Patient Group and Recruitment Procedures
The study was carried out with patients who were regularly attending the outpatient units, day-care programs, and rehabilitation centers of the University Psychiatric Departments of Naples, Milan, L’Aquila, and Pisa, Italy. Those who had a clinical diagnosis of schizophrenia and were reported to be clinically stable were tested to verify that they met the following inclusion criteria: 1) a DSM-IV diagnosis of schizophrenia, confirmed by the Structured Clinical Interview for DSM-IV (SCID); 2) age between 16 and 55 years; 3) no history of severe mental retardation, alcoholism, or drug abuse or dependence in the last 12 months and no previous ECT; 4) no significant changes in the clinical state or in drug treatment during the preceding 3 months; and 5) willingness to participate in the study procedures, expressed by providing written informed consent after complete description of the study. Patients meeting these criteria were then classified as having either deficit or nondeficit schizophrenia after being interviewed with the Schedule for the Deficit Syndrome
(22).
The original version of the Schedule for the Deficit Syndrome was translated into Italian, back-translated into English by a bilingual translator, and discussed with one of the authors of the schedule (Brian Kirkpatrick), who also guided the training of the investigators. At the end of the training, there was no disagreement among eight raters (Cohen’s kappa=1) in classifying one patient with nondeficit schizophrenia and two patients with deficit schizophrenia. Intraclass correlation coefficients (ICCs) showed good agreement among raters for the Schedule for the Deficit Syndrome measures of restricted affect (0.76), diminished emotional range (0.62), poverty of speech (0.67), curbing of interests (0.74), diminished sense of purpose (0.74), and diminished social drive (0.81). Investigators were also trained in the use of the SCID and the SCID Nonpatient Version. At the end of the training, there was no disagreement among eight raters (Cohen’s kappa=1) in classifying two patients with schizophrenia and one as having bipolar I disorder with mood-incongruent psychotic features. The same kappa value was achieved by six raters on the nonpatient SCID.
All subjects with deficit schizophrenia according to the Schedule for the Deficit Syndrome were enrolled in the study. For each recruited patient with deficit schizophrenia, an age- and sex-matched patient with nondeficit schizophrenia was recruited. Thus, 60 deficit/nondeficit pairs were recruited. However, only those who completed most of the study procedures were included in the data analysis. The final group consisted of 58 patients with deficit schizophrenia (43 men and 15 women) and 54 patients with nondeficit schizophrenia (41 men and 13 women). The comparability between the two groups with respect to demographic and clinical variables was assessed by analyses of variance (ANOVAs) or chi-square tests, as appropriate. There were no significant differences between the deficit and nondeficit patients in terms of age (mean=35.2 years [SD=7.3] and 34.4 years [SD=7.7], respectively), education (mean=11.4 years [SD=3.1] and 11.3 years [SD=3.3]), age at onset of illness (mean=21.6 years [SD=4.5] and 21.7 years [SD=4.2]), duration of illness (mean=13.6 years [SD=7.4] and 12.8 years [SD=6.9]), or gender distribution. The mean current antipsychotic dose was significantly higher in patients with nondeficit schizophrenia (mean=645 mg/day [SD=410] in chlorpromazine equivalents) than in patients with deficit schizophrenia (mean=445 mg/day [SD=363]) (F=7.23, df=1, 106, p<0.008). There was no difference between the deficit and nondeficit patient groups as to the type of antipsychotic treatment they were receiving (novel antipsychotics: 56.9% [N=33] and 46.3% [N=25], respectively; standard neuroleptics: 25.9% [N=15] and 27.8% [N=15]; combination: 13.8% [N=8] and 24.1% [N=13]).
