Subjects
The subjects were 119 homosexual men ages 21 to 58 years in the asymptomatic and early symptomatic clinical stages of HIV-1 infection at entry into a prospective natural history study of HIV-1 infection. Subjects were enrolled between 1987 and 1990, with evaluation at 6-month intervals until their death or until May 1991. Subjects were recruited from a variety of sources, including the University of Miami HIV-1 research screening clinics, community physician referrals, and AIDS-related community-based organizations. Informed consent was obtained after the nature of the procedures had been fully explained.
Exclusion criteria were 1) signs (other than persistent generalized lymphadenopathy) or symptoms referable to HIV-1; 2) CD4 cell count >700 cells/mm3; 3) taking of antiretroviral medications (such as zidovudine, didanosine (ddI), zalcitabine (ddC), stavudine (d4T), or lamivudine (3TC)), immunomodulators, or participation in an HIV-related drug trial at study entry; 4) history of excessive alcohol or substance use; 5) history of severe head trauma associated with loss of consciousness; 6) evidence of HIV-associated CNS disease; or 7) history of a major psychiatric disorder (such as schizophrenia or other psychosis or bipolar affective disorder) prior to HIV-1 infection. A history of major depressive disorder did not warrant exclusion. Although a history of injection drug use was not explicitly employed as an exclusion criterion, no subjects with this history were enrolled (as would be expected per the exclusion for history of excessive alcohol and substance use).
There were 14 deaths over the follow-up period among the 119 HIV-1–seropositive subjects.
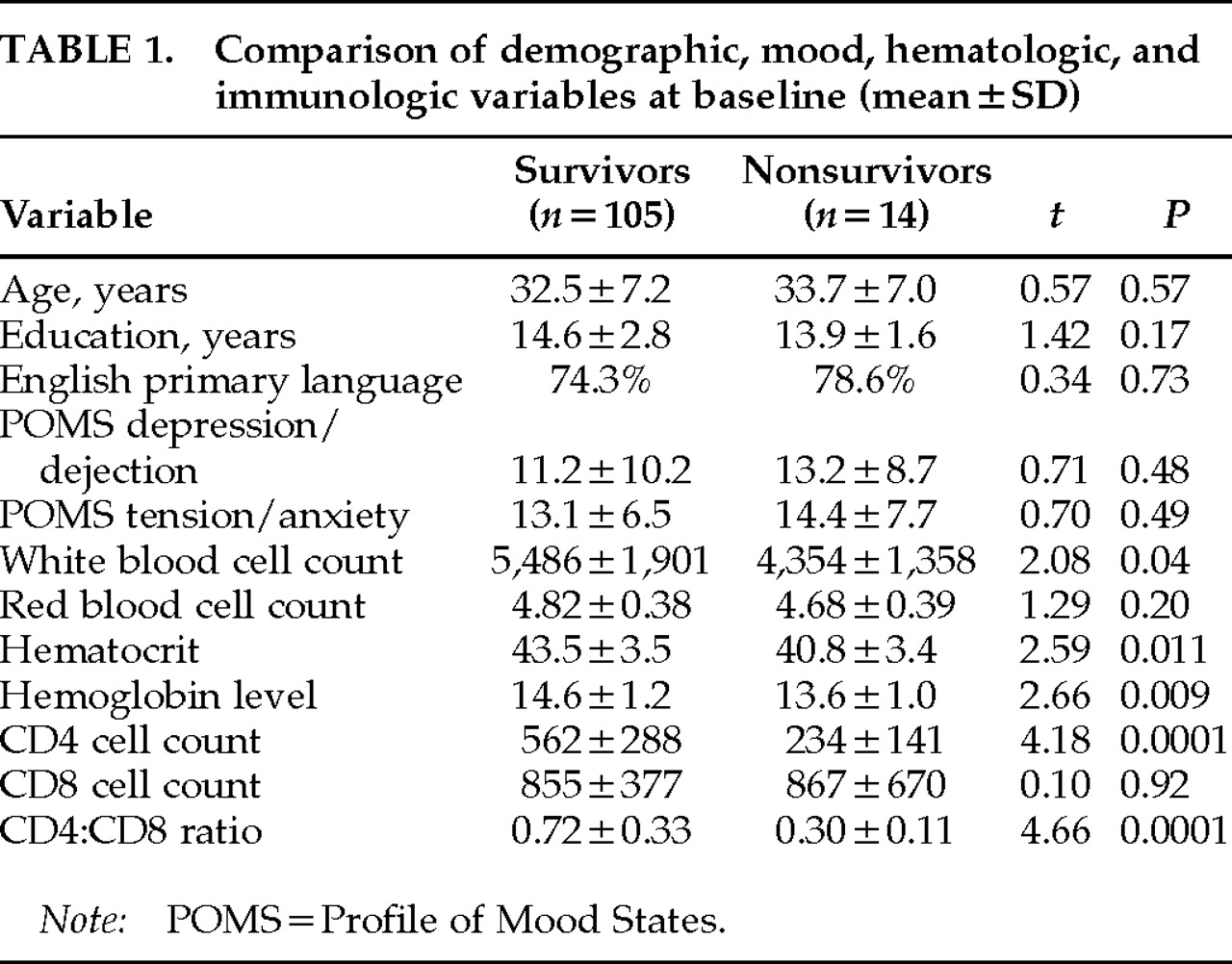
Table 1 lists the characteristics at study entry for the 105 survivors and 14 nonsurvivors. The survivors were followed for a median 24 months (mean±SD=23.4±14.5; subsample distribution by length of follow-up: <6 months, 13.5%; at least 6 months, 6.7%; 1 year, 9.6%; 1.5 years, 12.5%; 2 years, 11.6%; 2.5 years, 12.5%; 3 years, 16.3%; 3.5 years, 17.3%) and the nonsurvivors for a median 18 months (mean 18.4±8.6; subsample distribution by length of follow-up: <6 months, 7.1%; at least 6 months, 7.1%; 1 year, 14.3%; 1.5 years, 35.8%; 2 years, 21.5%; 2.5 years, 7.1%; 3 years, 7.1%; 3.5 years, 0%). All of the deaths were HIV-1 related. The two groups were similar with respect to age, education, and primary language. They were also similar in clinical stage, as measured by the 1986
