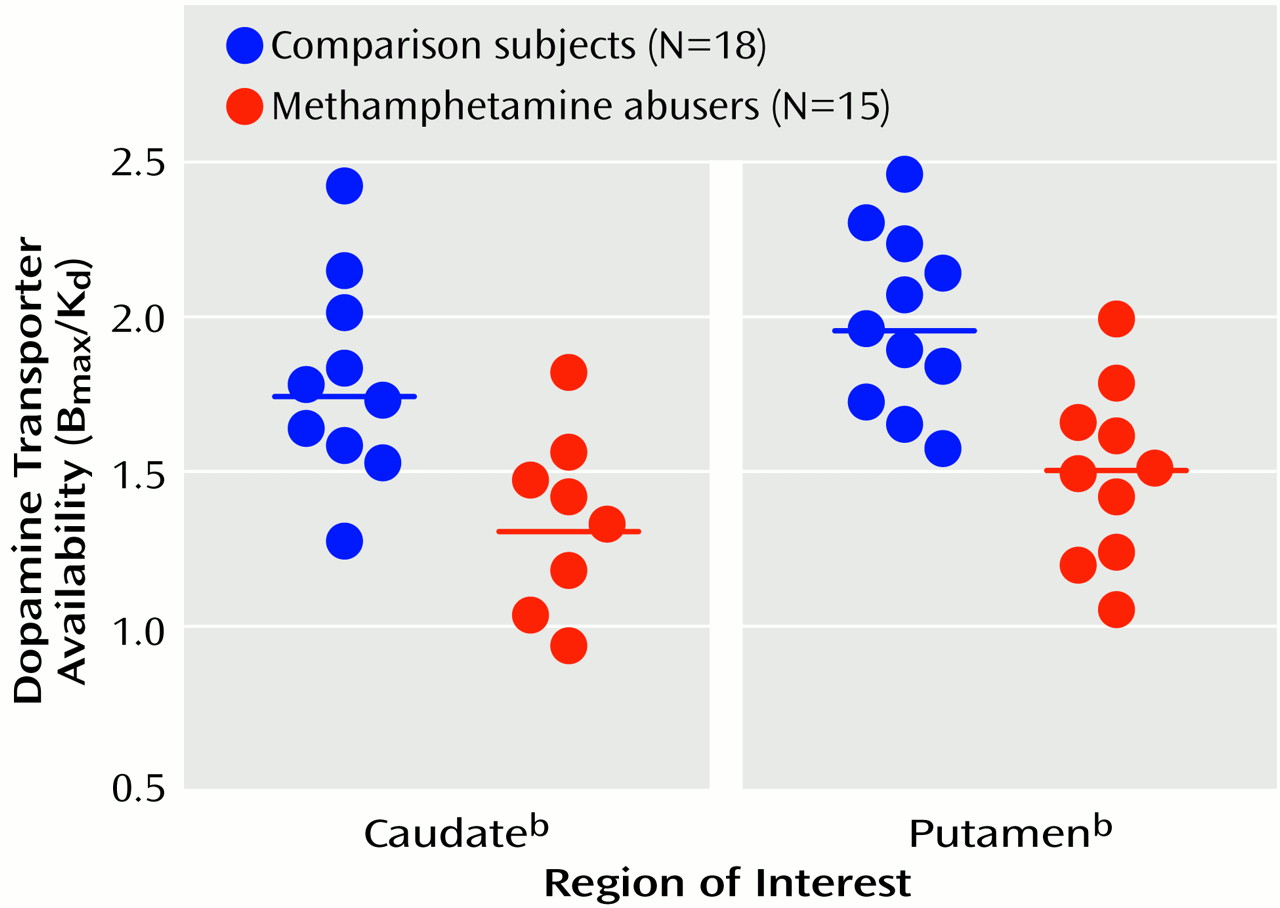
Figure 2 shows the dopamine transporter availability (B
max/K
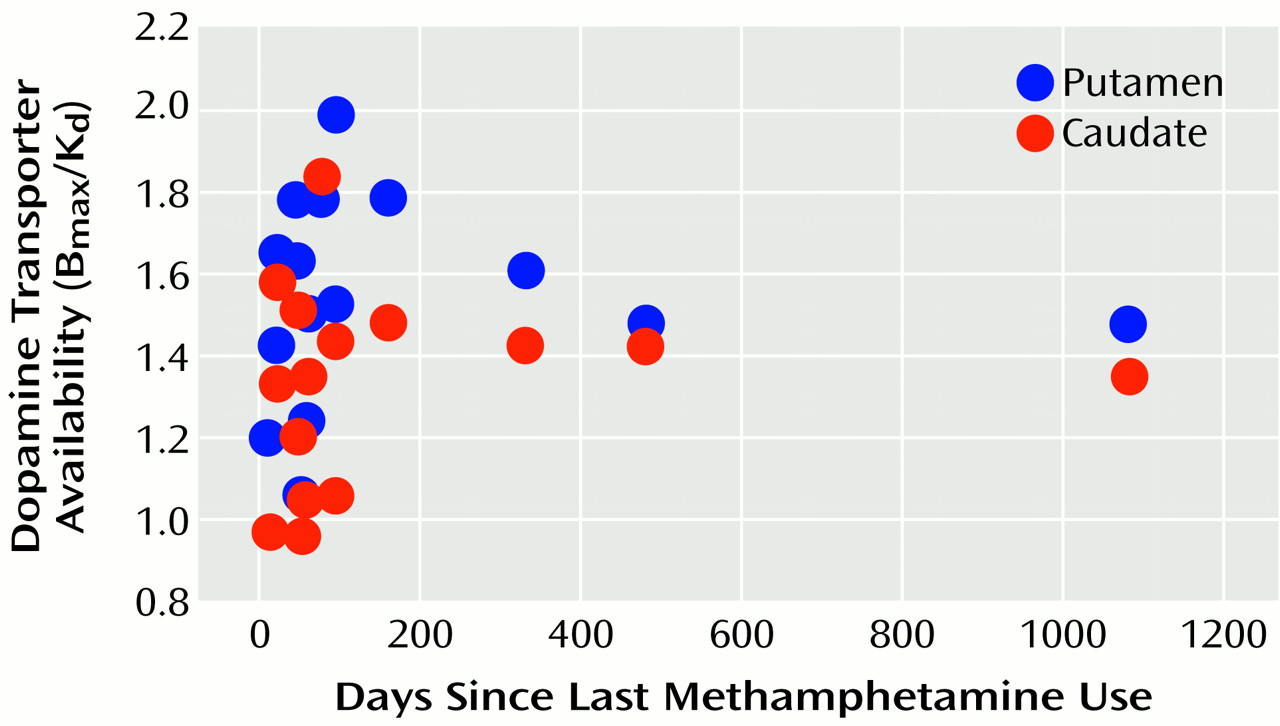
d) values for the individual comparison subjects and methamphetamine abusers. A significant correlation between dopamine transporter level and years of methamphetamine use was found in the caudate (r=0.54, df=14, p<0.05). This correlation approached significance in the putamen (r=0.47, df=14, p<0.08), but no correlation was seen in these areas between dopamine transporter levels and methamphetamine dose (r<0.37, df=14, p>0.17). Dopamine transporter levels also were not significantly correlated with days since last methamphetamine use (r<0.11, df=14, p>0.70) (
Figure 3).
