While prefrontal and temporal cortical dysfunction are associated with schizophrenia, recent attention has focused on the role of the thalamus in this illness
(1,
2). The thalamus plays a critical role in the processing and integration of sensory information relevant to emotional and cognitive function. Abnormal thalamic activity, or a disturbance of corticothalamic circuitry, may contribute to sensory and cognitive deficits that schizophrenia patients often experience
(3). In fact, multiple thalamic abnormalities have been reported in schizophrenia. Neuroimaging studies have consistently reported decreased thalamic volume
(4–
7) and altered metabolism
(8–
10) in patients with schizophrenia. Postmortem morphometric studies and magnetic resonance imaging investigations have found volume loss in specific thalamic nuclei, including the dorsomedial
(6,
11–13), central medial, posteromedial
(6), and anterior
(6,
14) nuclei as well as the pulvinar
(11). It is of interest that thalamic cell loss is most often found in the dorsomedial and anterior nuclei (which are reciprocally connected to limbic brain regions frequently implicated in schizophrenia) but not in nuclei with primarily nonlimbic projections
(13).
While there is evidence of structural and functional thalamic changes in patients with schizophrenia, few studies have examined associated neurochemical abnormalities
(15–
18). Thalamic efferents, as well as corticothalamic and sensory afferents, predominately use glutamate as a neurotransmitter, which activates ionotropic (NMDA, AMPA, kainate) and metabotropic receptors expressed throughout thalamic nuclei. Several lines of investigation have implicated glutamatergic dysfunction in the pathophysiology of schizophrenia
(19,
20). The glutamatergic hypothesis of schizophrenia rests largely on pharmacological observations, since NMDA receptor blockade with dissociative anesthetics like phencyclidine produces psychosis and cognitive deficits in normal subjects and exacerbates these symptoms in patients with schizophrenia
(21). Moreover, enhancing NMDA receptor-mediated neurotransmission either with
D-cycloserine (an agonist of the glycine/
D-serine co-agonist site of the NMDA receptor) or ampakines (which stimulate AMPA receptors and indirectly enhance NMDA receptor function) improves negative symptoms in some patients
(20,
22). Finally, postmortem studies have identified abnormalities of glutamate receptor expression and subunit composition in multiple limbic brain regions implicated in schizophrenia, including the thalamus
(15), prefrontal cortex
(19,
23,
24), and temporal lobe structures
(25–
28). Together, these findings suggest that a perturbation of NMDA receptor-mediated glutamate neurotransmission in the thalamus and other limbic structures may contribute to the pathophysiology of schizophrenia.
The NMDA receptor is a multimeric ligand-gated ion channel formed by subunits encoded by seven genes: NR
1, NR
2A-D, and NR
3A-B (29,
30). NR
1 is an obligate subunit expressed as one of eight isoforms, due to the alternative splicing of exons 5, 21, and 22
(29). Exon 5 encodes an N-terminal cassette that influences receptor desensitization and sensitivity to protons, zinc ions, and polyamines
(31). Exons 21 and 22 encode two C-terminal cassettes (C1 and C2). These C-terminal cassettes modulate NMDA receptor function by influencing the association of NMDA receptor subunits with each other
(32) and with other intracellular molecules, including protein kinase C
(33), calmodulin
(34), and postsynaptic density-associated proteins like PSD95
(35,
36). These intracellular molecules modulate NMDA receptor activity and participate in signal transduction pathways. Consequently, alternative splicing of NR
1 has a substantial impact on NMDA receptor structure, function, and related intracellular signaling.