In two of the participating centers (Naples and Milan), a group of 26 healthy subjects (18 men and eight women)—matched to patients for age (within 3 years), education (within 3 years), handedness, and sex—was recruited through flyers from the general population for comparisons on neuropsychological indices. These comparison subjects had no personal or family history of major psychiatric disorders—as ascertained by the nonpatient SCID and the Family History Questionnaire and Relative Psychiatric History Questionnaires
(23)—and no history of severe head trauma or substance-related disorders. Their mean age of 34.6 years (SD=9.4) and their mean education level of 12.7 years (SD=2.9) did not significantly differ from those of patients.
Study Procedures
Different investigators carried out historical, psychopathological, neurological, and neuropsychological evaluations to avoid “halo” effects. All of them, as well as patients and family members, were blind to the deficit/nondeficit categorization and had not been informed of the specific study aims and hypotheses.
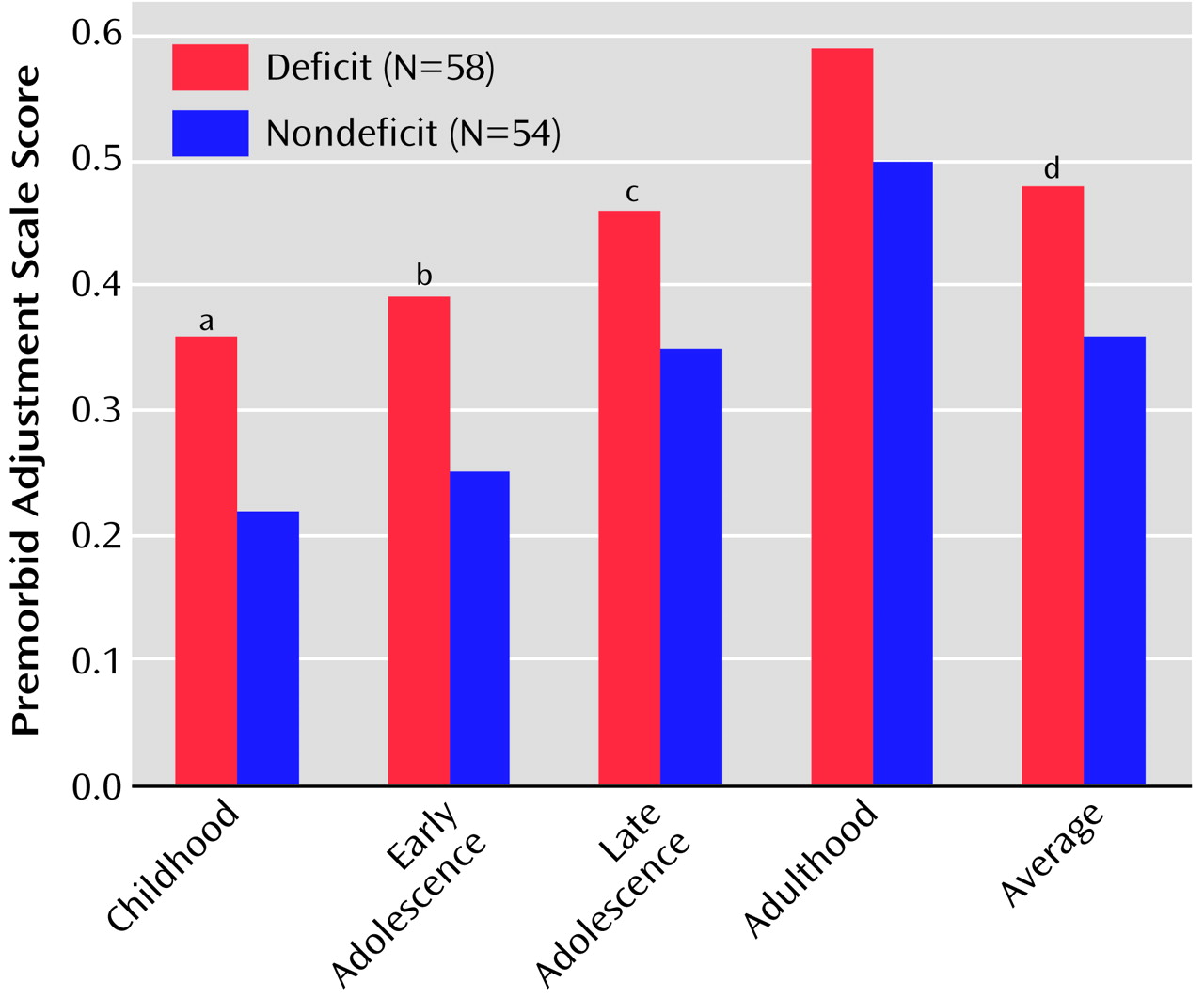
The personal and family history of each recruited patient was explored with the help of at least one family member (the mother, if available) by using the Family History Questionnaire and Relative Psychiatric History Questionnaires, the Obstetric Complications Scale
(24), and the Premorbid Adjustment Scale
(25). The psychopathological state was evaluated by the expanded Brief Psychiatric Rating Scale (BPRS), version 4.0