18 or the 1993
19 Centers for Disease Control and Prevention (CDC) clinical staging systems. On average, the subjects were in their early 30s and had 14 to 15 years of education. Seventy-one percent of the nonsurvivors and 75% of the survivors spoke English as their primary language. The remaining subjects within each group were second- or third-generation Cuban Americans who were bilingual but spoke fluent English. The groups were within normal limits and did not differ on measures of anxious and depressive mood symptoms as measured by the Profile of Mood States tension-anxiety and depression-dejection subscales.
20On initial evaluation, 75% of both the survivors and the nonsurvivors were asymptomatic or had persistent generalized lymphadenopathy (PGL) and were at the 1986 CDC stages II or III (
n=96; 1993 CDC clinical stage A—asymptomatic). The remaining 25% in both groups were at the 1986 CDC stage IVA (constitutional symptoms), B (neurological symptoms—all of which were peripheral neuropathies and were non–AIDS-defining) or C-2 (non–AIDS-defining opportunistic infections such as thrush and oral hairy leukoplakia; 1993 CDC clinical stage B [early symptomatic],
n=22). No subjects were at 1986 stage IVC-1 (AIDS-defining opportunistic infection) or IVD (tumor) at entry. That is, none were at 1993 CDC clinical stage C (late symptomatic—i.e., clinically defined AIDS). 1993 CDC staging combined clinical staging (A, B, and C, as above) with a numbered immunological staging axis: 1≥500 cells/mm
3 (
n=54; A1=48, B1=6, C1=0); 2=200–499 cells/mm
3 (
n=52; A2=40, B2=12, C2=0); 3≤200 cells/mm
3 (
n=12; A3=8, B3=4, C3=0). Although the 1993 AIDS case definition
19 included any individuals with a CD4 cell count <200 cells/mm
3 as an AIDS case regardless of clinical stage, it was nevertheless decided to use CD4 cell count here as a continuous rather than a stratified covariate in the analysis to maximize the information available from this control (see statistical methods). Compared with the survivors (mean=562 cells/mm
3), as expected, the nonsurvivors did have a significantly lower CD4 lymphocyte count (mean=234 cells/mm
3;
t=4.18,
P=0.0001) and a lower CD4:CD8 ratio (survivors, mean=0.72, vs. nonsurvivors,mean=0.30;
t=4.66,
P=0.0001; see statistical methods section for the method of controlling this significant difference in the analysis of mortality). They also had significantly lower white blood cell counts, hematocrit levels, and hemoglobin levels than the survivors (see
Table 1).
