Results
The study group consisted of 57 patients: 26 (45.6%) with schizophrenia, 18 (31.6%) with brief psychotic disorder, eight (14.0%) with substance-induced psychotic disorder, and five (8.8%) with schizoaffective disorder. The distribution of the polymorphism in these patients was in accordance with that in literature
(5). Forty-one patients (71.9%) were homozygous for allele 2, and 16 (28.1%) were heterozygous.
Genetic groups did not differ with respect to the distribution of diagnoses at baseline and on day 7 (χ2=1.03, df=3, p=0.80), on day 14 (χ2=1.12, df=3, p=0.78), on day 21 (χ2=2.39, df=3, p=0.50), or on day 28 (χ2=3.41, df=2, p=0.19). They also did not differ in age (t=0.55, df=55, p=0.59), sex (χ2=1.66, df=1, p=0.24), age at onset of illness (t=1.48, df=55, p=0.15), number of hospitalizations (t=–0.35, df=55, p=0.74), or duration of hospitalizations (t=–0.48, df=51, p=0.65). There was no difference between genetic groups in duration of episode before treatment, categorized by less than 1 month, less than 3 months, and 3 or more months (χ2=3.11, df=2, p=0.22). Haloperidol doses were comparable on day 7 (t=0.37, df=55, p=0.72), day 14 (t=–0.82, df=53, p=0.42), day 21 (t=–1.05, df=38, p=0.31), and day 28 (t=–0.49, df=20, p=0.63); mean daily doses were comparable throughout the study (t=–0.96, df=172, p=0.34).
