DISCUSSION
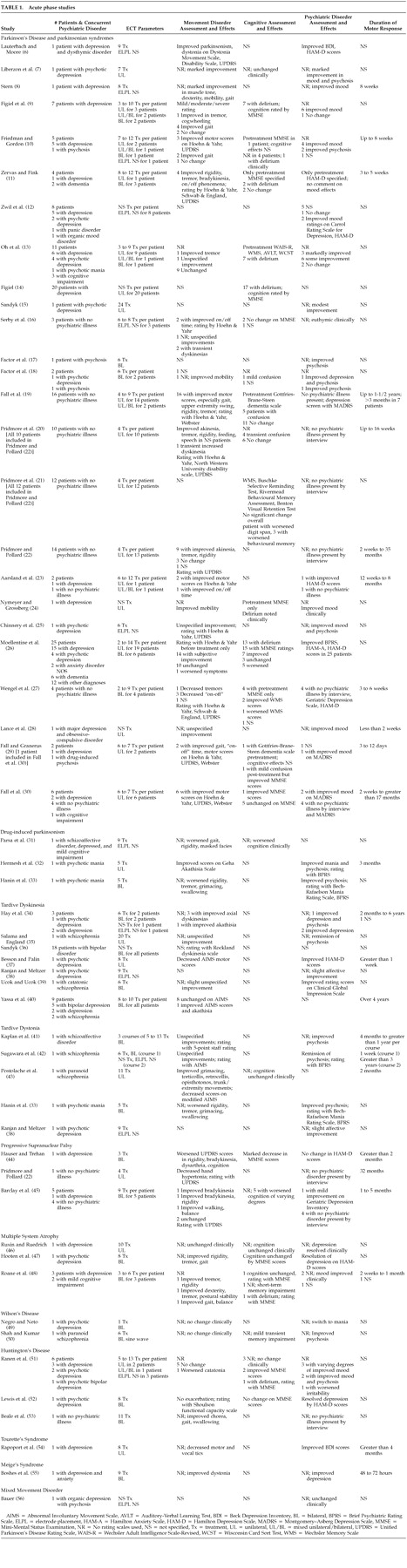
ECT remains an effective treatment for a variety of psychiatric illnesses, particularly depression. These disorders occur frequently in patients with movement disorders, and ECT does appear effective for alleviating mood symptoms in this population. Due to the uncontrolled nature of these reports, it is not known whether comorbid movement disorders adversely affect the response of depression to ECT. ECT may also be effective in treatment of drug-induced psychotic states in PD.
ECT also shows some promise in treating motor manifestations of movement disorders, though these data must be interpreted cautiously. The majority of reports address ECT and Parkinson's disease, with many of them being favorable. Based on these reports, some ECT experts have advocated general usage of ECT in Parkinson's disease. One editorial recommended “a therapeutic trial of ECT for every patient with intractable or drug-resistant Parkinson's disease, particularly those with the on-off syndrome.”
60 Another editorial
61 advocated a therapeutic trial of ECT before such procedures as neuronal transplantation were considered. Experts in PD have been less encouraging,
62 and recent neurological guidelines for management fail to mention this as a treatment option for motor symptoms.
63 Experts in PD note publication bias to report only positive results and the small number of well documented studies. Furthermore, they raise concerns about the biological basis of response, induction of confusional states, and the need for maintenance treatment for continued response. These criticisms must be addressed if one advocates ECT as a treatment for the motoric symptoms of PD.
The concern about possible publication bias overemphasizing positive results is well justified, as most of the data concerning ECT and PD consist of case reports. Practitioners are less likely to publish negative results or to pursue further study of a treatment modality when initial results are not encouraging.
62 Retrospective reviews and open prospective trials may provide a better view of response rates by including nonresponders, and these studies have been positive. Although data concerning ECT and PD do not rely solely on case reports, the lack of further controlled studies is a serious limitation.
Several mechanisms of action have been proposed for ECT in PD. Some researchers have argued that the improvement in motor symptoms is due to resolved depression, with reduced psychomotor retardation.