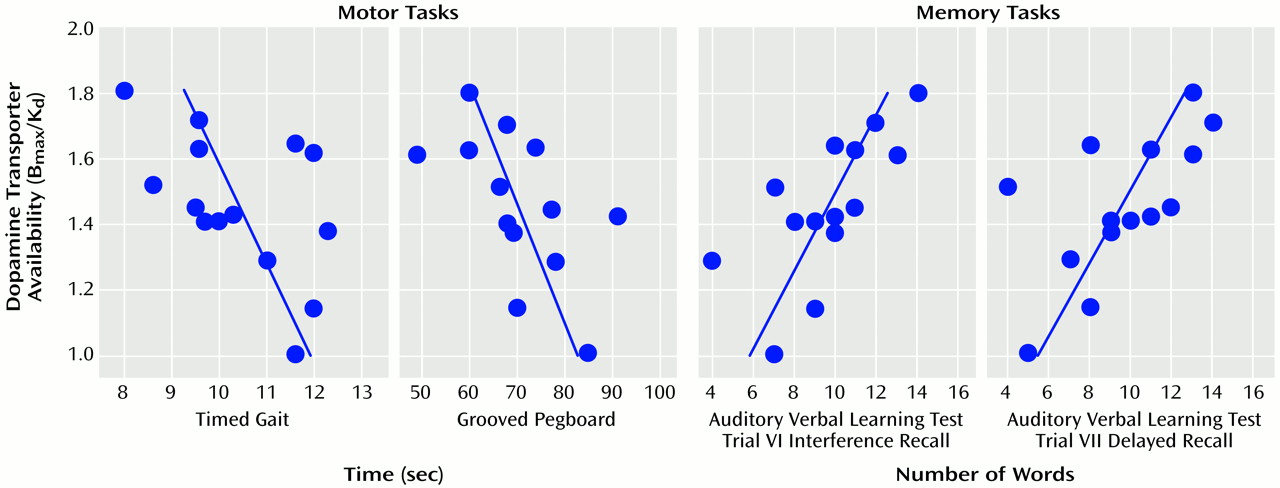
Because the estimates of dopamine transporters in the caudate were strongly correlated with those in the putamen (r=0.75, df=14, p<0.0001), we averaged these two measures into a striatal value. The correlations between striatal dopamine transporters and performance on the neuropsychological tests for which we hypothesized an association a priori were significant for the motor tasks (Timed Gait task: r=0.53, df=14, p<0.05; Grooved Pegboard task: r=0.57, df=14, p<0.05) and for the verbal memory task (Auditory Verbal Learning Test, interference recall: r=0.70, df=14, p<0.005; delayed recall: r=0.64, df=14, p<0.01; immediate recall: r=0.58, df=14, p<0.05) (
Figure 4). For the exploratory analyses, only the correlation with the Trail Making Test approached significance (r=0.61, df=14, p<0.05). None of the other tests were significantly correlated with striatal dopamine transporter level.
Discussion
This study documented significant dopamine transporter reduction in detoxified methamphetamine abusers relative to non-drug-abusing comparison subjects that was associated with poor motor and memory performance. Dopamine transporter reduction was observed as lower [
11C]
d-threo-methylphenidate distribution volume and lower striatal dopamine transporter availability in the methamphetamine abusers relative to the comparison subjects. The fact that there were no differences in K
1 for [
11C]
d-threo-methylphenidate between comparison subjects and methamphetamine abusers indicates that the reductions in distribution volume were not due to changes in tracer delivery. Dopamine transporter reduction was seen even in the three methamphetamine abusers who had been detoxified for at least 11 months. These findings are consistent with previous reports in human
(8,
9) and nonhuman primates
(17,
18) that have documented dopamine transporter changes after methamphetamine administration. The findings from these studies have implications in the treatment of methamphetamine abusers, for they suggest that interventions that improve dopamine brain function would benefit these individuals by improving motor and cognitive function.
The dopamine transporter reductions seen in the methamphetamine abusers were smaller than those found in patients with Parkinson’s disease, in whom dopamine transporter reductions are proportional to disease severity and range between 36% and 71%
(19–
21). However, it should be noted that in three methamphetamine abusers, the dopamine transporter level fell within the range seen in patients with low-severity Parkinson’s disease. It is likely that these three relatively young subjects did not have extrapyramidal symptoms because they were still able to compensate. Nonetheless, dopamine transporter reductions resulted in impaired motor performance on the Timed Gait and Grooved Pegboard tasks: the lower the dopamine transporter level, the slower the motor responses. A similar association, albeit for more severe pathology, has been reported for patients with Parkinson’s disease studied with PET and [
18F]fluoro-
l-DOPA
(22). Thus, while the dopamine transporter reductions in the methamphetamine abusers may not have been severe enough to induce parkinsonian symptoms, our findings suggest that they resulted in impairment of motor function. Since significant dopamine transporter reductions occur both with age (6%–7% per decade)
(23) and with methamphetamine abuse, it is possible that an interaction effect of methamphetamine abuse and aging may yield a higher risk for the development of parkinsonian symptoms in these abusers later in life.