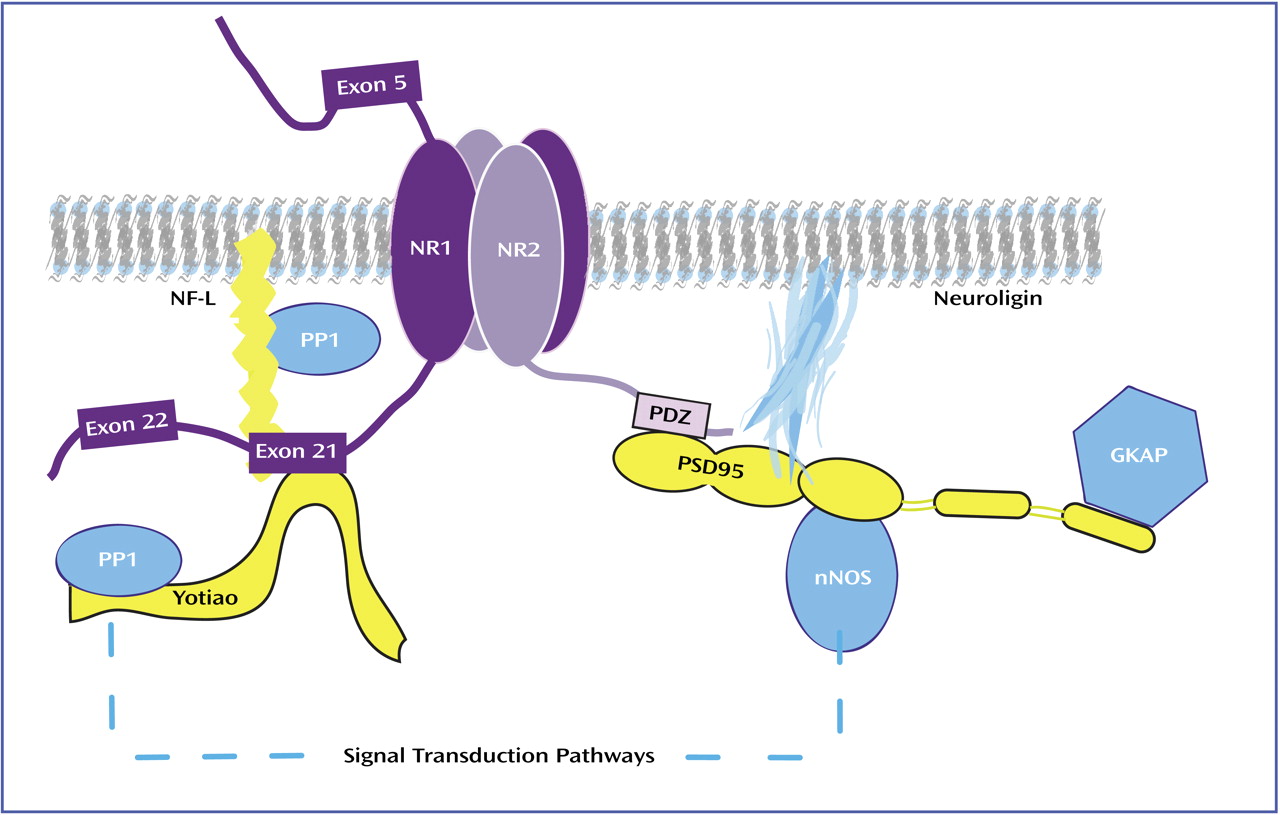
Glutamate neurotransmission involves not only glutamate receptors but also intracellular molecules enriched in postsynaptic density proteins. PSD95, the prototype of this family, and related proteins contain domains that bind the C-termini of NMDA receptor subunits, cytoskeletal proteins, and signal transduction enzymes (
Figure 1). Through protein-protein interactions, PSD95 promotes clustering and anchoring of the NMDA receptor at the postsynaptic density
(35), modulates NMDA receptor sensitivity to glutamate
(37), and, perhaps most importantly, assembles signaling complexes that coordinate NMDA receptor-related intracellular processes
(38). An alteration of the expression of PSD95-like molecules, or of the interaction between NMDA receptor subunits and these proteins, can impede NMDA receptor–mediated intracellular signaling. To date, two studies have examined the expression of PSD95 in schizophrenia and reported increased mRNA expression in the occipital cortex
(24) and decreased expression in Brodmann’s area 9 of the prefrontal cortex
(39) but no change in area A6 of the prefrontal cortex
(24) or hippocampus
(39).
We have reported alterations of glutamatergic molecules in the thalamus in schizophrenia
(15–
17), including decreased expression of NR
1 subunit transcripts and decreased binding to the polyamine and glycine sites of the NMDA receptor, with the most prominent changes occurring in nuclei reciprocally linked to limbic regions
(15). In the present study, we hypothesized that this reduction of NR
1 subunits is specifically associated with particular isoforms and used in situ hybridization to determine transcript expression for exon 5-, 21-, and 22-containing NR
1 isoforms in the thalamus. We also determined whether the expression of the PSD95 family of molecules is altered in the thalamus in schizophrenia by using in situ hybridization to measure the transcript expression of a set of NMDA receptor-associated postsynaptic density proteins in these same subjects.
