Outcomes for Offspring of Parents With Schizophrenia and Related Disorders
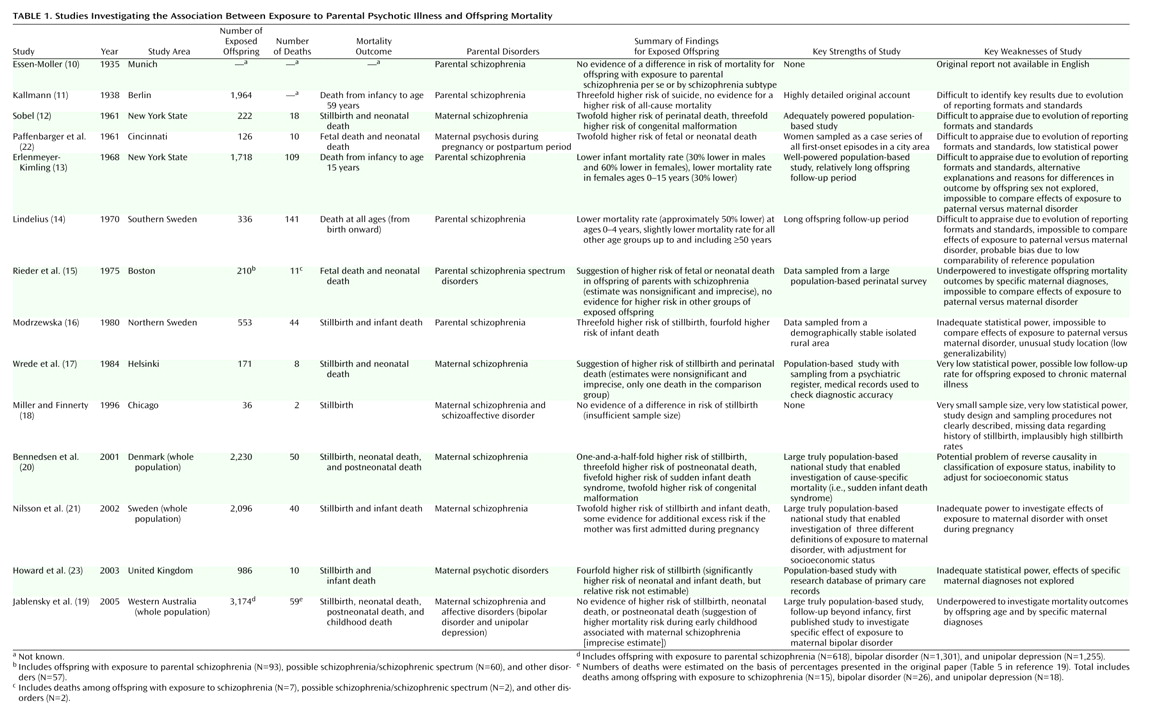
The earliest studies were conducted in Germany
(10,
11). The account of the first published study, by Essen-Moller, is available only in German
(10), although Kallmann
(11) briefly summarized Essen-Moller’s findings in English. In Essen-Moller’s study, probands were admitted with schizophrenia in Munich during 1904–1927, but the offspring sample size and follow-up period were not reported. No differences in mortality risk were found between exposed offspring overall and the general population, and there was no evidence for heterogeneity of effect by clinical form of schizophrenia, by maternal versus paternal disorder, or by timing of schizophrenia onset.
In Kallmann’s study
(11), outcomes for offspring (N=2,120) of schizophrenic probands admitted in Berlin during 1892–1902 were investigated. His findings, similar to those of Essen-Moller, were that child mortality risk was independent of schizophrenia per se, of the specific form of the disease, and of the sex of the affected parent. However, the risks of mortality because of suicide and tuberculosis were higher than expected, with four- to fivefold higher proportional mortality rates for those causes. The proportion of all offspring who died by suicide was also reported (relative risk*=3.3, 95% CI* not estimable).
Rosenthal
(26) postulated a “double dose” effect of inherited mortality risk in offspring of two schizophrenic parents. Testing this hypothesis in a secondary analysis of Kallmann’s data, he found no evidence of greater risk; 35% of these offspring (N=55) died before age 5 years, compared to 44.5% of the offspring with one affected parent. He also postulated that several gestational conditions could generate additional risk of poor outcome specific to maternal (versus paternal) schizophrenia. These conditions included medication toxicity, coma-inducing or convulsive therapy, inadequate diet, unusually high anxiety or excitatory states, and failure to exercise because of catatonia. He compared mortality to age 5 years in offspring of affected mothers (43.5%) versus fathers (39%) and again found insufficient evidence from Kallmann’s data to support this second hypothesis.