The dopamine transporter reductions in the methamphetamine abusers also differed from those seen in patients with Parkinson’s disease in that the magnitude of the reduction was similar in the caudate and putamen, whereas in Parkinson’s disease, the putamen is more frequently affected than the caudate
(24–
26). This suggests different mechanisms for dopamine transporter reduction in methamphetamine abusers than in Parkinson’s disease. Also, since the caudate is more involved than the putamen in cognitive operations
(27), it is to be expected that in methamphetamine abusers dopamine transporter reduction will result in cognitive as well as motor impairment. In fact, in laboratory animals methamphetamine induces motor
(28) and learning and memory impairments
(29). Moreover, the degree of dopamine transporter reduction in the methamphetamine abusers predicted both the motor as well as the memory changes; the lower the dopamine transporter levels, the worse their performance. Performance on the task used to assess verbal memory in this study (Auditory Verbal Learning Test) has also been found to be impaired in Parkinson’s disease patients
(30), whose performance, as was seen with the methamphetamine abusers, was found to be associated with dopamine transporter levels
(31).
The dopamine transporter reductions in the methamphetamine abusers were smaller than those reported in animal studies, which have exceeded 50%
(6,
7). This could be due to differences in doses and patterns of use, interspecies differences, or coadministration of other drugs. Of particular relevance may be the fact that most methamphetamine abusers are cigarette smokers, since nicotine has been shown to be protective against methamphetamine neurotoxicity
(32). Thus, it is possible that one of the reasons why less dopamine transporter reduction was seen in the human methamphetamine abusers than has been reported in animal studies is that cigarette smoking may have provided some protection against methamphetamine-induced dopamine transporter reduction.
Studies in nonhuman primates have reported some recovery of dopamine terminal damage induced by methamphetamine abuse
(18). Although our study was not designed to assess recovery, the fact that the length of methamphetamine detoxification was not correlated with dopamine transporter levels suggests that in human methamphetamine abusers, dopamine transporter losses do not recover significantly following 1 year of detoxification.
The dopamine transporter reductions in the methamphetamine abusers could reflect either a decrease in dopamine transporter expression or degeneration of dopamine terminals. While there is evidence from preclinical studies that methamphetamine induces dopamine terminal degeneration
(33), a human postmortem study showed dopamine transporter reductions but not vesicular monoamine transporter reductions
(9). Since vesicular monoamine transporters are more stable markers of dopamine terminals than dopamine transporters, this was interpreted as reflecting persistence of the dopamine terminal
(8). Thus, further studies are required to determine if dopamine transporter reductions in humans are due to dopamine terminal degeneration. This study focused on the effects of methamphetamine on dopamine transporter levels, but animal studies have shown that methamphetamine also damages other neuronal types
(6,
7). Thus, further studies are required to assess if methamphetamine disrupts neuronal systems other than dopamine in human subjects.
In summary, the results from this study provide evidence that methamphetamine at doses abused by humans leads to dopamine transporter reductions in the brain and that this reduction is associated with functional impairment. The fact that the dopamine transporter levels were lower even in subjects detoxified for at least 11 months suggests that methamphetamine’s effects in the human brain may be long lasting. At present we do not know whether the dopamine transporter reductions reflect dopamine terminal damage or down-regulation of dopamine transporter expression; we also do not know whether this reduction may eventually resolve or whether it may increase vulnerability to Parkinson’s disease or other neurodegenerative diseases. Thus, there is an urgent need to alert methamphetamine users to the consequences of their abuse and to develop treatments for these patients. Similarly, preventive measures are needed urgently to warn and educate the public of the damaging effects of methamphetamine to the human brain.