Method
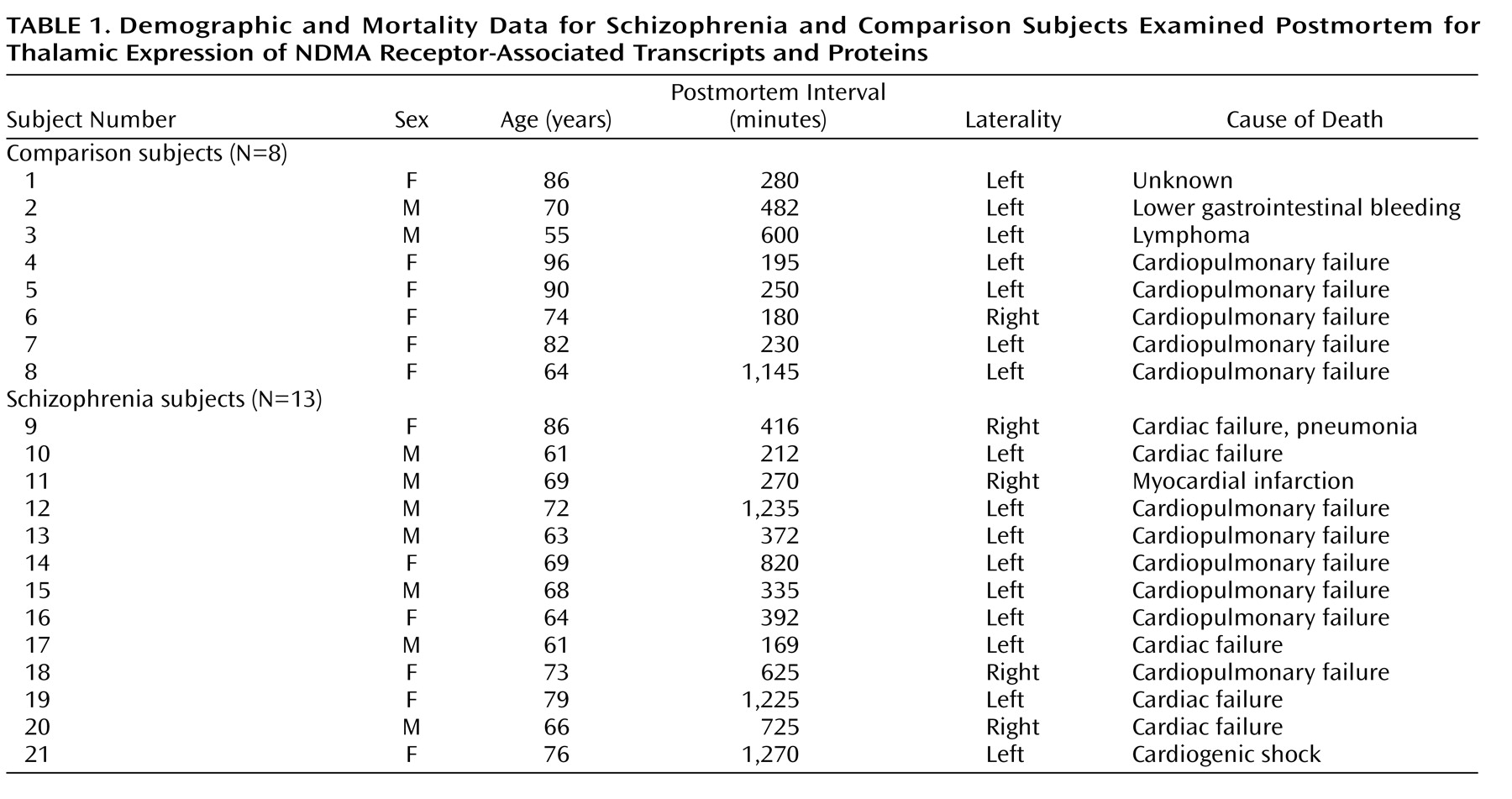
Thirteen subjects with schizophrenia and eight nonpsychiatrically ill individuals from the Mount Sinai Medical Center Brain Bank were studied (
Table 1). Next of kin consent was obtained for each subject. There were no significant differences between the diagnostic groups in age (t=1.59, df=19, p=0.12), postmortem interval (t=1.18, df=19, p=0.25), or sex distribution (χ
2=1.68, df=1, p=0.19). Each schizophrenia and comparison brain underwent neuropathological examination, and Alzheimer’s disease was specifically ruled out in each case. Brains were prepared by slicing one hemisphere into 1-cm coronal slabs, which were snap frozen. Blocks containing thalamus were cryostat-sectioned (20 μm thick), thaw-mounted onto poly-
L-lysine-subbed microscope slides, dried, and stored at –80°C. Two slides per subject were prepared for in situ hybridization for each probe.
Antisense oligodeoxyribonucleotides were synthesized for NR
1 exons 5 (corresponding bases 1022-1066), 21 (3152-3196), and 22 (3258-3302) to use as in situ hybridization probes. Regions of minimal homology to other genes were identified and verified through a search of Genbank and EMBL databases, using the National Center for Biotechnology Information BLAST network service. Antisense oligonucleotides were purified by high-performance liquid chromatography, diluted to a concentration of 1 μg/μL and stored at –20°C. Oligonucleotides were terminally labeled with 50 μCi of [
35S]dATP (New England Nuclear, Boston) with 1.5-μl terminal deoxynucleotidyl transferase (Boehringer-Mannheim, Mannheim, Germany) and used for in situ hybridization as we have previously described in an earlier report examining the distribution of these exons in macaque thalamus
(40).