64 Our review, as well as the previous review,
3 argues against this. A substantial number of patients without any discernible psychiatric disease have had improvement in motor symptoms. Also, as previously noted by Young,
65 one report
8 describes a dissociation between improvement in mood and motor symptoms, with the latter occurring earlier in treatment. Other authors have argued that improvement is due to disruption of the blood-brain barrier that is known to occur with ECT treatment.
66 This would allow increased levels of L-dopa to enter the brain, potentially alleviating motor symptoms even though the oral drug dosage remained constant.
64 The above reports, documenting patients with improvement who were not on L-dopa, indicate other mechanisms are at least partly responsible. The most commonly accepted explanation is a neurochemical one. Animal and human studies have suggested that ECT enhances dopaminergic transmission. Since serum and CSF levels of dopamine metabolites are not consistent in studies, it is thought that the effects may be enhancement at the receptor or postreceptor level.
67 However, some of the evidence for this comes from the improvement in PD following ECT.
67 Fochtmann and colleagues
68 have shown that electroconvulsive shock in rats increases dopamine type 1 receptor binding in the substantia nigra. They hypothesize that this may be associated with other changes in the dopamine system, including upregulation of dopamine type 2 receptors in the striatum. The latter may be the mechanism by which ECT exerts an antiparkinsonian effect. Only two studies
19,30 during this review period addressed this issue. These confirmed an increase in cerebrospinal fluid dopamine metabolites after ECT, indicating increased dopamine neurotransmission. These changes did not appear to be mediated by alterations in the dopamine transporter protein, indicating other mechanisms for the efficacy of ECT.
ECT has significant effects on both noradrenergic and serotonergic transmission.
67 Although these changes likely play a role in the alleviation of depression, their role in the motor changes are unclear.
69,70 Finally, animal studies have shown upregulation of the GABAergic system, although this has not been demonstrated in humans.
67 Changes in the GABAergic system may influence the abnormal movements in PD, although evidence is still controversial.
71Concerns about delirium or confusional states are also justified but should not prevent use of ECT. Continued delirium or cognitive dysfunction (defined in most reports as lasting more than 24 hours) is thought to be a rare side effect of ECT. One study
72 indicated that post-ECT delirium occurred in approximately 10% of patients at some time during treatment. Given that some authors have postulated a subcortical mechanism for delirium,
73 it is not surprising that patients with impaired subcortical function such as PD would be more vulnerable. Although precise incidence figures are not ascertainable due to the uncontrolled nature of many reports, the above figures for delirium are concerning. The prolonged periods of delirium in many patients adds further weight to this. In vulnerable patients, several strategies can minimize post-ECT delirium.
74 These include twice-weekly ECT instead of thrice weekly, use of unilateral ECT, using the minimally effective electrical dose, using brief pulse stimulation, and avoiding potentially deliriogenic medications. For PD patients undergoing ECT, reducing the dosage of L-dopa has been effective for some, but not all, patients who developed delirium. Current recommendations to reduce L-dopa dosages by half
4 should be followed unless clinical symptoms of PD are too severe to allow reductions. Right unilateral electrode placement should be used initially.
4 Previous recommendations
3 against unilateral treatment due to limited experience are no longer justified, given the number of patients benefitting with this modality. Strong consideration should be given to twice-weekly treatments. This is clearly an area needing further research, both in depressed and nondepressed PD patients, to determine the overall incidence of delirium and the effectiveness of these strategies to minimize it.