(26), the Scale for the Assessment of Negative Symptoms (SANS)
(27), and the Scale for the Assessment of Positive Symptoms (SAPS)
(28). The recent history of hospitalization and social functioning was explored by the Strauss-Carpenter Scale
(29). Investigators were trained in the use of these instruments, and at the end of the training, good to excellent agreement was observed among 12 raters who assessed three patients (for the expanded BPRS, ICC=0.61–1.00; for the SANS, ICC= 0.83–0.99 [except for the alogia and attention subscales, which had coefficients of 0.59 and 0.57, respectively]; for the SAPS, ICC=0.63–1.00).
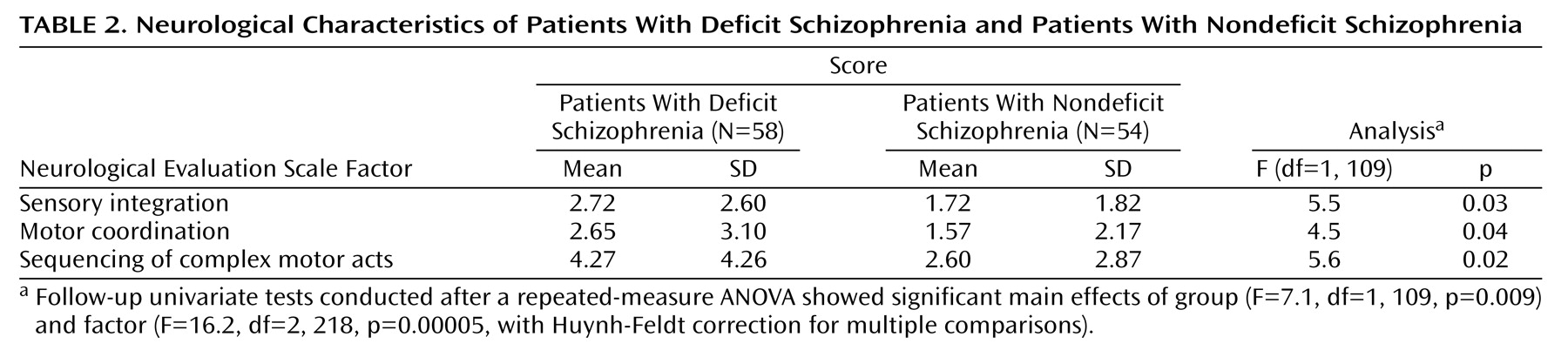
The Neurological Evaluation Scale
(30), which assesses three functional areas of interest (sensory integration, motor coordination, and sequencing of complex motor acts) plus some miscellaneous items (abnormalities in eye movements, frontal release signs, and short-term memory), was used for the neurological evaluation. The presence of spontaneous or induced abnormal involuntary movements was assessed by the Abnormal Involuntary Movement Scale (AIMS)
(31). Extrapyramidal signs were evaluated by the Simpson-Angus Scale
(32). Investigators were trained in the use of these instruments, and at the end of the training, good to excellent agreement was observed among five raters who assessed three patients (for the Neurological Evaluation Scale indices, ICC=0.75–0.99; for the AIMS, ICC=0.95 [total score] and 0.97 [perioral region subtotal]; for the Simpson-Angus Scale average score, ICC=0.54).
