Methamphetamine is a highly addictive drug
(1), and methamphetamine abuse has risen substantially in several areas of the United States
(2,
3) and the world
(4). As methamphetamine abuse rises, concern about its potential neurotoxic effects on human abusers increases, since methamphetamine administration has been shown to produce long-lasting damage to select cell populations in the brains of laboratory animals. Specific damage has been documented in dopamine cells
(5,
6). Methamphetamine has consistently been reported to cause persistent decreases in markers of dopamine cell function and structure
(7). Moreover, these changes have been associated with long-lasting motor deficits, which indicate that the neurotoxic effects are functionally significant
(8). There is also evidence that methamphetamine causes decreases in markers of serotonin cell function
(9) and that it also affects nonmonoaminergic cortical neurons
(10,
11). Data from human studies are very limited, and both postmortem as well as imaging studies have mainly focused on the effects of methamphetamine on dopamine cells. These studies have shown a marked reduction in dopamine transporters, which serve as markers for dopamine cell terminals in methamphetamine abusers
(12–
14). However, there are no data on the effects of methamphetamine in nondopamine neurons in humans. The purpose of this study was to assess whether there are changes in brain regions other than those innervated by dopamine.
Following administration of [
18F]fluorodeoxyglucose (FDG), positron emission tomography (PET) was used to measure regional brain glucose metabolism. Cerebral glucose metabolism serves as a marker of neuronal activity
(15) and is a sensitive indicator of brain dysfunction
(16,
17). The methamphetamine abusers and 18 of the comparison subjects also underwent PET imaging to measure dopamine transporter levels in the brain. The results from these measures, which showed marked striatal dopamine transporter reductions in methamphetamine abusers, are reported in a separate article in this issue of the
Journal (18).
Results
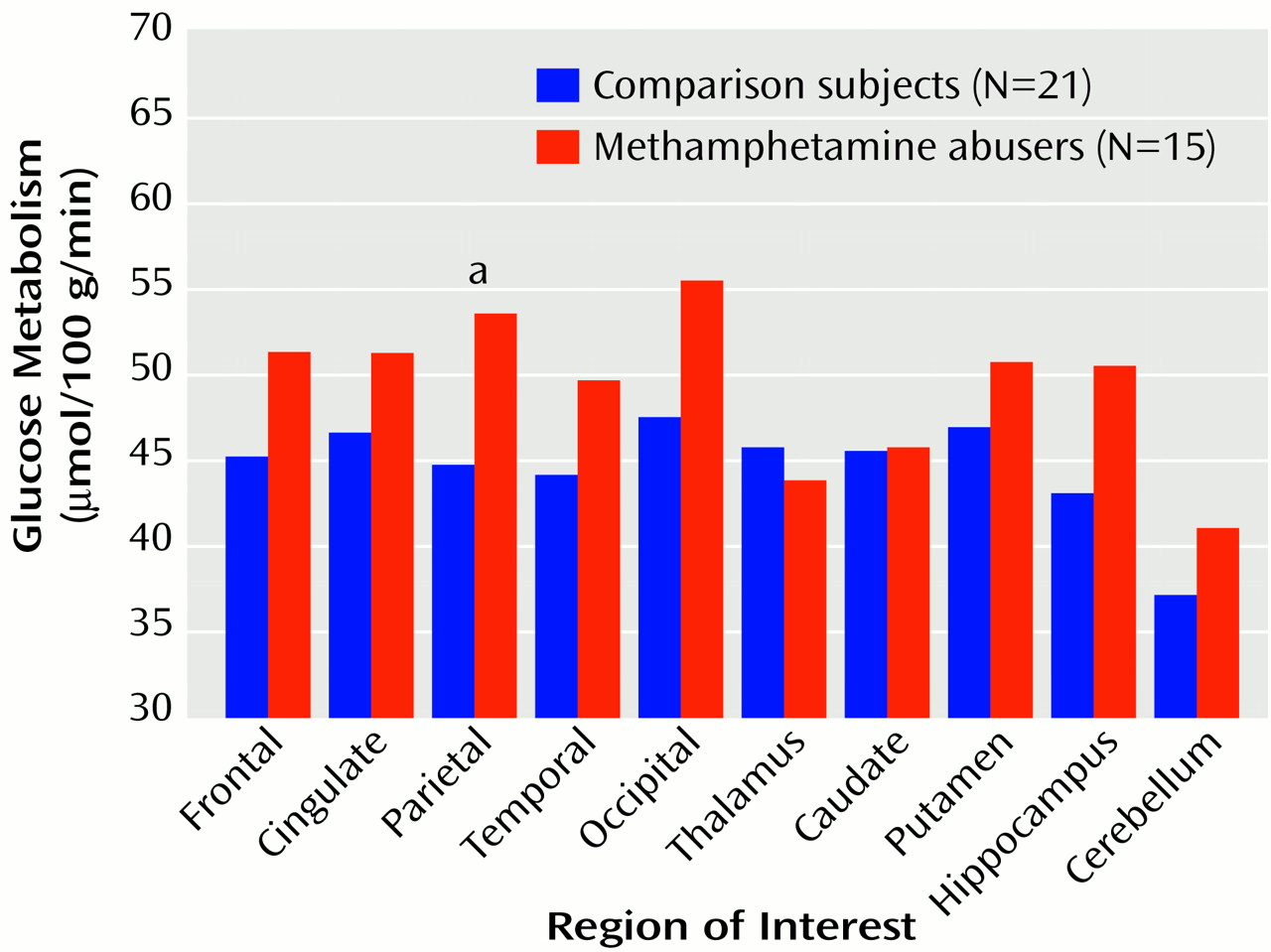
Global metabolic rate was significantly higher (14%) for the methamphetamine abusers (mean=38.4 μmol/100 g/minute, SD=8) than for the comparison subjects (33.6 μmol/100 g/minute, SD=5) (t=2.2, df=34, p<0.05) (
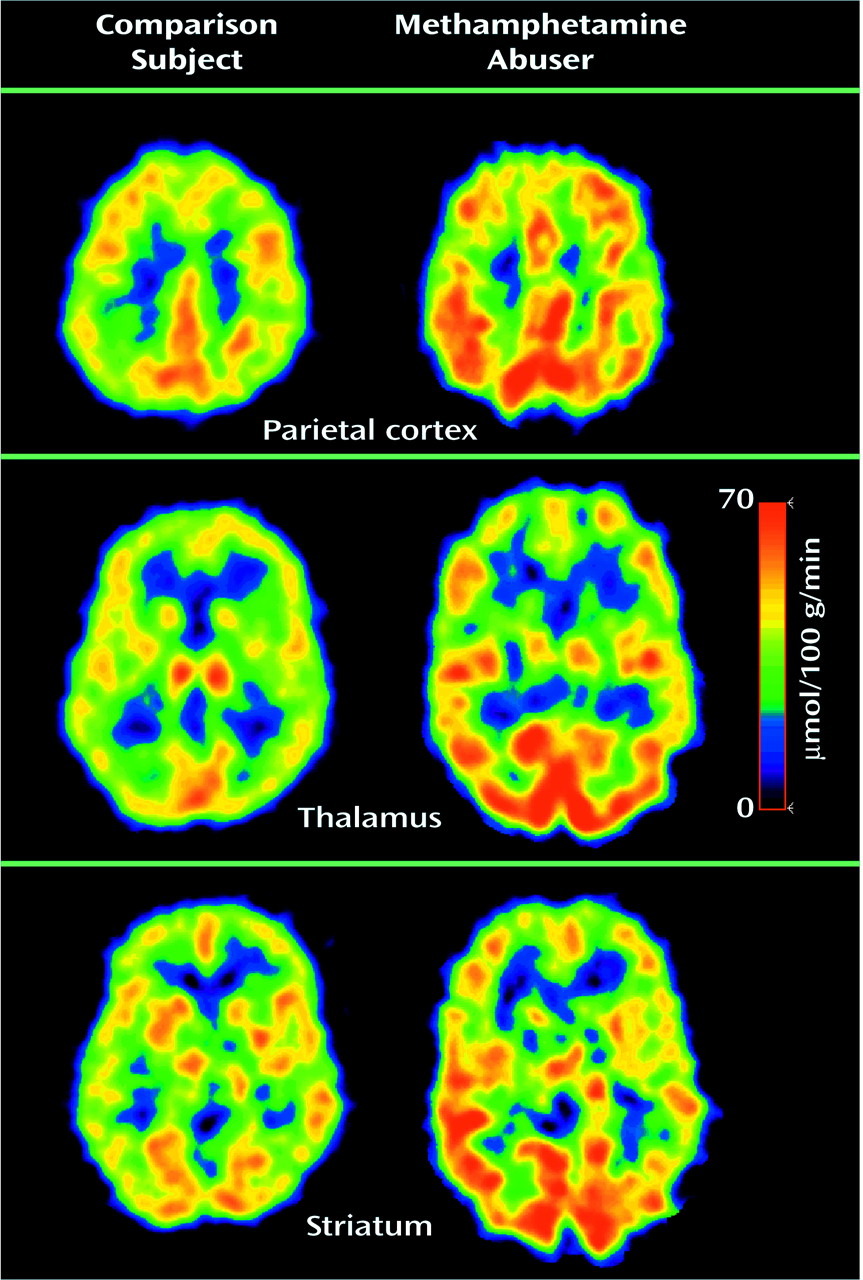
Figure 1). Analysis of the regional measures showed that relative to the comparison subjects, metabolism in the parietal cortex was 20% higher in the methamphetamine abusers, which was a significant difference (
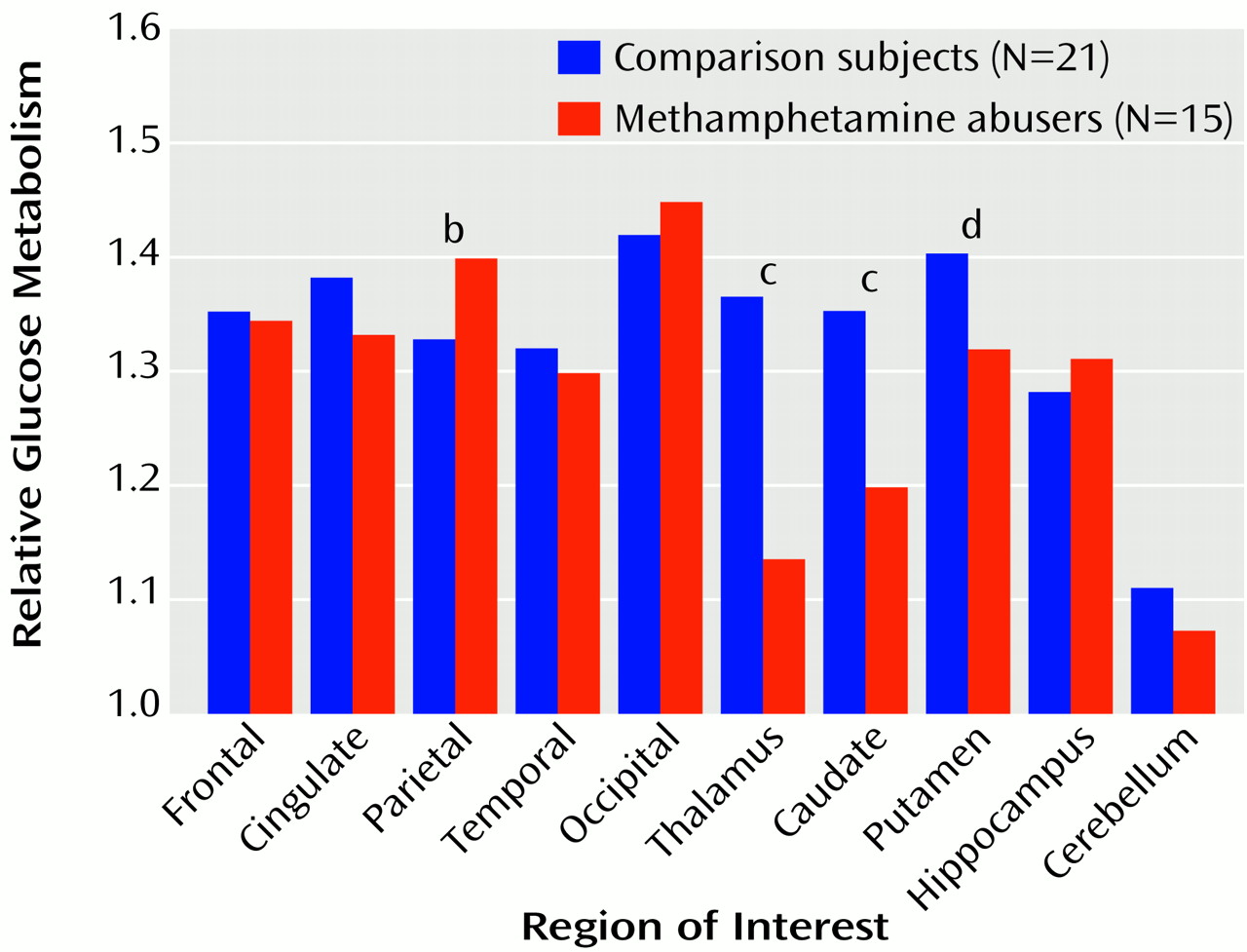
Figure 2). While not achieving significance, higher metabolism (differences of 11%–12%) was seen in the frontal cortex (t=2.1, df=34, p<0.05), temporal cortex (t=2.0, df=34, p<0.05), occipital cortex (t=2.4, df=34, p<0.05), and hippocampus (t=2.2, df=34, p<0.05) of the methamphetamine abusers. After normalizing for whole brain metabolism (relative metabolism), the only brain region in which the methamphetamine abusers showed significantly higher metabolism was the parietal cortex, where metabolism was 5% higher (