Sobel
(12) examined data for offspring (N=222) of women with schizophrenia who delivered at New York State mental hospitals during 1950–1958. He reported a perinatal mortality rate of 8.1% (10 stillbirths, eight neonatal deaths), compared with 3.6% in the U.S. general population (relative risk*=2.3, 95% CI* not estimable); the rate of congenital malformations was 3.2% (seven cases), compared with approximately 1% in the national population (relative risk*=3.2, 95% CI* not estimable). Sobel argued that the incidence of “deviant fetal growth” and subsequent mortality risk could be higher among offspring of mothers with some subtypes of schizophrenia, thereby highlighting the need for using large data sets for investigation of these rare outcomes. Sobel’s study was the first published study to empirically indicate higher than expected risk of all-cause mortality among exposed offspring.
However, in accordance with the “physiological advantage hypothesis” of Huxley et al.
(27), Erlenmeyer-Kimling
(13) postulated that offspring of people with schizophrenia would have
lower mortality risk. This hypothesis suggests the existence of genetic compensatory advantages that overcome selective disadvantages such as lower fertility and fecundity and render offspring of affected parents fitter than the general population. Examples of possible advantages given by Huxley et al. include “extreme resistance” to surgical or wound shock, burns, pain, arthritis, and many allergies and infections. To test the hypothesis, Erlenmeyer-Kimling calculated mortality rates in offspring (N=1,718) born during 1900–1959 to schizophrenic probands admitted to psychiatric hospitals in New York State and compared these rates with national population rates. The risk of infant mortality (i.e., deaths of live-born offspring during the first year of life) was lower among exposed offspring, a difference that was greater in girls (relative risk=0.4*, 95% CI* not estimable) than in boys (relative risk*=0.7). Relative risks were below 1.0 for both sexes in the 1–4-year age group, although this apparent protective effect was weak. For offspring ages 5–15 years, the relative risks were greater than 1.0 (relative risk*=1.3 in girls; relative risk*=1.2 in boys). When the data were pooled across all ages (0–15 years), there was no evidence of an effect for boys (relative risk*=1.0), but mortality risk was significantly lower among exposed girls (relative risk*=0.7). These results provided some support for the physiological advantage hypothesis, although no account was taken of possible environmental factors that could explain the lower risks found among exposed infants and young children. For example, institutionalization may have led to a lower prevalence of environmental hazards. Furthermore, no explanation for the heterogeneity of effect by offspring sex was offered.
Lindelius
(14) investigated mortality in offspring (N=336) of schizophrenic probands admitted during 1900–1910 in a demographically stable area of rural Sweden, with 93% follow-up to 1960 achieved by using parish registers. Expected numbers of deaths per age-sex stratum were estimated in reference to national population rates and were compared with observed numbers. All-cause mortality was significantly lower than expected in exposed offspring to age 4 years, but, as the author suggested, this finding may have been a statistical artifact that occurred because the national reference rates included higher-risk urban populations. For all other age-sex strata, up to and through adulthood, all-cause mortality was slightly lower than expected in the exposed offspring. There was some indication of a higher mortality rate because of tuberculosis and a lower risk of suicide, although these cause-specific analyses lacked statistical power.
Using data from a large perinatal survey established in 1959 in Boston, Rieder et al.
(15) investigated fetal and neonatal mortality in three parental diagnostic subgroups: 1) schizophrenia (N=93), 2) possible schizophrenia/schizophrenic spectrum (N=60), and 3) other disorders (neuroses, manic depression, “character disorder,” alcohol addiction with psychosis) (N=57). Comparison subjects without a psychiatric history were selected and matched with the psychiatric disorder subjects for ethnicity, socioeconomic status, maternal age, parity, and offspring multiplicity. There was some suggestion of a higher than expected risk for exposure group 1 only (relative risk*=2.0, 95% CI*=0.7–5.5), although the wide confidence interval indicates imprecision and nonsignificance.
