The pons and thalamus are both critical nodes in multiple circuits linking the cerebellum to the motor and sensory cortices and to association areas in the frontal and prefrontal cortex that subserve higher-order behavior
(1–
3). The pons is primarily a white matter structure of the brainstem composed of several systems of large white matter tracts and nuclear relays. The thalamus is a highly differentiated gray matter structure that comprises many subnuclei, each with specialized functional links to multiple cortical sites. Both the pons and the thalamus are adversely affected by excessive alcohol consumption. Central pontine myelinosis has been associated with alcoholism
(4), and its defining neuroradiological sign on clinical T
2-weighted magnetic resonance imaging (MRI) scans is a hyperintense triangular lesion
(5). Damage to the pontine system in uncomplicated alcoholism is seen in vivo with increased T
2 relaxation times
(6) and in pathological reports of a lower than normal number of cholinergic neurons in the locus ceruleus, a substructure of the pons
(7–
9). Alcohol also has deleterious effects on the thalamus that are observed as deficits in overall volume
(10) and in the volumes of specific nuclei
(11) and deficits in the number and size of neurons, particularly in the anterior thalamus
(12). Furthermore, the size of the thalamus has been associated with odor identification ability in patients with alcoholism
(13).
The pons has also been implicated in the pathophysiology of schizophrenia, although relatively few reports have examined this relationship, and evidence for an overall volume deficit has not emerged
(14). However, pathology studies have found lower levels of choline acetyltransferase in the pons of patients with schizophrenia, relative to nonschizophrenic comparison subjects
(15), and greater numbers of neurons in the pedunculopontine nucleus
(16). In vivo studies have reported lower
N-acetylaspartate/creatine ratios in the pons of patients with schizophrenia, relative to comparison subjects
(17), but no pontine area deficit, as measured on a single sagittal slice
(14). In contrast to the limited amount of research on the pons, more studies have examined the role of thalamic pathology in the development of schizophrenia (e.g., reference
18) and in the cognitive dysmetria associated with the disorder (e.g., reference
19). In vivo evidence for gross volume deficits in the whole thalamus has been mixed, although a meta-analysis identified a small-to-moderate, statistically significant effect size for thalamic volume deficits
(20). Although evidence for gross thalamic volume deficits is equivocal, the evidence for an abnormally small size of specific nuclei, particularly the mediodorsal nucleus, in schizophrenia is stronger
(21,
22). Further, in vivo spectroscopic studies have reported abnormally low levels of thalamic
N-acetylaspartate/creatine in schizophrenia
(23,
24).
Neuroleptic medication can affect thalamic volume. One study reported that the thalamus was smaller in neuroleptic-naive patients but enlarged in patients treated with neuroleptics and that the extent of enlargement was dose dependent regardless of whether the patients were treated with typical or atypical antipsychotic medications
(25). Similar findings have been reported for other diencephalic structures, such as the basal ganglia
(26,
27), with different effects related to the type of antipsychotic medication. In one study, caudate volumes, enlarged in patients treated with typical antipsychotic medications, declined after 24 weeks of treatment with atypical antipsychotic medications
(28). In another study, mean basal ganglia volume increased over a 2-year period in patients predominantly treated with typical antipsychotic medications but decreased in patients predominantly treated with atypical antipsychotic medications
(29). Structural neuroimaging studies have not reported differential effects of typical and atypical antipsychotic medications on thalamic volume. A recent positron emission (PET) study, however, reported that a typical antipsychotic medication induced significantly higher dopamine D
2 receptor binding in the thalamus, compared with atypical antipsychotics
(30), suggesting a mechanism by which the volume differences associated with the various antipsychotic medication types could occur.