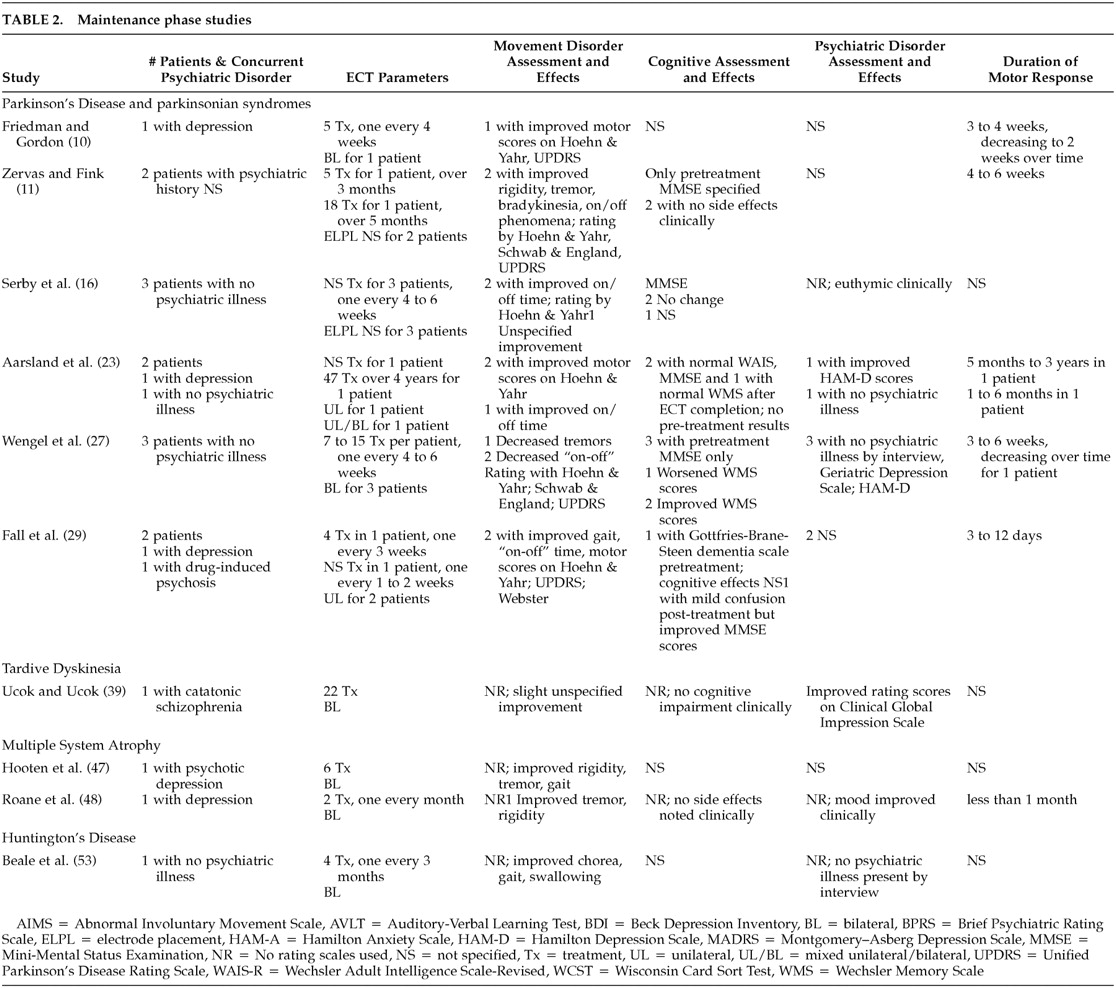
Several issues also remain in the use of maintenance ECT for PD. With only 15 patients undergoing such treatment, it is difficult to generalize results. However, it is clear that some patients have continued benefit in motor symptoms with maintenance ECT, both with and without comorbid affective disorders. Other patients have required progressively shorter intervals between treatments until the frequency of ECT was no longer feasible. The reasons for this phenomenon are unknown at present. The optimal interval between ECT treatments is also not known. Previous guidelines
4 suggest that this intertreatment interval should be initially short, such as once a week, and progressively increased to the longest interval that sustains the improvement. This approach is reasonable. However, with some patients having prolonged symptom-free intervals after the initial ECT series (up to 35 months has been reported
22), this approach may expose a small number of patients to a higher number of ECT treatments than is necessary.