Although none of the subjects were taking zidovudine when enrolled, during the longitudinal follow-up period 32.6% of the survivors and 71.4% of the nonsurvivors had started zidovudine therapy. Other antiretroviral drugs were largely unavailable throughout the trial period. ddI was started by only 3 subjects (2 in combination with zidovudine), ddC by 1 subject (in combination with zidovudine), and d4T and 3TC by no subjects during the follow-up period. The protease inhibitors were not available during the study period. Prophylactic medications for Pneumocystis carinii pneumonia (such as trimethoprim/sulfamethoxazole, aerosolized pentamidine, and dapsone), were started by 22 subjects; for cryptococcal meningitis, by 2 subjects (fluconazole); and for tuberculosis, by 3 subjects (isoniazid; INH). No subjects started rifabutin for Mycobacterium avium intracellulare during the study period.
Laboratory and Clinical Health Measures
A measure of immunosuppression (CD4 lymphocyte count), oxygen-carrying capacity (venous hemoglobin level), and clinical staging (CDC 1986
18; CDC 1993
19) were used as covariates because of their association with disease progression generally and cognitive impairment specifically. Nutritional status (vitamin B
6 and B
12 levels) was also examined as a covariate in our analyses because of its effect on cognition in HIV-1 infection.
28–30 After a 6-hour fast, a total of 50 ml of venous blood was drawn between 8:00 and 10:00
a.m. for immune and nutritional assays. HIV-1–positive serostatus was defined by a doubly reactive enzyme-linked immunosorbent assay (ELISA; Abbott, Chicago, IL) confirmed by Western blot (Hillcrest Laboratories; Cypress, CA). The CD4 cell count was determined by direct immunofluorescence, using a whole-blood staining technique. Briefly, we used a two-color protocol, using an Epics Elite flow cytometer (Coulter Corp, Hialeah, FL), as described by Fletcher and co-workers
31,32 The complete blood count (CBC), determined by Cell Dyne 1500 (Sequoia-Turner, Mountain View, CA) with a differential count, provided a measure of the total white blood cell count, the total lymphocyte count, and a hemoglobin level. Percentage of positively stained cells for the CD4 marker was determined by use of Quadstat software (Coulter Epics). Estimates of absolute counts of cells positive for the CD4 surface marker were determined by multiplying peripheral lymphocyte count by percentage of positive cells for the CD4 surface marker.
Plasma pyridoxine (vitamin B
6) levels were determined by an erythrocyte transaminase assay,
33 and plasma cobalamin (vitamin B
12) levels were established by a radioisotope dilution assay.
34Data Analysis
The Cox proportional hazards model
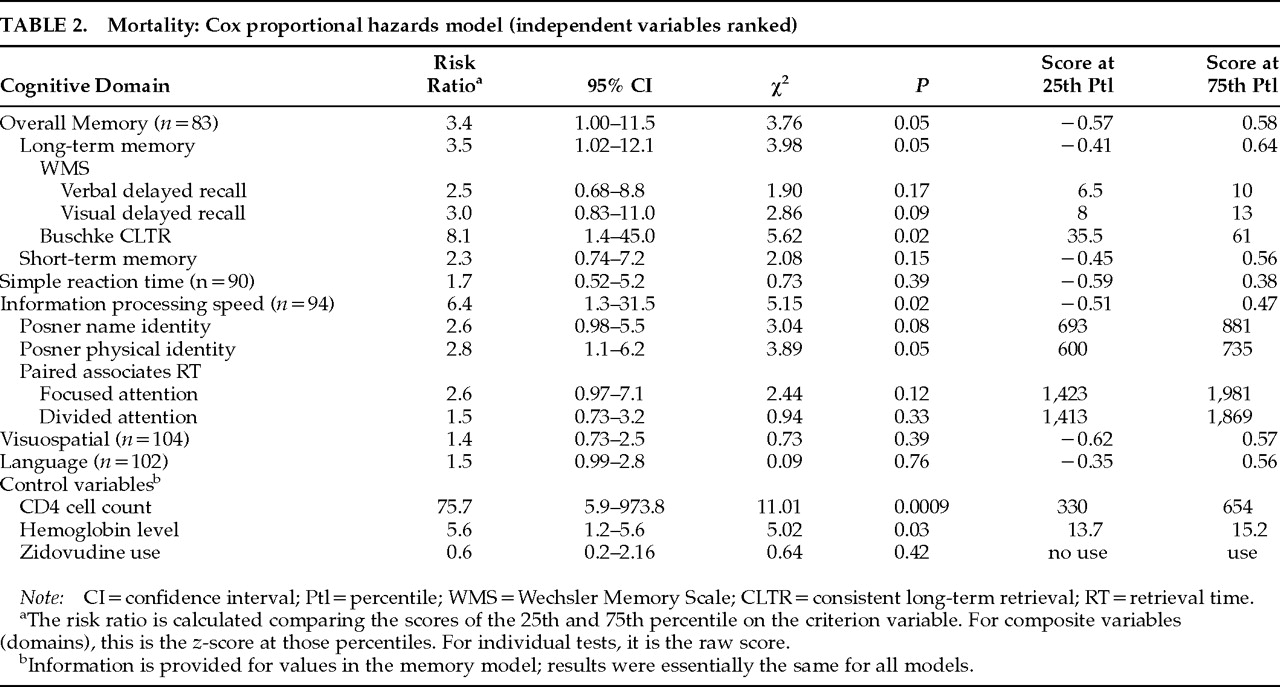
35 was used to determine whether mortality between the fall of 1987 and May 31, 1991 (study termination), could be predicted by baseline neuropsychological test performance level (as opposed to impairment versus nonimpairment). This model incorporated controls for the CD4 cell count and other known predictors of mortality in HIV-1 infection. Hazard models appropriately include all available information about subject survival and control for censoring by differential length of follow-up.