Alcohol abuse and dependence occur with high prevalence in schizophrenia and pose exceptional risks of brain damage to patients with this disorder, especially in regions that are vulnerable to the toxic effects of alcohol. In regions already compromised by schizophrenia, such as the frontal lobes and the cerebellum, even relatively mild alcohol abuse has a particularly deleterious effect
(31,
32). This compounding effect has also been demonstrated for ataxia, a behavioral manifestation of cerebellar damage
(33). In this study, we sought to determine whether a compounded deleterious effect of alcoholism on schizophrenia holds true for either the pons or the thalamus—the principal intermediary nodes of the frontocerebellar circuit—and whether these effects may be mitigated by the type of antipsychotic medication patients receive.
Method
Study Participants
All subjects (
Table 1) were men and gave written informed consent to participate in MRI research. A detailed description of the selection criteria and the clinical and demographic characteristics of the study subjects have been provided in earlier reports
(31,
32). Patients with schizophrenia were recruited from an unlocked voluntary research ward at the Veterans Affairs (VA) Palo Alto Health Care System. The group with schizophrenia included 27 patients who met the DSM-III-R criteria for schizophrenia and did not meet the criteria for current or lifetime alcohol abuse or dependence or any other axis I diagnosis. The group with schizophrenia and comorbid alcohol dependence included 19 patients who met the DSM-III-R criteria for schizophrenia and who also met either the DSM-III-R criteria for alcohol dependence or the Research Diagnostic Criteria (as determined from the Schedule for Affective Disorders and Schizophrenia
[36]) for alcoholism. Participants with a diagnosis of nonalcohol substance dependence, but not those with a diagnosis of nonalcohol substance abuse, were excluded. All but three of the patients with either schizophrenia alone or schizophrenia and comorbid alcohol dependence were currently taking antipsychotic medications: 29 were taking typical antipsychotic medications, and 13 were taking atypical antipsychotic medications. (Data on type of medication were missing for one patient.) The alcohol-dependent patients (N=25) were drawn from a group of patients who met the DSM-III-R or DSM-IV criteria for a diagnosis for alcohol dependence but not for schizophrenia or any other axis I disorder. These subjects had earlier undergone brain imaging while they were inpatients at the VA Palo Alto Health Care System
(37) and were participating in follow-up studies that included administration of the Structured Clinical Interview for DSM-III-R or the Structured Clinical Interview for DSM-IV, medical screening, clinical review of MRI scans, and an updated assessment of alcohol consumption
(38,
39). The median time between their last drink and MRI scanning was 204 days (range=1–1,994 days).
Healthy comparison participants (N=51), recruited from the community by advertisement and word of mouth, were screened to exclude any potential subjects with axis I disorder, substance abuse in the year before entry into the study, or consumption of more than four drinks a day for more than a month. Lifetime alcohol consumption of all subjects was assessed by using a semistructured interview based on an interview developed by Skinner
(40,
41) and used previously by the authors (e.g., references
37,
42).
Group Comparisons of Demographic Variables
One-way analysis of variance (ANOVA) was used to test for demographic differences across the four groups. Significant differences were found for education (F=9.03, df=3, 118, p=0.0001), general intelligence estimated with the National Adult Reading Test
(34) (F=4.29, df=3, 95, p<0.01), and total lifetime consumption of alcohol (F=59.24, df=3, 103, p=0.0001). No significant differences were found for age (F=2.16, df=3, 118, n.s.) and handedness, as measured by a quantitative test
(35) (F=0.78, df=3, 111, n.s.). Follow-up Scheffé tests (alpha ≤0.05) revealed that the alcohol-dependent group consumed more alcohol than the group with schizophrenia and comorbid alcohol dependence, which consumed more than the schizophrenia group or the healthy comparison group. However, the three patient groups had equivalent numbers of years of education and equivalent IQs. The group with comorbid disorders did not differ significantly from the schizophrenia group in Brief Psychiatric Rating Scale (BPRS) scores.
Among the patients with schizophrenia, there were no group differences in age, BPRS scores, IQ, years of education, or handedness by medication type (typical versus atypical) or by the presence or absence of alcohol dependence comorbidity.