Figure 3). However, the analysis of the relative measures also revealed that the methamphetamine abusers had significantly lower metabolism in the thalamus, caudate, and putamen (differences of 17%, 12%, and 6%, respectively). To determine if metabolic changes in the caudate were significantly larger than those in the putamen, we conducted a factorial (comparison subjects versus methamphetamine abusers) repeated measure (caudate versus putamen) ANOVA. This showed a significant diagnosis-by-region interaction effect (F=4.8, df=1, 34, p<0.05), indicating that the differences between comparison subjects and the methamphetamine abusers were greater in the caudate than in the putamen.
To assess if there was any evidence of recovery with detoxification, we conducted a separate analysis that included only the three subjects who had been detoxified for 11 months or longer; similar results were found as those obtained for the whole group. These long-detoxified methamphetamine abusers had significantly lower relative metabolism in the caudate than comparison subjects (12% difference; t=3.6, df=22, p<0.002) and, although not significant, also had global metabolism that was 13% higher and relative metabolism in the thalamus that was 10% lower. We also measured the correlations between the metabolic measures that differed between the groups and days since last methamphetamine use; none were significant.
There were no significant correlations between whole brain metabolism and neuropsychological scores, but relative metabolism in the parietal cortex was significantly correlated with performance on the Grooved Pegboard task (r=0.73, df=14, p<0.003); there were no significant correlations between neuropsychological scores and thalamic or striatal metabolic measures. Whole brain metabolism was negatively correlated with years of methamphetamine use (r=–0.62, df=14, p<0.02), but this effect was not significant after covarying for age. Correlations between metabolic measures and methamphetamine dose were not significant.
To determine if gender differences between groups (the methamphetamine group had a larger proportion of women [60%] than did the comparison group [28%]) affected the results, we conducted separate comparisons for each gender. The findings were consistent with those when both genders were included. The women (nine methamphetamine abusers and six comparison subjects) had higher whole brain metabolism (t=2.2, df=13, p<0.05) and lower relative metabolism in the thalamus (t=3.8, df=13, p<0.002) and caudate (t=3.0, df=13, p<0.01); the men (six methamphetamine abusers and 15 comparison subjects) had lower relative metabolism in the thalamus (t=3.6, df=19, p<0.002), caudate (t=4.4, df=19, p<0.001), and putamen (t=2.2, df=19, p<0.05) (data not shown).