Modrzewska
(16) examined data for offspring of parents with schizophrenia (N=553) and comparison subjects with unaffected parents (N=624) in an isolated area of northern Sweden. All probands were born in the area during 1829–1960. Higher than expected risks of stillbirth (relative risk*=3.1, 95% CI*=1.3–7.2) and infant mortality (relative risk*=4.1, 95% CI*=1.8–9.5) were observed in exposed offspring. Low numbers precluded assessment of mortality risk in offspring with two affected parents (N=39), and cause of death could not be investigated because of poor documentation. Furthermore, a high prevalence of consanguineous marriage and intensive inbreeding affected the study’s external validity.
Wrede et al.
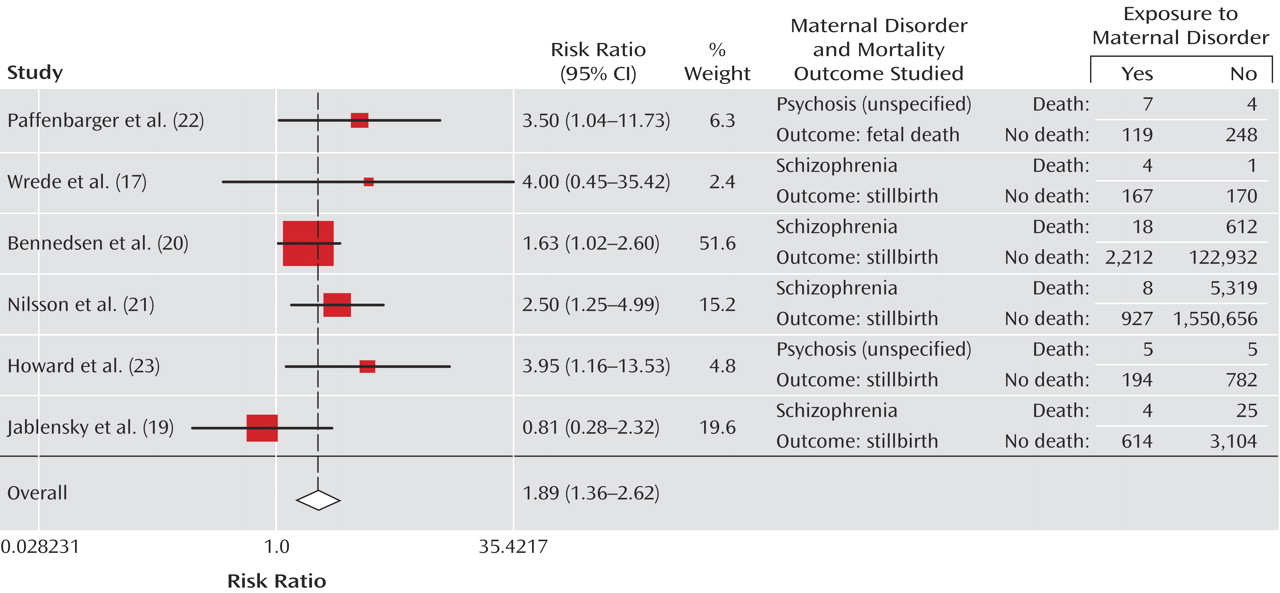
(17) used a psychiatric register to investigate outcomes for offspring born to women with schizophrenia in Helsinki, Finland, during 1960–1964. Medical records were examined to assess diagnostic accuracy, and exposure status in terms of severity of maternal disorder was classified as “chronic” (permanent social deficit: N=54) or “mild” (1–3 admissions: N=117). Comparison subjects were randomly sampled from a population register (N=171). Nine perinatal deaths occurred, all but one among the mild exposure group (four stillbirths, four neonatal deaths) and the other among the comparison subjects (a stillbirth). The lack of deaths in the chronic exposure group was unexpected but could have occurred by chance or because of the high rate of loss to follow-up in that group. For all exposed offspring combined (N=171), the relative risks of stillbirth (relative risk*=4.0, 95% CI*=0.5–35.4) and perinatal mortality (relative risk*=8.0, 95% CI*=1.0–63.3) were higher than expected, but the confidence intervals for these estimates are very wide and imprecise.
Miller and Finnerty
(18) recruited women meeting the Research Diagnostic Criteria for schizophrenia or schizoaffective disorder (N=44) in Chicago during 1993–1995 and a comparison group (N=50) of women without major mental illness who were matched with the schizophrenic subjects for age, ethnicity, and socioeconomic status. Subjects were questioned about their sexuality, pregnancies, and child rearing, but there were missing data concerning history of stillbirth (data on history of stillbirth were available for 36 affected women and 46 comparison subjects). Comparable stillbirth rates were reported in the two groups (two cases in the affected group, three in the comparison group), but this evidence is weak because of the small sample sizes and because the baseline risk of stillbirth reported in the comparison group (65 per 1,000) was approximately nine times higher than the contemporary national rate
(28).