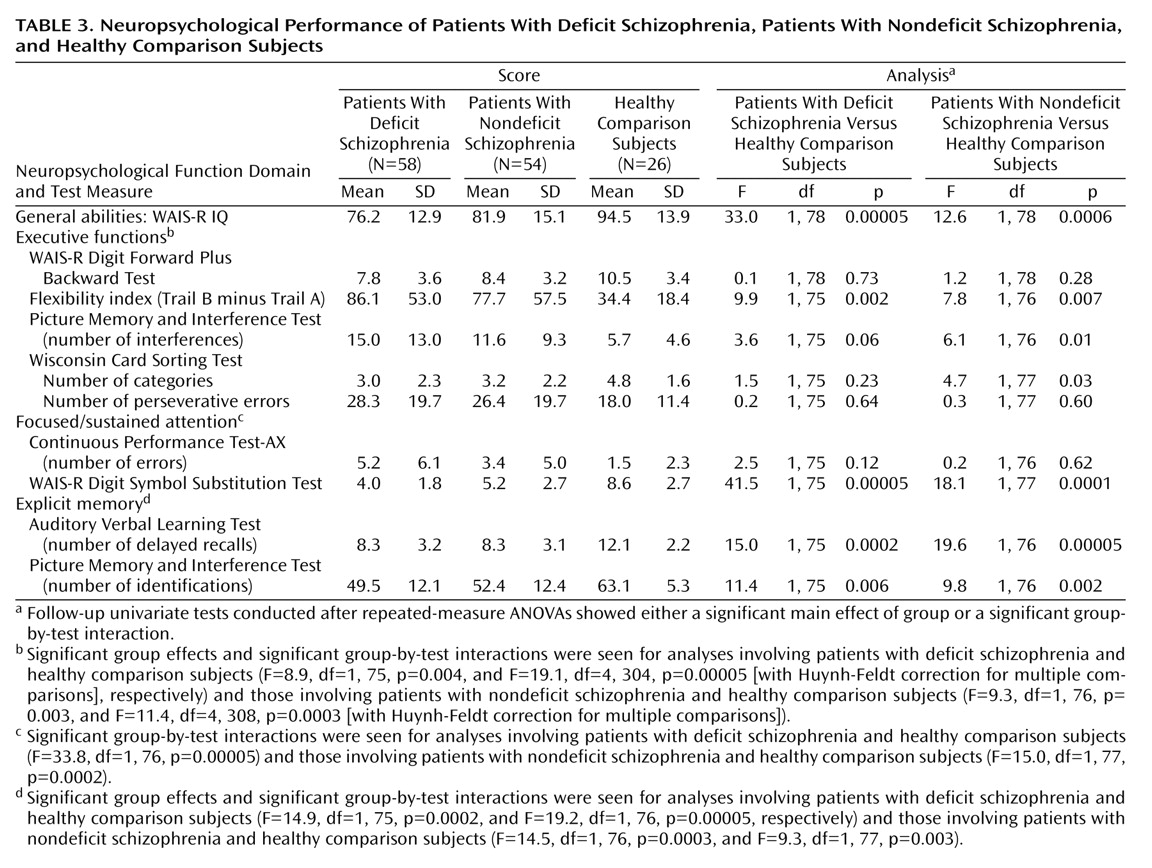
Neuropsychological functioning of patients and healthy comparison subjects was evaluated by their performance on tests that assessed five functional domains: 1) general abilities (WAIS-R IQ results); 2) executive functions (number of categories and perseverative errors on the Wisconsin Card Sorting Test
[33], number of interferences on the Picture Memory and Interference Test
[34], the part B minus part A time on the Trail Making Test
[35], and WAIS-R Digit Forward Plus Backward Test results); 3) focused/sustained attention (WAIS-R Digit Symbol Substitution Test results and the number of errors on the Continuous Performance Test-AX
[36]); 4) explicit memory (number of delayed recalls on the Auditory Verbal Learning Test
[34] and the number of identifications on the Picture Memory and Interference Test
[34]); and 5) visuospatial abilities (Benton Judgment of Line Orientation
[37] and the WAIS-R Block Design results).