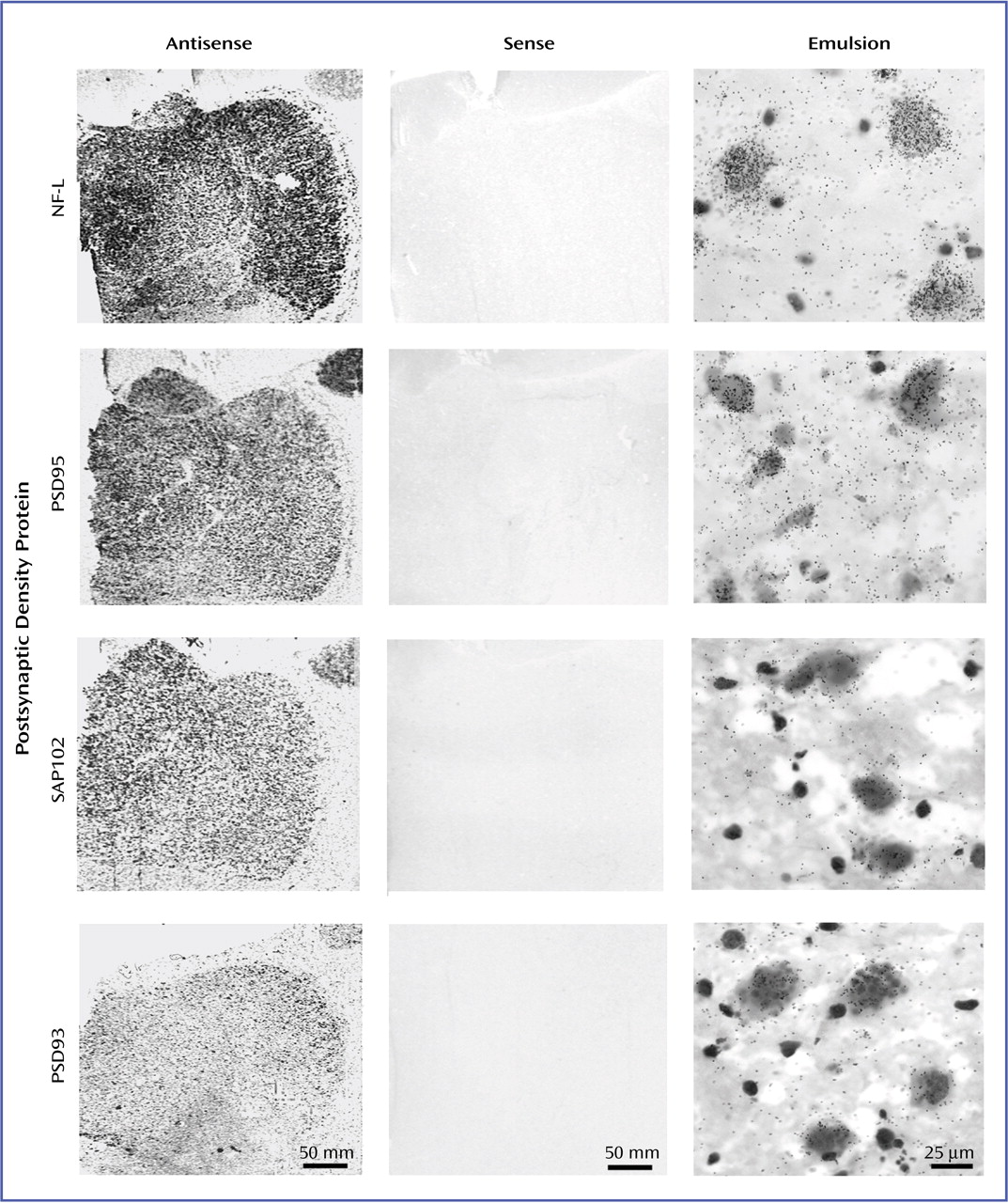
We performed in situ hybridization with riboprobes to determine the thalamic expression of transcripts encoding the NMDA receptor-related postsynaptic density proteins NF-L, PSD93, PSD95, SAP102, and Yotiao. We amplified unique regions of NF-L (Genbank accession number: X05608, nucleotide coding region: 456-950), PSD93 (NM_001364, 587-1082), PSD95 (U83192, 1254-1744), SAP102 (U49089, 204-713), and Yotiao (5032230, 3652-4207) from a human brain cDNA library (EdgeBiosystems, Gaithersburg, Md.) by using polymerase chain reaction (PCR). Amplified cDNA segments were extracted (QIAquick Gel Extraction Kit, Qiagen, Valencia, Calif.), subcloned (Zero Blunt TOPO PCR cloning kit, Invitrogen, Carlsbad, Calif.), and confirmed by nucleotide sequencing (Thermo Sequenase Radiolabeled Termination Cycle Sequencing Kit, USB, Cleveland). Linearized subclones were used to synthesize [
35S]UTP-labeled riboprobes, as we have previously described
(40).