Jablensky et al.
(19,
29) linked population and psychiatric registers to investigate mortality among all births occurring in Western Australia during 1980–1992 to mothers with schizophrenia (N=618), bipolar disorder (N=1,301), and unipolar depression (N=1,255). Comparison offspring (N=3,129) of mothers without a psychiatric history were randomly selected from the general population. There was no evidence of higher than expected perinatal or infant mortality among offspring exposed to maternal schizophrenia (stillbirth: odds ratio=0.8, 95% CI=0.3–2.3; neonatal death: odds ratio=1.0, 95% CI=0.3–3.0; postneonatal death: odds ratio=1.1, 95% CI=0.2–5.1). There was a suggestion that these offspring were at increased risk during early childhood (odds ratio=2.2, 95% CI=0.7–7.3), although this estimate lacked statistical significance and precision. No evidence was found for higher risk of mortality at any age among offspring exposed to maternal bipolar disorder (stillbirth: odds ratio=0.9, 95% CI=0.4–1.9; neonatal death: odds ratio=0.8, 95% CI=0.3–2.0; postneonatal death: odds ratio=1.2, 95% CI=0.5–3.0; early childhood death: odds ratio=1.3, 95% CI=0.4–4.6). However, as
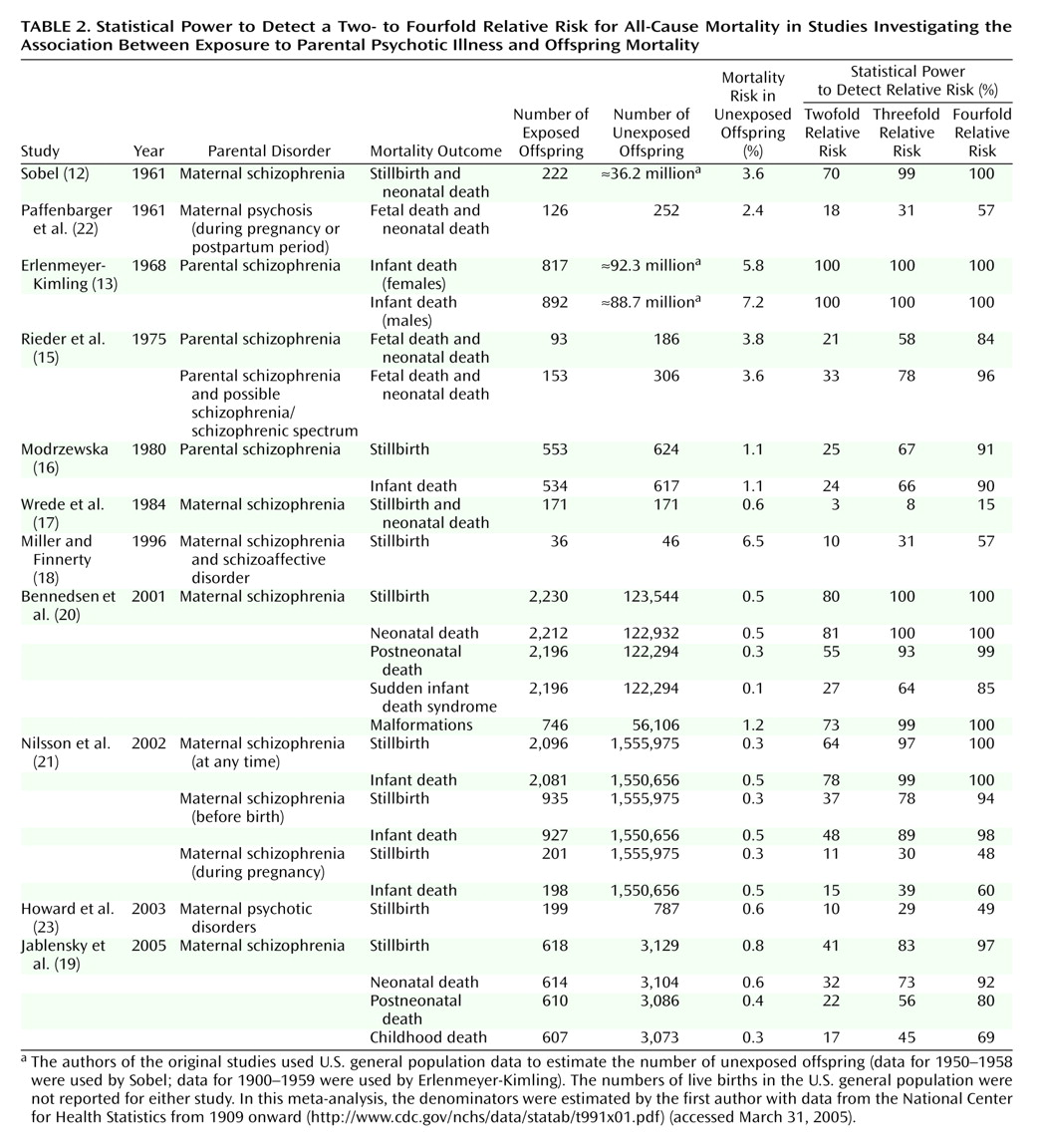
Table 2 shows, these data lacked power for examining offspring mortality in relation to specific maternal disorders, as the primary aim of the study was to investigate more commonly occurring outcomes during pregnancy, delivery, and the neonatal period.