Image Acquisition and Analysis
MRI scans were acquired on a 1.5-T General Electric Signa scanner by using a three-dimensional spoiled gradient recalled sequence (TR=24 msec; TE=5 msec; flip angle=40°; 124 slices; field of view=24 cm; 256×196 matrix; reconstructed resolution=0.9×0.9×1.5 mm; acquired resolution=0.9×1.2×1.5 mm). Image data were reformatted to 1-mm isotropic voxels and aligned along the anterior-posterior commissure plane and interhemispheric fissure. All images were coded to allow processing to be performed by researchers who were blind to the subject’s identity, age, and sex.
The ventral (basilar) pons and the thalamus were manually traced by K.L.S. following visible borders on every third 1-mm- thick sagittal slice, with the medial slice at mid-sagittal plane. All measurements were completed twice, and the volume was the mean of the two measurements. The most lateral boundaries of both structures were first estimated by marking the limits on the coronal and axial views. The shape of the pons was smooth and ovoid (
Figure 1). The dorsal borders were formed by the medial and lateral lemnisci that appeared as a strip of reduced signal intensity. The most lateral aspects were traced until partial voluming made it difficult to distinguish the ventral pons from the superior cerebellar peduncles, approximately 10 mm from the midline, where the brainstem is transected by the calcarine fissure. Visible borders of the left and right thalamus were first apparent at approximately 15–18 mm from each side of the midline. The rostral border of the thalamus forms part of the floor of the lateral ventricles and is bounded by the fornix and the body of the caudate nucleus (laterally). Caudally, the sulcus hypothalamicus marks the boundary of the thalamus and the subthalamus. The dorsal surface of the thalamus, the pulvinar, forms a part of the floor of the third ventricle and is clearly delimited by the retropulvinal (subarachnoid) cistern. The ventral boundaries of the thalamus are the third ventricles (medially) and the internal capsule (laterally). The left and right thalami were randomly reversed in orientation on the display screen to prevent measurement bias.
Statistical Analysis
Because the size of the pons was associated with intracranial volume and the size of the thalamus with age (see Results), we applied a two-step regression analysis based on the data from the healthy comparison subjects to calculate intracranial-volume- and age-corrected z scores
(37). Measures for the pons and thalamus were regressed against an estimate of intracranial volume that was based on a three-dimensional volume of the cerebrum
(32). The resulting regression coefficients were used to calculate intracranial-volume-residualized scores on the basis of the regression analysis. These values were then regressed on age, and values from individual subjects were converted to standardized z scores as the final unit of analysis. The expected mean of the comparison subjects was z=0 (SD=1). Use of standardized z scores permitted assessment of the effects of disease, having removed the effects of variation due to normal aging and differences in intracranial volume. Data were analyzed first as absolute volumes and then as age-adjusted and intracranial-volume-adjusted scores. Group differences in pons and thalamus volumes were assessed with four-group, one- or two-way ANOVAs with follow-up Scheffé tests (alpha=0.05). Absolute values for the pons and thalamus were also subjected to analysis of covariance (ANCOVA), with intracranial volume as the covariate in the analysis of pons volumes and age as the covariate in the analysis of thalamus volumes.
Results
Among healthy comparison subjects, the correlation of absolute regional volume with age was significant for the bilateral thalamus (left plus right) (r=–0.53, N=51, p<0.0001) but not the pons (r=0.15, N=51, n.s.). Intracranial volume was significantly correlated with neither the pons volume (r=0.21, N=51, n.s.) nor the bilateral thalamic volume (r=0.02, N=51, n.s.) in the healthy comparison group; however, in all groups combined, pons volume and intracranial volume were correlated significantly (r=0.29, N=122, p=0.001). Further, a one-way ANOVA indicated that the four groups did not differ in intracranial volume (F=0.41, df=3, 118, p=0.75).