The Continuous Performance Test-AX was administered by computer; all other tests used the traditional manual method. Tests were administered in the morning (from 9:00 a.m. to 12:00 noon) in a fixed order (Auditory Verbal Learning Test, Benton Judgment of Line Orientation, Wisconsin Card Sorting Test, WAIS-R, Continuous Performance Test, Picture Memory and Interference Test, Trail Making Test [part A], Trail Making Test [part B]).
Data Analyses
Data distributions were examined for normality and homogeneity of variance. In cases in which these assumptions were violated, data were log transformed.
Categorical variables were analyzed by the chi-square test. For continuous variables, in order to deal with the problem of multiple comparisons, the following four strategies were adopted. First, for each investigated area, the lowest possible number of indices was included. Second, ANOVAs were performed, with multivariate or repeated-measures designs used when deemed appropriate. Third, Huynh-Feldt correction for multiple comparisons was used when needed. Fourth, univariate follow-up tests were carried out only when a significant main effect or interaction had been found in the multivariate test.
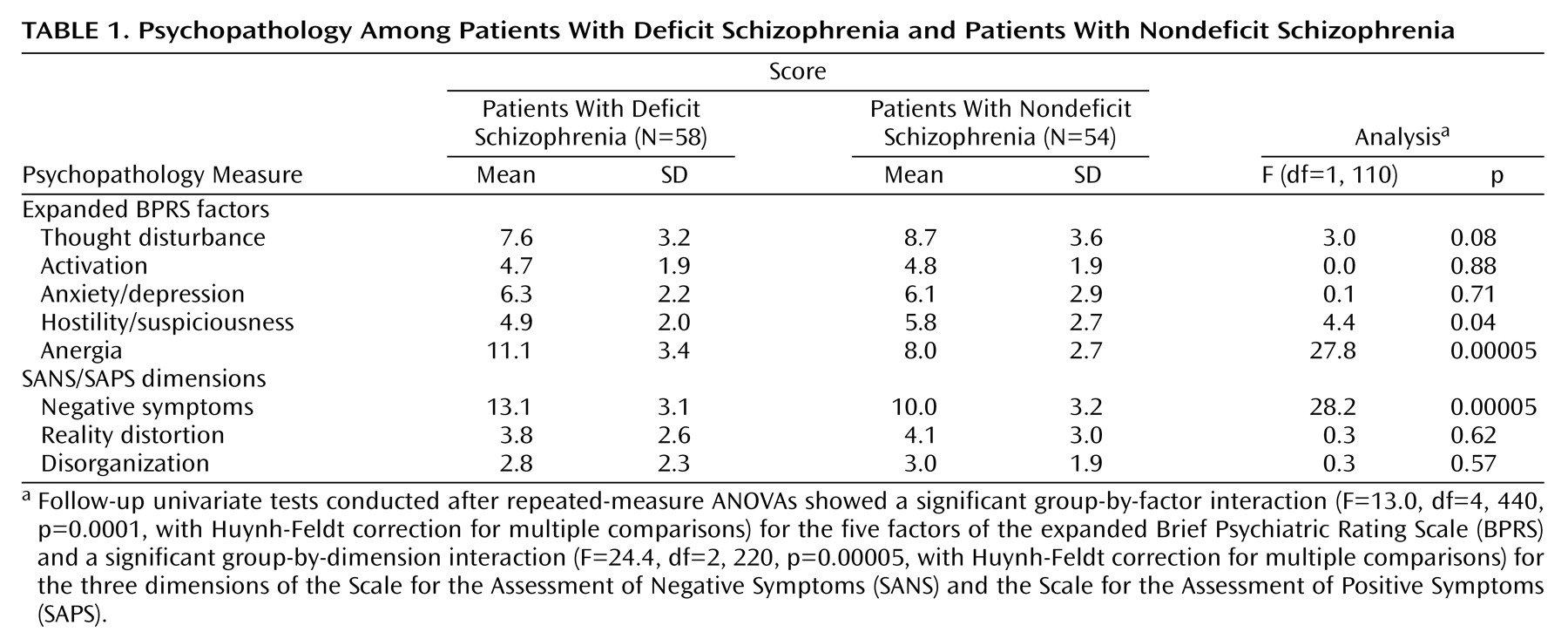
Psychopathological variables were reduced to eight, on the basis of previous factor analysis studies of the instruments used in this study. Measures from the SANS and SAPS were grouped into three dimensions
(38–
41): 1) negative symptoms (sum of global scores on the alogia, anhedonia, affective flattening, and avolition subscales of the SANS); 2) reality distortion (sum of global scores on the hallucinations and delusions subscales of the SAPS); and 3) disorganization (sum of global scores on the formal thought disorder and bizarre behavior subscales of the SAPS). Measures from the expanded BPRS were grouped into five factors
(42): 1) thought disturbance (sum of scores on the conceptual disorganization, hallucinatory behavior, and unusual thought content items); 2) activation (sum of scores on the tension, mannerisms/posturing, and excitement items); 3) anxiety/depression (sum of scores on the anxiety, guilt feelings, and depressed mood items); 4) hostility/suspiciousness (sum of scores on the suspiciousness, hostility, and uncooperativeness items); and 5) anergia (sum of scores on the blunted affect, emotional withdrawal, and motor retardation items).