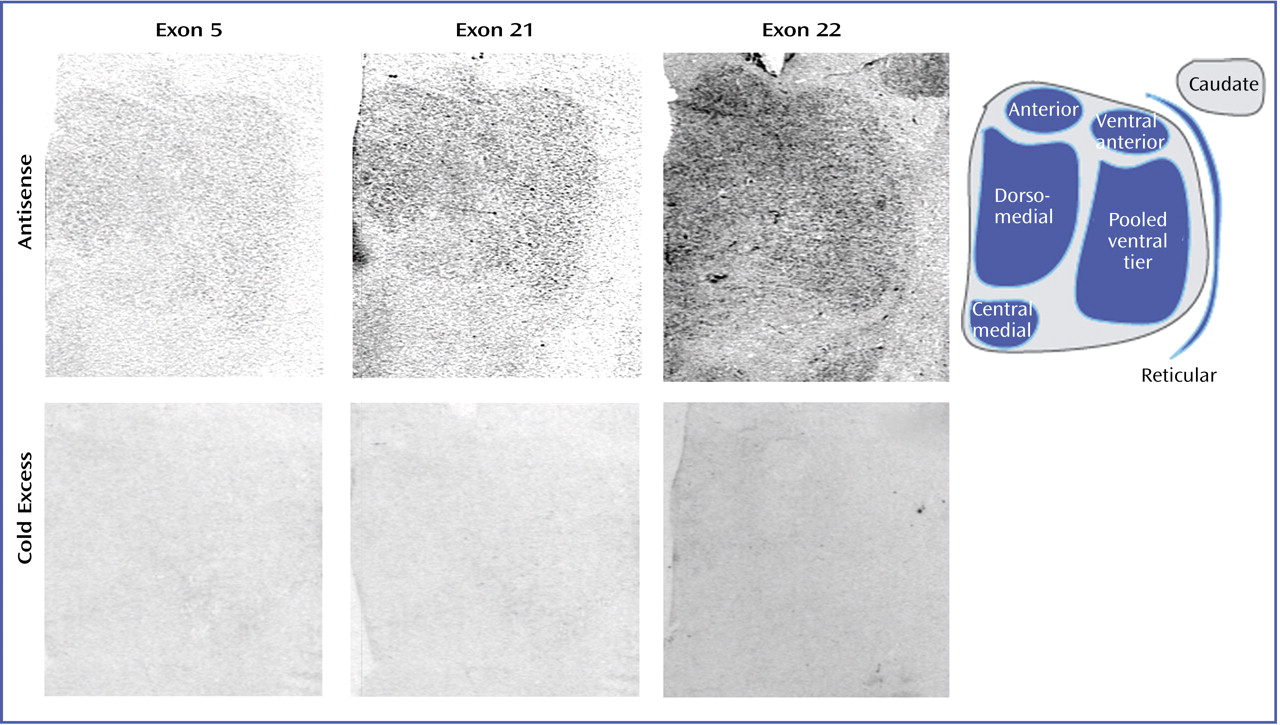
To ensure the specificity of in situ hybridization signals, multiple control studies were performed. For the NR
1 splice variants, adjacent slides were incubated with either labeled probe for exon 5, 21, or 22, or labeled probe in the presence of 100-fold excess of unlabeled oligonucleotide. For the postsynaptic density proteins, adjacent sections were incubated with sense strand probes and processed as previously described
(40). One set of these slides was exposed to Kodak film for 7–60 days, and a second set was dipped in Kodak NTB-2 emulsion and stored at 4°C for 21–90 days prior to developing
(41).