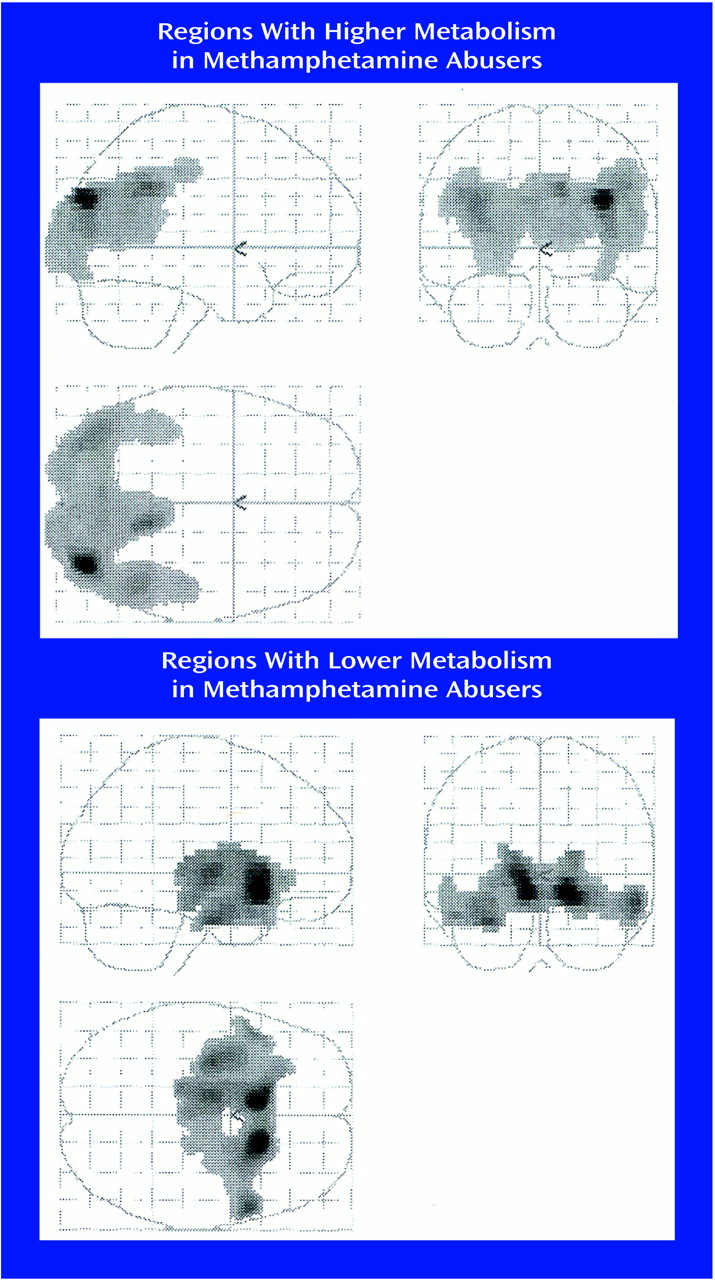
Statistical parametric mapping analyses revealed significant differences between the comparison subjects and the methamphetamine abusers that corroborated the findings from the region of interest analysis. Methamphetamine abusers had significantly higher metabolism than comparison subjects in the parietal cortex (Brodmann’s areas 39 and 40) and significantly lower metabolism in the thalamus, caudate, and putamen (
Figure 4).
Discussion
This study documented significantly higher whole brain metabolism that was most marked in the parietal cortex and significantly lower relative metabolism in the thalamus and striatum of detoxified methamphetamine abusers. These metabolic differences were present even in the three methamphetamine abusers who had been detoxified for at least 11 months. These findings are similar to those that have been reported in laboratory animals, which showed persistent reductions in subcortical metabolism after repeated administration of methamphetamine to rats
(26). The striatum receives direct projections from dopamine mesencephalic cells, which in animals have been shown to be sensitive to methamphetamine’s neurotoxic effects
(5–
7); hence, the reduction in brain metabolism could reflect methamphetamine-induced disruption to these cells. The thalamus, on the other hand, mainly receives dopamine signals from the striatum
(27). Thus, one could also postulate that the lower metabolism in the thalamus of the methamphetamine abusers reflects disruption in dopamine-linked pathways. Thus, these findings provide evidence that methamphetamine at the doses abused by humans causes long-lasting changes in brain metabolism in both regions that are directly or indirectly connected to dopamine pathways (striatum and thalamus) as well as regions that are not (parietal cortex).
The finding of brain hypermetabolism in the methamphetamine abusers was unexpected, since most studies have revealed decrements in brain metabolism in drug abusers (detoxified alcoholics or cocaine abusers)
(28). A review of the literature showed that brain disease states for which higher metabolism has been reported, such as head trauma
(29) and radiation damage
(30,
31), usually involve inflammation and gliosis
(32). Studies in laboratory animals have documented that methamphetamine induces gliosis
(33,
34), and although such an effect has not been directly documented in the human brain, studies with magnetic resonance spectroscopy have shown increases in the concentration of
myo-inositol, which serves as a glial marker, in methamphetamine abusers
(35). Thus, one could speculate that the hypermetabolism in the methamphetamine abusers could reflect gliosis/inflammation, and studies to evaluate this possibility merit further investigation.