For the Neurological Evaluation Scale, the score on each of the three factors was included in the analysis; they were preferred to the total score as they test different functions of the CNS.
Independent two-way ANOVAs for repeated measures on SANS/SAPS dimensions, expanded BPRS factors, and Neurological Evaluation Scale factors were used to test group differences between patients with deficit schizophrenia and patients with nondeficit schizophrenia (group, between; dimension/factor, within). A repeated-measure ANOVA was not deemed appropriate for the Premorbid Adjustment Scale, since the subscores were obtainable only for epochs preceding the age at illness onset, which was different for different subjects, yielding missing data for many of them for some epochs. In this case, a Bonferroni correction was applied (p=0.05/5=0.01).
Neuropsychological variables were grouped into the five aforementioned functional domains. To deal with the problem of multiple comparisons, a multivariate analysis of variance (MANOVA) on all neuropsychological indices was used to assess group differences among subjects with deficit schizophrenia, subjects with nondeficit schizophrenia, and healthy comparison subjects. Only in the presence of a MANOVA significant group effect were follow-up two-way ANOVAs for repeated measures performed on each neuropsychological domain, with diagnosis (deficit schizophrenia, nondeficit schizophrenia, healthy comparison) as a between-group factor and test as a within-group factor. Analyses of covariance (ANCOVAs), with general abilities (the summed age-corrected scores on the WAIS-R vocabulary and picture completion subscales) entered as a covariate, were planned to exclude the influence of group IQ differences on the other tested functions
(43). For those domains showing significant group effects or interactions, follow-up analyses of simple effects were planned to locate the source of the main effect or interaction.
For measures showing significant differences between patients with deficit schizophrenia and patients with nondeficit schizophrenia, a further question was addressed, i.e., whether the deficit state remained significantly associated with these measures when combinations of several confounding variables (demographic or clinical) were simultaneously controlled. To this aim, stepwise multiple regression analyses were performed in which the deficit/nondeficit categorization, the SANS/SAPS dimensions, age, education, duration of illness, the Simpson-Angus average score, and the chlorpromazine-equivalent doses were entered as independent variables.