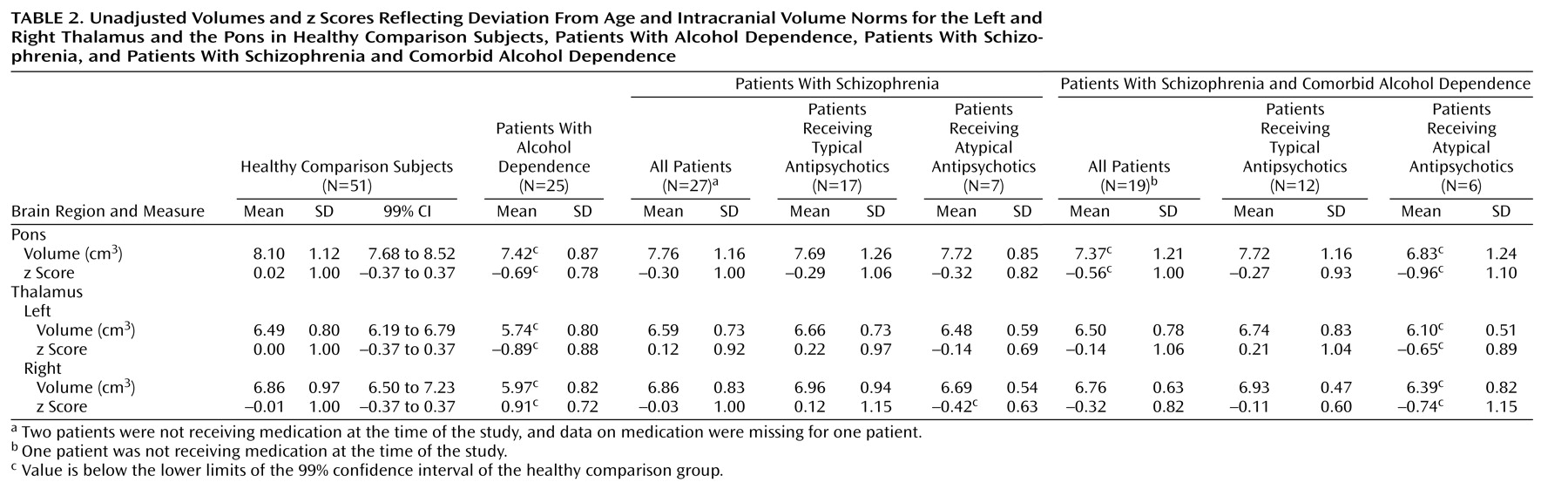
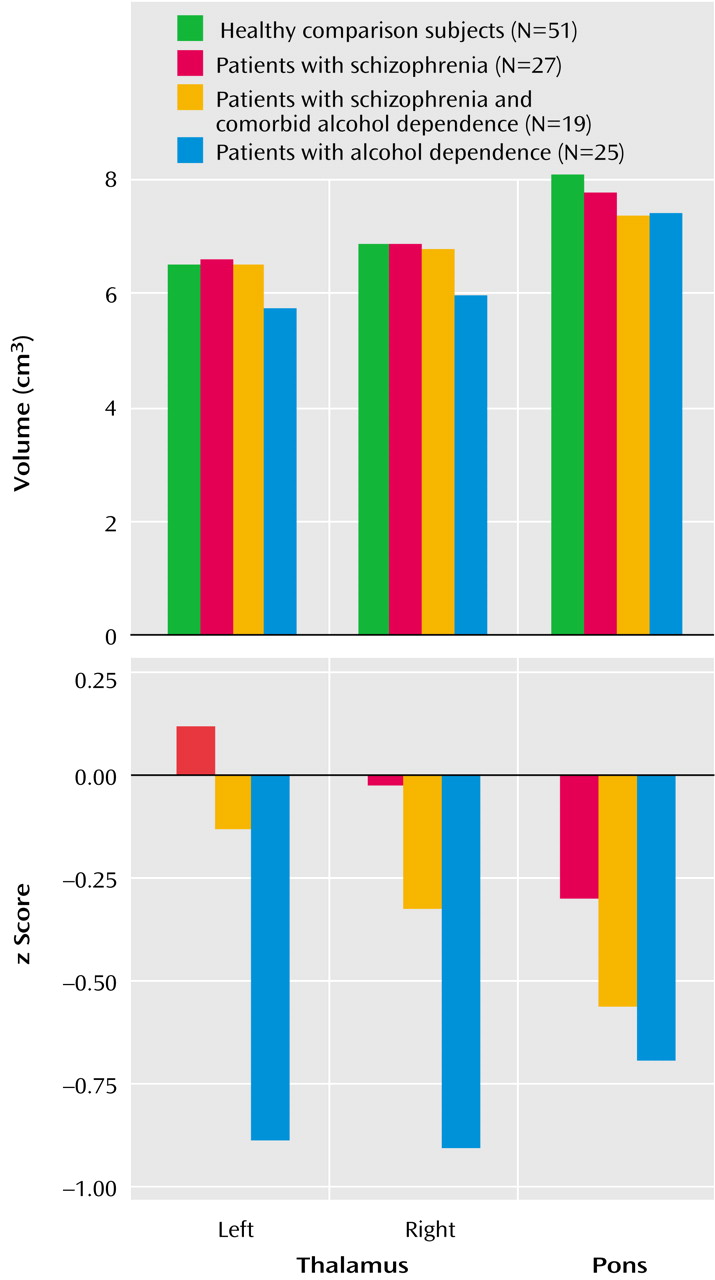
Means and standard deviations for z scores and unadjusted volumes of the left and right thalamus for each group are listed in
Table 2 and plotted in
Figure 2. Thalamic volume z scores showed a group effect (F=7.05, df=3, 118, p=0.0002) but no hemisphere or group-by-hemisphere interaction. Follow-up Scheffé tests indicated that the alcohol-dependent group had a deficit relative to the healthy comparison group (p=0.0008) and the schizophrenia group (p=0.002) but that neither the schizophrenia group nor the group with comorbid disorders had significant deficits. The ANCOVA, with unadjusted volumes and age as the covariates, yielded essentially the same results. The addition of lifetime alcohol use as a covariate in an ANCOVA assessing z scores for the bilateral thalamus eliminated the group effect.
Table 2 shows MRI values for the patients with schizophrenia and the patients with schizophrenia and comorbid alcohol dependence grouped by medication type. The contribution of antipsychotic medication type to thalamic volume in the schizophrenia patients as a group was assessed with a two-by-two group ANOVA for diagnosis (schizophrenia versus schizophrenia and comorbid alcohol dependence) and medication type (typical versus atypical). This analysis revealed an effect for medication type (F=4.21, df=1, 38, p<0.05), no diagnosis effect (F=0.85, df=1, 38, n.s.), and no interaction of medication type and diagnosis (F=0.23, df=1, 38, n.s.). The patients with schizophrenia, regardless of alcohol comorbidity, who were taking atypical medications had greater thalamic volume deficits than the patients who were taking typical medications.