In evaluating the value of the cognitive and other variables in predicting mortality, we estimated the Cox proportional hazard models by using maximum likelihood methods with the Statistical Analysis System (SAS) software.
36 The risk ratio (RR) was calculated to compare the scores at the 25th and 75th percentiles within this sample. Preliminary analyses were performed to identify potential confounds, which included sociodemographic variables, depressive and anxious mood symptoms from the POMS,
20 an immunologic progression marker (CD4 cell count), red blood cell oxygen-carrying capacity (hemoglobin level), zidovudine use, and CDC staging of HIV-1 infection as established by both the 1986
18 and the 1993
19 CDC clinical staging systems. (1993 CDC
19 immunological staging by the CD4 lymphocyte count was not included here because the latter was retained as a continuous independent control variable to allow for the maximum range of variation.) To avoid type II error in identifying confounds, variables reaching the
P≤0.10 level were included in the final analyses. CD4 cell count and hemoglobin level independently maintained
P-values ≤0.10 with mortality. In addition, zidovudine use was significantly and positively associated with mortality.
The cognitive test scores were grouped by cognitive domains determined a priori and verified by using Cronbach's α for internal consistencies, which were in the 0.77 to 0.88 range. We transformed the raw scores for each test to z-scores based on the means and standard deviations for this sample of HIV-1–seropositive subjects and obtained an average z-score for each person for each of the five cognitive domains. Examination of the final z-scores revealed kurtosis due to an increased frequency of low neuropsychological performance levels—to be expected in a sample of HIV-1–infected individuals. Hence, the z-scores were ranked in the statistical analyses. A separate Cox analytical model was estimated for each cognitive domain. The n in each analysis varies because of missing data. The composite scores for each of the cognitive domains showing a significant relationship with mortality were then decomposed, and the constituent scales (cognitive tests) were used as predictor variables in post hoc examinations of the mortality relationship. To examine the effect of impairment in each specific domain, the risk ratio comparing the score at the cutoff for the 25th percentile with that for the 75th percentile and its associated confidence interval was calculated, based on the model estimates derived from the data involving the total sample (N=119).
In our statistical models, mortality as of May 31, 1991 (study termination), was the dependent variable. For each cognitive domain, in addition to the baseline cognitive measure for that domain, the two significant laboratory predictors (CD4 lymphocyte count and hemoglobin level), along with zidovudine treatment, were included in the model. Therefore, the relationships of the cognitive variables to mortality are independent of the levels of these control variables. Controlling for the CD4 lymphocyte count and the hemoglobin level reduced the effect size for zidovudine treatment on mortality.