Continued investigation of ECT for the motoric symptoms of PD is indeed warranted. A recent report
75 describes the long-term outcome of pallidotomy for PD, with sustained improvements in off-period contralateral signs and in on-period dyskinesias noted. In this study, however, one-half of the original cohort was excluded for various reasons. These patients had similar baseline characteristics but worse response to surgery at 6 months compared to the study group, potentially biasing the latter toward positive outcome. Another report
76 described the outcome for transplantation of embryonic dopamine neurons for severe PD. Small amounts of motor improvement, measured using standardized scales, was noted in some younger patients but no improvement in older patients. Thus, the treatment for advanced or refractory PD remains unsatisfactory, and other novel treatments may yet play a role. Furthermore, patients may prefer the option of ECT over other treatment modalities for refractory symptoms. Pridmore and Pollard
22 reported that more than half of their patients who could give an opinion would consider a future course of ECT. Another survey
77 of PD patients showed that 28% would consider ECT in their current state and another 32% would consider it if their condition worsened.
Drug-induced movement disorders also appear responsive to ECT, although data are mostly case reports and case series. ECT is thought to reduce drug-induced parkinsonism by upregulating postsynaptic dopamine receptors, similar to its effects in idiopathic PD. A previous review
3 and an additional case have confirmed its utility. Another case series
57 has suggested that ECT may prevent drug-induced parkinsonism in patients who have not developed this complication. However, two patients in the case series had worsened symptoms, which had not previously been reported. The reasons for this are unclear, but caution is warranted in such patients undergoing ECT. With the widespread usage of atypical antipsychotics, which have lower rates of parkinsonism, such issues may not be particularly pressing.
Tardive dyskinesia and dystonia appear responsive to ECT, although overall results are mixed. ECT is thought to prevent supersensitization of postsynaptic dopamine receptors that play a role in development of tardive states.
78 Such effects only occurred with concurrent neuroleptic administration, which may account for the efficacy of ECT in both hypo- and hyperdopaminergic states. Some patients improve, many remain unchanged, and a small number experience worsening of symptoms. The single prospective study
40 found improvement in one patient, with eight remaining unchanged. The reason as to why some patients experience worsening of symptoms is unclear. However, Hay and colleagues
34 note the difficulties in attributing worsened TD to ECT, as many patients concurrently have alterations in their medication regimen. These alterations may cause withdrawal dyskinesias that spontaneously remit with time. If medications are discontinued in order to begin administration of ECT, these withdrawal dyskinesias may be erroneously attributed to ECT. Similar issues may exist for tardive dystonia, as the single case with worsened symptoms also involved the discontinuation of medications prior to ECT.
33 However, this patient also developed aspiration from dysphagia and dystonia, illustrating that these effects cannot be completely ignored even if transient. Finally, three reports
79–81 have documented lower prevalence of TD in patients who received ECT. Given that treatments for tardive dyskinesia are generally disappointing,
82 the possible protective role of ECT with tardive dyskinesia deserves further study.
There is much less data regarding movement disorders other than PD, consisting only of case reports and case series. Disorders with parkinsonian or dystonic symptoms, such as PSP and MSA, have the most reports of improvement. Since the effects of ECT on dopamine are primarily postsynaptic,
67 improvement might be expected even if L-dopa administration is not useful. As previously noted, ECT also affects a variety of other neurotransmitters, such as gamma-aminobutyric acid (GABA). Movement disorders involving these transmitters, such as HD, are largely unaffected by ECT, though occasional patients may benefit. Resolution of mood symptoms may also contribute to improvement.
54 These patients should not be denied ECT based solely on the above reports noting delirium and worsening of the movement disorder. However, these reports should be considered in the risk/benefit ratio and the decision to prescribe ECT. Such patients should be treated as clinically indicated by their psychiatric condition. The risk of delirium may be minimized using the above strategies.
Overall, a substantial amount of data support the positive effects of ECT on the motor symptoms of Parkinson's disease. However, almost all of these data are from case reports or case series, with only one randomized, controlled trial in the literature. This, together with concerns over side effects, may raise doubts among neurologists. Further research and collaboration with neurological colleagues should be encouraged to address these concerns, as ECT may be a viable option that is acceptable to many patients. Initial reports suggest that other movement disorders, particularly those with parkinsonism, may improve with ECT. Further observation of patients with these disorders who undergo ECT will be needed to clarify the potential role for ECT.