Two large Scandinavian registry-based studies
(20,
21) have provided the best available evidence to date. Bennedsen et al.
(20) investigated all single births (N=2,230) to mothers admitted with schizophrenia to public psychiatric facilities in Denmark during 1969–1993; the comparison group consisted of subjects randomly sampled from the general population (N=123,544). As there were no private psychiatric inpatient facilities in Denmark, this investigation was truly a population-based study. Relative risks were adjusted for year of birth, offspring sex, maternal age, and parity, although no adjustment was made for socioeconomic status, smoking status, substance abuse, and psychoactive treatment. Higher than expected relative risks in exposed offspring were observed for stillbirth (relative risk=1.6, 95% CI=1.0–2.6; adjusted relative risk=1.5, 95% CI=0.9–2.4), neonatal death (relative risk=1.4, 95% CI=0.9–2.3; adjusted relative risk=1.3, 95% CI=0.8–2.1), and congenital malformation (relative risk=1.8, 95% CI=1.1–2.9; adjusted relative risk=1.7, 95% CI=1.0–2.8), with larger effects observed for postneonatal death (relative risk=2.8, 95% CI=1.7–4.5; adjusted relative risk=2.8, 95% CI=1.7–4.6) and sudden infant death syndrome (relative risk=4.7, 95% CI=2.6–8.8; adjusted relative risk=5.2, 95% CI=2.8–9.7). However, offspring were classified as being “exposed” even if the first maternal admission for schizophrenia occurred after the offspring death, an event that could be a contributory factor in maternal disease onset, thereby introducing a potential reverse causation bias
(30).
Nilsson et al.
(21) conducted a similar investigation of all single births during 1983–1997 to mothers admitted with schizophrenia in Sweden during 1977–1997. Comparison subjects were offspring of unaffected mothers in the general population (N=1.56 million). Potential problems of reverse causality were addressed by estimating relative risks for three exposed subgroups: 1) offspring of mothers who first received the diagnosis at any time during the study period (N=2,096), 2) offspring of mothers who first received the diagnosis before birth of the offspring (N=935), and 3) offspring of mothers who first received the diagnosis during pregnancy (N=201), with groups 2 and 3 providing the more stringent definitions of exposure. Odds ratios were estimated with adjustment for maternal age, smoking, education, country of birth, marital status, and the presence of pregnancy-induced hypertension. Offspring whose mothers received the diagnosis of schizophrenia before the offspring’s birth were found to be at higher than expected risk for stillbirth (odds ratio=2.5, 95% CI=1.3–5.1; adjusted odds ratio=1.7, 95% CI=0.9–3.5), and they also had a higher—although nonsignificantly higher—risk for infant death (odds ratio=1.8, 95% CI=0.9–3.6; adjusted odds ratio=1.4, 95% CI=0.7–2.8). There was some indication of even greater risk if the mother’s first admission occurred during pregnancy (stillbirth: odds ratio=4.4, 95% CI=1.4–13.8; adjusted odds ratio=2.5, 95% CI=0.8–7.9) (infant death: odds ratio=3.1, 95% CI=1.0–9.8; adjusted odds ratio=2.1, 95% CI=0.7–6.6), although these subgroup analyses lack statistical precision.