At baseline, scores on the positive (t=–0.50, df=55, p=0.63) and negative (t=1.10, df=55, p=0.28) subscales of the Positive and Negative Syndrome Scale were not different between the two genotype groups. The diagnostic groups also did not differ in positive and negative values, respectively, at baseline: schizophrenia (t=0.39, df=24, p=0.70, and t=1.46, df=24, p=0.16), schizoaffective disorder (t=1.53, df=3, p=0.23, and t=0.76, df=3, p=0.51), brief psychotic disorder (t=–0.45, df=16, p=0.67, and t=0.02, df=16, p=0.99), and substance-induced psychotic disorder (t=–2.08, df=6, p=0.09, and t=–0.24, df=6, p=0.82).
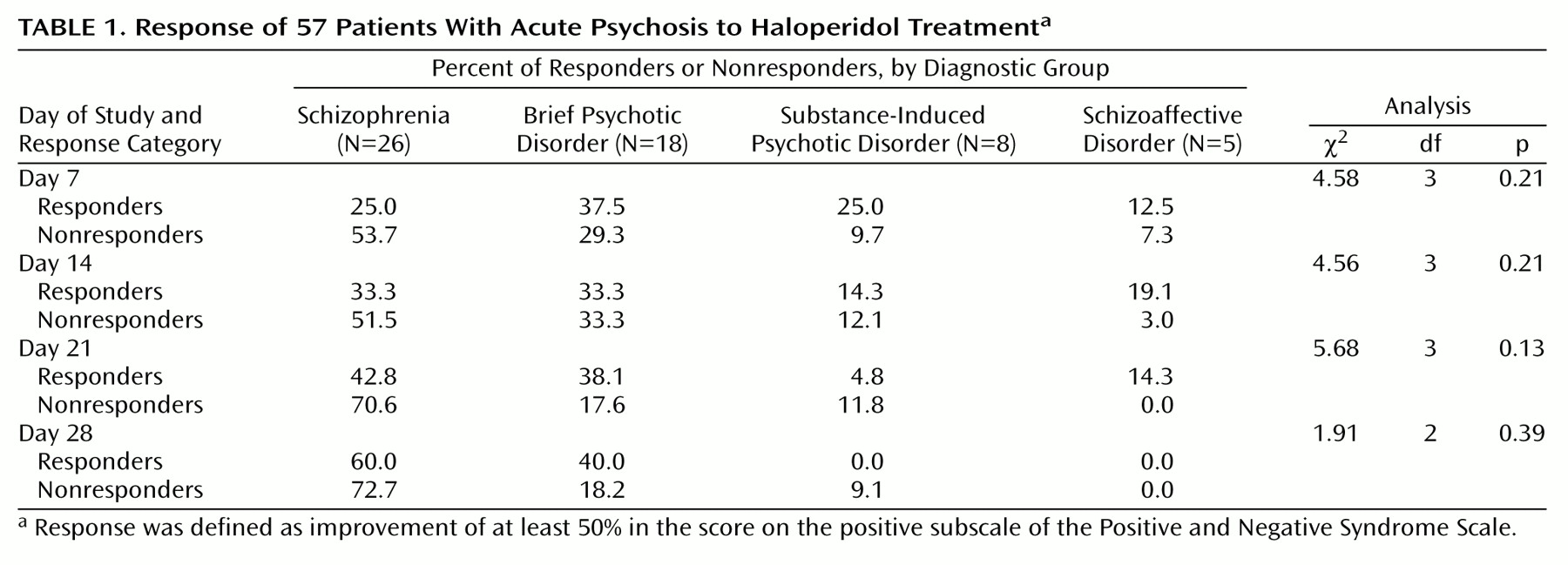
The percentages of patients in each diagnostic group who were defined as responders (their score on the positive subscale of the Positive and Negative Syndrome Scale improved by at least 50%) did not differ (
Table 1).
Haloperidol dose was not correlated with response on day 7 (responders: r=0.04, N=16, p=0.89; nonresponders: r=–0.21, N=41, p=0.18) and day 14 (r=–0.29, N=21, p=0.21; nonresponders: r=–0.14, N=32, p=0.45) but was negatively correlated for nonresponders on day 21 (responders: r=–0.15, N=21, p=0.53; nonresponders: r=–0.66, N=16, p=0.005) and day 28 (responders: r=–0.58, N=10, p=0.09; nonresponders: r=–0.85, N=10, p=0.002). Furthermore, doses did not differ between responders and nonresponders on day 7 (t=0.77, df=55, p=0.45), day 14 (t=1.03, df=51, p=0.31), day 21 (t=0.64, df=35, p=0.53), or day 28 (t=1.49, df=18, p=0.16). The dropout rate was not related to the last available haloperidol dose (F=1.38, df=2, 33, p=0.27). Reasons for dropout before day 28 were early remission (12 [29.3%] of 41 