Film-based images were analyzed for mRNA expression on a nucleus-by-nucleus basis. As we have previously described
(15), we identified six discrete thalamic nuclei in each section: anterior, dorsomedial, central medial, ventral anterior, reticular, and pooled ventral tier nuclei. The nuclei were identified based on cellular and white matter patterns, defined by cresyl violet and gold chloride staining of sections from each subject
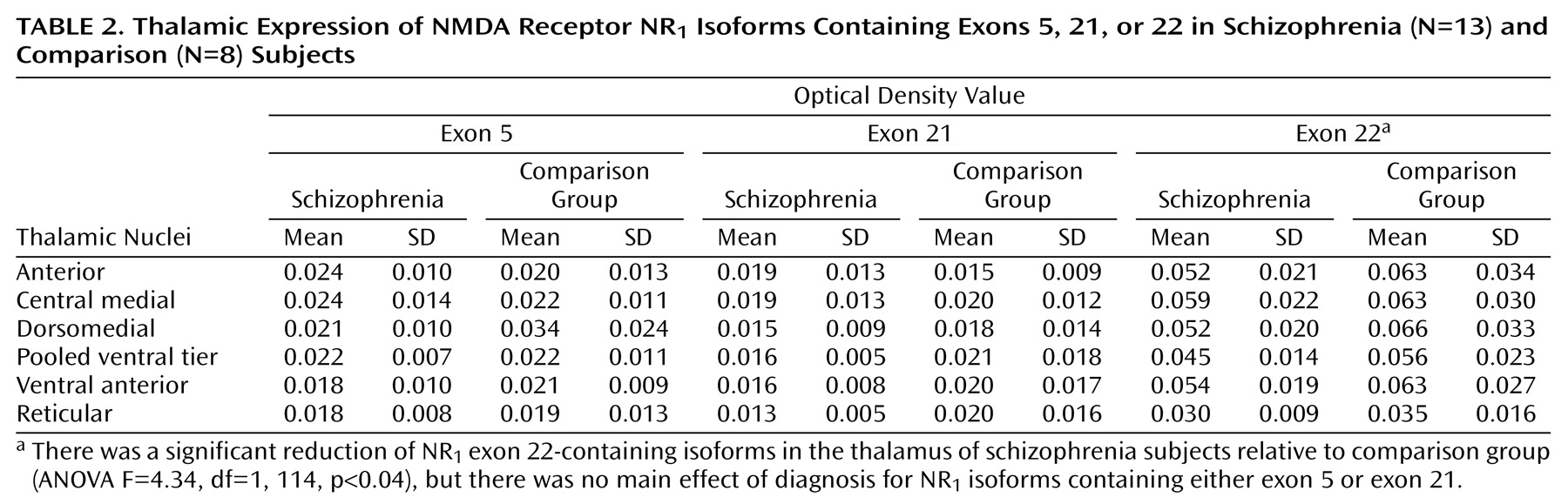
(15). Tissue background values from adjacent white matter were subtracted from grayscale values for each nucleus and converted to optical density. Data from the NMDA receptor isoform study are expressed as optical density values (
Table 2). Postsynaptic density protein data are expressed as mRNA concentrations. The amount of radioactivity bound (in nCi/g) was determined by using [
14C] microscale standards (Amersham Biosciences, Piscataway, N.J.)
(42), which were exposed on the same film as the slides for each study. The number of labeled uridine nucleotides contained in each riboprobe and the specific activity of the [
35S]UTP were then used to convert bound radioactivity to concentration of mRNA per nucleus, expressed as fmol/g. For all studies, values for each nucleus from two sections per subject were averaged and used for statistical analysis, which was performed for each probe by two-way analysis of variance, with nucleus and diagnosis as independent variables, and optical density values (exon studies) or mRNA concentrations (postsynaptic density protein studies) as the dependent variables. The Kolmogorov-Smirnov test was used to ensure normality of all data. Analysis of covariance was performed with age and postmortem interval as covariates, and Pearson product-moment correlations were used to determine relationships between continuously distributed variables. For all tests, alpha=0.05.
Discussion
We recently reported alterations in the expression of NMDA receptor subunits NR
1 and NR
2C and other molecules involved in glutamatergic neurotransmission in the thalamus of subjects with schizophrenia
(15–
17). In the present study, using the same subjects that were used in these earlier studies, we have characterized the thalamic expression of NR
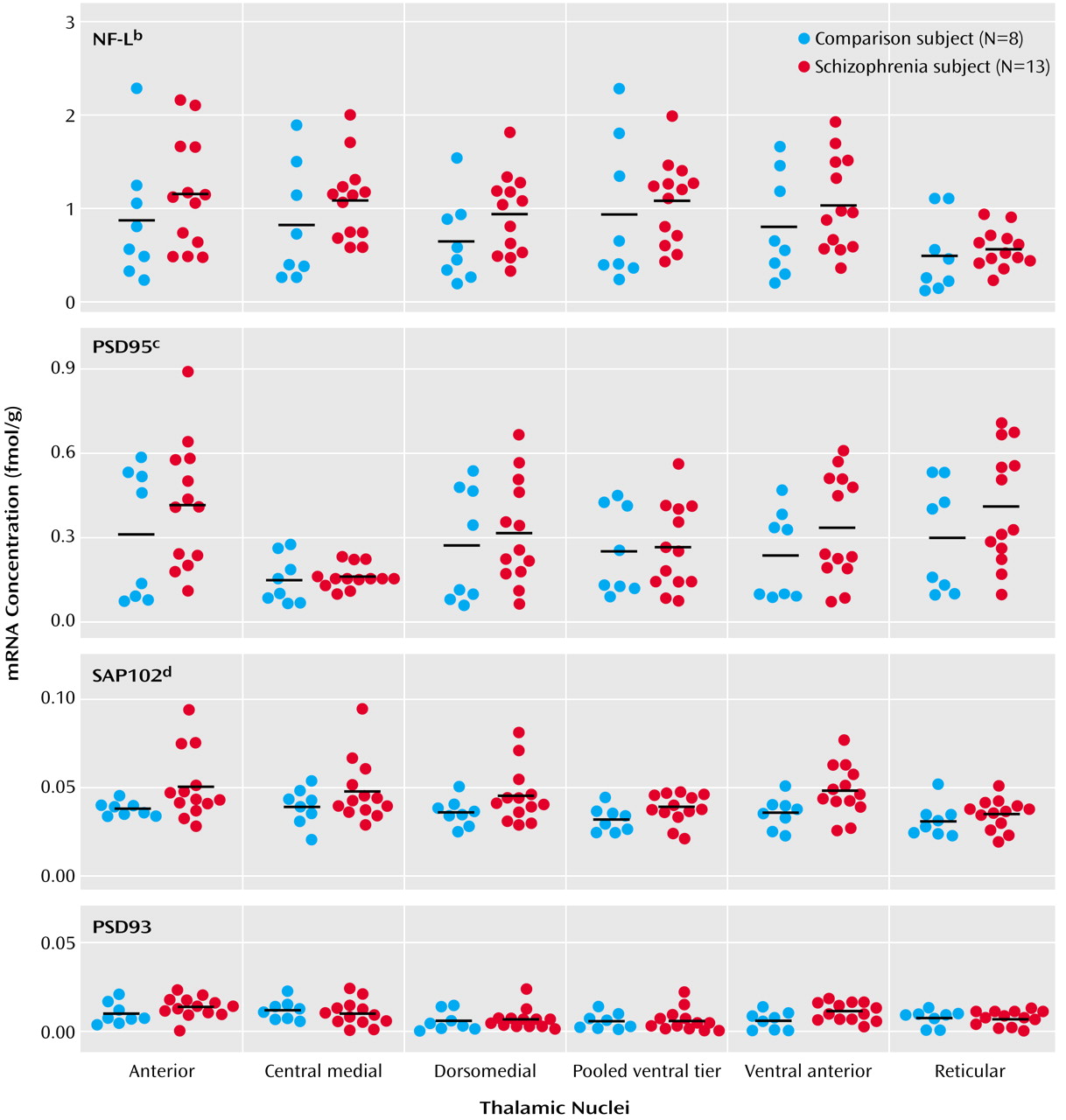
1 subunit isoforms in schizophrenia and found that this reduction is restricted to exon 22-containing isoforms. Moreover, we have examined the expression of postsynaptic density proteins that associate with NMDA receptor subunits and found a significant increase in the transcript expression of the NR