The hypermetabolic activity in the brain of the methamphetamine abusers was most prominent in the parietal cortex (association cortex), a brain region that is poorly innervated by dopamine terminals. Hence, the hypermetabolism most likely reflects the effects of methamphetamine on neuronal types or circuits other than dopamine. In laboratory animals, the parietal cortex was found to be particularly sensitive to neurotoxicity from methamphetamine
(36). In the parietal cortex, methamphetamine produces degeneration of myelinated processes, damages pyramidal glutamatergic cells
(10,
11), and damages serotonergic terminals
(37). Methamphetamine also increases glutamate receptors in the parietal cortex
(38), which would make it more sensitive to glutamate excitotoxicity. Unfortunately, none of the neuropsychological tests used to evaluate the methamphetamine abusers specifically targeted functions of the parietal cortex. Nonetheless, we found that in the methamphetamine abusers metabolism in the parietal cortex was correlated with performance on the Grooved Pegboard task. Although the Grooved Pegboard is considered a motor task
(22), there is a spatial component to it: the subject has to make a decision about where to insert the pegs and then orient the grooves in a certain way in order for them to match and be inserted into the holes. Although preliminary in its interpretation, this significant association suggests that hyperactivity of the parietal cortex may be functionally significant.
Methamphetamine abusers also showed significantly lower relative metabolism in the thalamus and striatum. This pattern is reminiscent of the metabolic abnormalities reported in patients with atypical Parkinson’s disease (who also show relatively lower thalamic and striatal metabolism
[39]) but differs from abnormalities reported in patients with Parkinson’s disease (who show either no metabolic changes
[40] or higher striatal and thalamic metabolism
[41]). The findings of dopamine transporter reductions in methamphetamine abusers
(12–
14) also more closely resemble the findings from patients with atypical Parkinson’s disease than patients with idiopathic Parkinson’s in that the former, like the methamphetamine abusers, exhibit equivalent dopamine transporter reductions in the caudate and putamen
(42), whereas the patients with Parkinson’s disease have greater dopamine transporter reductions in the putamen than in the caudate
(43,
44). This is relevant because patients with atypical Parkinson’s disease in addition to dopamine cell damage have degeneration of pallidal efferents, whereas patients with idiopathic Parkinson’s have a predominant involvement of dopamine cells. Moreover, in patients with atypical Parkinson’s disease, the decrements in subcortical metabolism are believed to reflect degeneration of striatal efferents
(45). Thus, one could postulate that the abnormalities in the striatum and thalamus in the methamphetamine abusers reflect not only methamphetamine’s effects in dopamine terminals
(12–
14) but also its effects on striatal efferents. In fact, animal studies have shown that methamphetamine, in addition to damaging dopamine terminals, also induces degeneration of striatal efferents
(46). Thus, the results from this study coupled with the findings of dopamine transporter reduction in methamphetamine abusers
(12–
14) should alert clinicians of the possibility of a higher risk for neurodegenerative diseases in these subjects.
The pattern of regional brain metabolic abnormalities seen in these methamphetamine abusers differs from that reported after acute methamphetamine administration in humans, which showed decreases in cortical metabolism and increases in cerebellar metabolism
(47). This indicates that the regions that may be most sensitive to acute administration are not necessarily the same as those that are vulnerable to the long-term effects of methamphetamine.
In generalizing the findings from this study it is relevant to address its limitations. This study was done with outpatients, and thus it is not possible to determine with certainty that the period since last methamphetamine use was accurate. Also, in this type of clinical study there are inaccuracies regarding exact amount and histories of drug use by the substance abusers as well as denial of drug use by the comparison subjects. Although we performed a careful physical examination and obtained routine laboratory tests, we did not test for all potential confounding diseases (e.g., HIV in the comparison subjects). Neurocognitive tests were only performed by the methamphetamine abusers, and thus it is not possible to determine the extent to which the groups differed. There are also confounds from differences in the groups investigated; the percentage of smokers in the methamphetamine group (73%) was higher than in the comparison group (24%), and although we excluded subjects if they were addicted to drugs apart from nicotine (or methamphetamine for the methamphetamine abusers), it is likely that the methamphetamine abusers consumed larger amounts of other drugs (i.e., alcohol, marijuana) than the comparison group.
In summary, the results from this study provide evidence that methamphetamine at doses abused by humans induces long-lasting metabolic changes in brain regions neuroanatomically connected with dopamine pathways but also in areas that are not innervated by dopamine. Prospective studies coupled with neuropsychological assessment of methamphetamine abusers are required to determine if these changes recover and if they predispose to neurodegenerative diseases.