homozygous patients and six [37.5%] of 16 heterozygous patients), severe side effects (six [14.6%] homozygous patients and four [25.0%] heterozygous patients), or nonresponse (seven [17.1%] homozygous patients and one [6.3%] heterozygous patient).
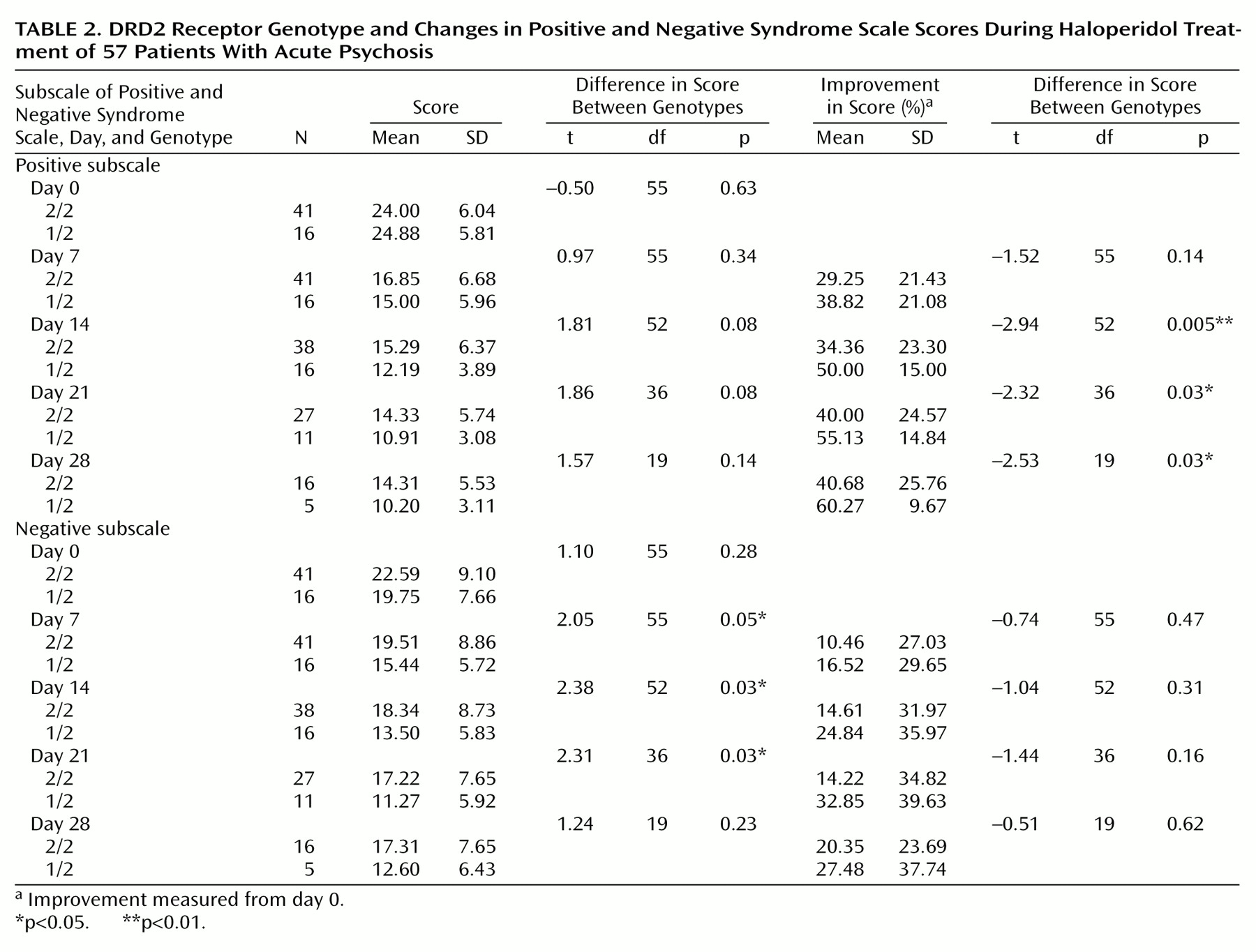
Overall, we observed a better response to haloperidol treatment in the heterozygous group. The improvement in positive symptoms in this group was greater than the improvement in the homozygous group on all assessment days and attained statistical significance on days 14, 21, and 28 (
Table 2). This effect was also seen after applying the general linear model with repeated measurements (F=4.37, df=1, 55, p=0.05). There were no differences between heterozygous and homozygous groups in negative symptoms (
Table 2) (F=1.28, df=1, 55, p=0.27).
On treatment day 14, 10 (62.5%) of 16 heterozygous patients were responders, compared with 11 [28.9%] of 38 homozygous patients (χ2=5.33, df=1, p=0.04, odds ratio=4.09, 95% confidence interval [CI]=1.19–14.01, sensitivity=0.48, specificity=0.82). On day 21, nine (81.8%) of 11 heterozygous patients were responders, compared with 12 (44.4%) of 27 homozygous patients (χ2=4.42, df=1, p=0.07, odds ratio=5.63, 95% CI=1.02–31.10, sensitivity=0.43, specificity=0.88). On day 28, four (80.0%) of five heterozygous patients were responders, compared with six (37.5%) of 16 homozygous patients (χ2=2.76, df=1, p=0.15, odds ratio=6.67, 95% CI=0.60–74.51, sensitivity=0.40, specificity=0.91).