Means and standard deviations for z scores and unadjusted volumes for the pons for each group are listed in
Table 2 and plotted in
Figure 2. A one-way ANOVA of the z scores for pons volume yielded a group effect (F=3.56, df=3, 118, p=0.02). Follow-up Scheffé tests identified significant differences between the alcohol-dependent and healthy comparison groups (p<0.04). In addition, the mean pons volume for both the alcohol-dependent group and the group with schizophrenia and comorbid alcohol dependence fell below the 99% confidence interval for the healthy comparison group. Unpaired t tests identified significant volume deficits in both the alcohol-dependent group (t=3.05, df=74, p=0.003) and the group with schizophrenia and comorbid alcohol dependence (t=2.12, df=68, p<0.05), relative to the healthy comparison group. The ANCOVA, with unadjusted volumes and intracranial volume as covariates, yielded essentially the same results. Group effects were eliminated when lifetime alcohol use was entered as a covariate in an ANCOVA assessing the z scores for the pons. The contribution of antipsychotic medication type to pontine volume in schizophrenia patients (with and without alcohol comorbidity) was assessed by a two-by-two group ANOVA for diagnosis (schizophrenia versus schizophrenia and comorbid alcohol dependence) and medication type (typical versus atypical). This analysis revealed no significant main effects or interactions.