1-associated protein NF-L and NR
2-associated proteins PSD95 and SAP102.
The NMDA NR
1 subunit is obligate, and numerous studies have indicated that alternative splicing of the N- and C-terminal cassettes produces functionally distinct NMDA channels
(31). Our results are consistent with previous reports of NR
1 splice variant expression patterns in the rodent, macaque, and human brain
(40,
43–45). Exon 5-containing isoforms are generally rare but are expressed at low to moderate levels in the rodent
(45) and macaque thalamus
(40). Moderate to high levels of expression of exon 22-containing isoforms have been reported in the cortex, hippocampus, striatum, and thalamus of rat
(45) and macaque
(40). Our results coincide with this work, since we found modest levels of exon 5-containing isoforms and relatively high levels of exon 22-containing isoforms in the human thalamus, consistent with the relative expression of these exons in macaque
(40).
Altered NR
1 subunit isoforms may reflect changes in NMDA receptor subunit composition, which in turn may have significant functional consequences, since alternative splicing of exons 5, 21, and 22 can modulate NMDA receptor localization and function
(33,
34,
46). Most studies have focused on the functional significance of the exon 5-encoded N1 cassette, the exon 21-encoded C1 cassette, or the C2′ cassette, which is a unique C-terminal tail that is produced when the exon 22-encoded C2 cassette is excluded. Omission of exon 5 modifies NMDA receptor desensitization and its sensitivity to protons, zinc ions, and polyamines
(31). Alternative splicing of the C1 cassette reduces protein kinase C potentiation of NMDA receptor activity
(46,
47) and also influences the receptor’s intracellular distribution
(48) and cell surface expression
(49). NR
1 isoforms that contain both C1 and C2 cassettes are expressed at low levels on the cell membrane in vitro, whereas isoforms that contain only the C2′ domain are highly expressed
(49). It is of interest that the C2′ cassette contains a PDZ-binding sequence that is thought to be involved in trafficking the NMDA receptor to the synaptic membrane
(50,
51), suggesting that NR
1 isoforms differentially interact with postsynaptic density proteins and other cytoskeletal components to target the NMDA receptor to the postsynaptic density proteins.
We observed a specific reduction of NR
1 isoform transcripts containing exon 22 in the thalamus in schizophrenia. We did not detect a change in the expression of exon 5 or 21 transcripts. Because of the low expression levels of these two isoforms, however, our inability to find a further decrease of one or both in schizophrenia may be a floor effect. Given the paucity of information regarding the functional role of the exon 22-encoded C2 cassette, it is difficult to interpret what effect would result from decreased expression of exon 22-containing isoforms. When the C2 cassette is excluded, it is replaced by the C2′ cassette, so perhaps downregulating the expression of the C2 cassette results in greater numbers of isoforms with C2′, which are more likely to be transported to the cell surface
(49).
PSD95 and similar NR
2-associated proteins, SAP102 and PSD93, contain domains that bind numerous intracellular molecules, including the C-termini of NR
2 subunits, cytoskeletal proteins, and signal transduction enzymes
(38), which influence NMDA receptor function and NMDA receptor-associated intracellular processes
(38,
52). We found a significant increase in the transcript expression of PSD95 and SAP102, but not PSD93, in the thalamus in subjects with schizophrenia. The total pool of postsynaptic density proteins may be increased in schizophrenia in an attempt to compensate for decreased thalamic expression of NMDA receptors. Enhanced expression of postsynaptic density molecules may lead to an enhanced association of the postsynaptic density proteins with the remaining NMDA receptor subunits, an intracellular adaptation to attempt to maintain homeostasis of NMDA receptor-related intracellular signaling in the face of decreased NMDA receptor expression or in response to a general deficit in glutamate neurotransmission.
Widespread interest in the NR
2 subunit-associated PSD95 family of proteins has prompted the investigation of intracellular proteins that interact with the NR
1 subunit
(53,
54). Two proteins, NF-L
(53) and Yotiao
(54), have been identified as proteins that interact with exon 21-containing isoforms. NF-L, along with the two other neurofilament subunits, NF-heavy and NF-medium, are among the most abundant cytoskeletal elements and play an important role in the maintenance of neuronal structure
(55). NF-L may also be involved in directing NMDA receptors to the postsynaptic density or linking it to the synaptic cytoskeleton
(48,
53). NF-L interacts with protein phosphatase-1 (PP1), a major protein/serine/threonine phosphatase that is involved in numerous intracellular processes
(56). Although the functional significance of this NF-L/PP1 interaction is not fully understood, it has been suggested that NF-L may bind PP1 and position it to dephosphorylate other postsynaptic density proteins, such as NMDA receptor subunits, or CamKII
(57).
We found that NF-L mRNA expression was significantly elevated in the thalamus in subjects with schizophrenia. Although a portion of NF-L protein in the cell may interact with the NR1 subunit to participate in NMDA receptor function, the majority of NF-L associates with the other neurofilament subunits to maintain the neuronal cytoskeleton. It is not possible to determine if the elevated NF-L mRNA we found in schizophrenia subjects translates into greater amounts of NF-L protein interacting with NMDA receptors, which might serve to boost an impaired glutamatergic system, or if it is part of a more general cytoskeletal response to the illness.
Our study has several limitations. These data are from elderly subjects, so generalization of our findings to younger patients should be made with caution. A limitation of all postmortem studies in schizophrenia is the possible effect of chronic neuroleptic treatment, since these medications potentially regulate the systems under study. While antipsychotic medications have been shown to alter thalamic metabolism and immediate early gene expression
(58–
60), they do not appear to affect thalamic NMDA receptor expression
(61). To date, no studies have directly examined the effect of neuroleptics on the expression of NMDA receptor-related postsynaptic density molecules. However, Dracheva and colleagues
(24) reported altered PSD95 and NMDA receptor subunit expression in the prefrontal and occipital cortices in schizophrenia and found that these transcripts did not differ between patients who were taking antipsychotic medication within 6 weeks of death and those who were medication-free for greater than 6 weeks
(24).
Numerous studies support a hypothesis of glutamatergic dysfunction in schizophrenia. While there are reports of abnormal ionotropic glutamate receptor expression in various limbic regions in schizophrenia, the pathophysiology of this illness may not be solely associated with abnormal receptor expression. Rather, it may also involve abnormalities of the intracellular signaling machinery that is linked to the NMDA receptor. The recently identified PSD95 family of molecules represents an important link between the NMDA receptor and the intracellular enzymes that mediate NMDA receptor-associated signaling. Abnormal expression of these postsynaptic density proteins, or an alteration of the interaction between NMDA receptors and these proteins, may significantly impact NMDA receptor-associated signal transduction. In this study, we show that three of these NMDA receptor-associated postsynaptic density proteins are abnormally expressed in the thalamus in schizophrenia. These data provide further evidence of glutamatergic dysfunction in schizophrenia and suggest that glutamatergic abnormalities in this illness may occur not only at the level of receptor expression but also within intracellular pathways associated with glutamate receptor-associated signal transduction.