Discussion
This study confirms the well-documented vulnerability of the thalamus and pons to chronic alcohol use and adds new information about the contribution of alcoholism to the volume of these structures in patients with schizophrenia. Although both the thalamus and the pons have been identified as structures involved in the pathophysiology of schizophrenia, our study found no significant deficits in gross volume in either structure in these particular patients with schizophrenia. Many other recent studies of medicated patients have also reported negative findings (e.g., references
21,
43–45), although a meta-analysis found a small but significant effect for thalamic volume deficits
(20). Our negative finding for the pons is consistent with other studies
(14).
Patients with schizophrenia who have comorbid alcoholism manifest deficits in the pons but not in the thalamus. It is possible that antipsychotic medication (particularly typical neuroleptics) can mitigate the effects of alcoholism in certain brain areas. We found that patients with schizophrenia who were taking atypical antipsychotic medications, regardless of whether they had comorbid alcoholism, showed greater thalamic volume deficits than those taking typical antipsychotic medications, an effect not found for the pons. Recent longitudinal in vivo imaging studies have shown an increase in basal ganglia volumes in patients taking typical antipsychotic medications and a decrease in patients taking atypical antipsychotic medications
(28,
29). However, the findings of studies of antipsychotic medication type and thalamic volume are less clear. One longitudinal study found that antipsychotic medication increased gross thalamus size, but this effect occurred with both typical and atypical antipsychotic medications in a dose-dependent manner
(25). Although a meta-analysis found no effect for medication status (medicated versus medication-naive), it did not consider medication type
(20). However, a recent PET study found a significantly lower D
2 binding index in the thalamus in patients treated with atypical antipsychotic medications than in those treated with typical antipsychotic medications
(30). The D
2 receptor binding associated with typical antipsychotic medications could normalize the thalamic volume deficits reported for neuroleptic-naive
(46) and first-episode schizophrenia patients
(47), or even increase the volume beyond the normal range. Unfortunately, the number of subjects in our study was relatively small, and we did not have medication dose and history data with which to more fully deconstruct the interactive effects of alcohol toxicity and neuroleptic enhancement on the thalamus. Further, this study included only men, thus precluding generalization of the findings to women.
In other studies involving this group of patients with schizophrenia and comorbid alcohol dependence, we have found deficits in the prefrontal cortex
(31) and gray matter of the cerebellar hemispheres and vermis
(32) that are greater than those observed in patients with either disease alone. The exacerbated prefrontal cortex deficits
(31) occurred despite far lower lifetime alcohol consumption by the patients with schizophrenia and comorbid alcohol dependence than by those with alcohol dependence alone. In our study of the cerebellum
(32), the schizophrenia group did not exhibit significant volume deficits, whereas deficits in the cerebellum were found in both the alcohol-dependent group and the group with schizophrenia and comorbid alcohol dependence, indicating that the volume deficits in the comorbidity group were attributable solely to the presence of alcohol dependence and that the comorbidity group appeared particularly vulnerable, as their lifetime exposure to alcohol was much less than that of the alcohol-dependent group. The effects of alcohol comorbidity in patients with schizophrenia were manifest differently on the structures examined in the current study. The thalamus was not smaller in volume in patients with schizophrenia, whether or not they had comorbid alcoholism, than in healthy comparison subjects. It is possible that the putative hypertrophy associated with typical neuroleptic use protected the thalamus from alcohol-related shrinkage, although this explanation is speculative and requires more rigorous investigation. By contrast, the volume of the pons in the comorbidity group was not afforded such protection and showed a level of volume deficit similar to that found in the alcohol-dependent group, who had consumed five times more alcohol in their lifetimes than the group with comorbid schizophrenia and alcohol dependence. Such pontine dysmorphology, along with cerebellar and prefrontal dysmorphology, puts schizophrenia patients with comorbid alcoholism at particular risk for impairment in cognitive and motor functions involving fronto-ponto-cerebellar circuitry
(1,
48–50).