Medical and pharmacy claims
Medical claims or encounter data were collected from all available sites for all types of services provided and were coded in conformance with insurance industry standards. Claims for ambulatory services submitted by individual providers (usually physicians) use Health Care Financing Agency (HCFA)-1500 forms and claims for hospital-based services use UB-92 forms (www.cms.hhs.gov).
Facility service records contain information on up to nine diagnoses and six procedures, categorized by
International Classification of Diseases, Ninth Revision, Clinical Modification (
ICD-9-CM ) diagnosis codes (
24 ), and they contain information on procedure codes based on
ICD-9-CM, Current Procedural Terminology (CPT), or HCFA Common Procedure Coding System (HCPCS) protocols (www.cms.hhs.gov/home/medi care.asp). Facility claims contain categories of most services but may not account for all drugs administered in the hospital. Provider service records contain information on up to four diagnoses recorded with
ICD-9-CM codes and one procedure recorded using
ICD-9-CM, CPT, or HCPCS codes. Claims for pharmacy services include drug names, dosage forms, drug strengths, fill dates, and number of days' supply.
Study population and characteristics
Health plan members initiating treatment for bipolar disorders were identified using eligibility criteria and medical and pharmacy claims data acquired between January 1, 2001, and December 31, 2005. To be included in the study, persons were required to be continuously enrolled in the health plan for six months before and for 12 months after a first prescription fill for a psychotropic medication. Persons were considered for the study if they were diagnosed as having bipolar disorder, were aged 17 years or older, were newly initiating pharmacological treatment for bipolar disorder (no psychotropic prescription fills in the preceding six months). Patients were entered in the study between January 1, 2001, and December 31, 2004, and were followed for 12 months after initiating treatment. Persons diagnosed as having schizophrenia, epilepsy, or migraines were excluded.
Demographic characteristics were assigned according to information reported in index claims for the first psychotropic prescription fill, including sex, current age, and geographic region. If more than one prescription by more than one prescriber was filled on the index date, the provider type was assigned hierarchically in the following order: psychiatrist, general practitioner, other specialist, and unknown.
Diagnosis of bipolar disorder and its subtypes was based on the presence of
ICD-9-CM diagnosis codes indicating such illnesses that were then converted to corresponding
DSM-IV codes according to guidelines provided by the American Health Information Management Association (AHIMA) (
25 ). For this study, patients who ever had an
ICD-9-CM diagnosis of bipolar affective disorder (
ICD-9-CM codes 296.4–296.66), corresponding with
DSM-IV bipolar I disorder (
DSM-IV codes 296.4–296.66), were considered to have type I bipolar disorder; those who did not have bipolar disorder type I but had ever been diagnosed as having
ICD-9-CM manic-depressive psychosis, other or unspecified type (296.89), corresponding to
DSM-IV bipolar II disorder (296.89), were considered to have type II bipolar disorder; those who did not have type I or II bipolar disorder but had ever been diagnosed with
ICD-9-CM affective psychosis, other or unspecified (296.7, 296.80), or with
ICD-9-CM atypical manic or depressive disorder (296.81–296.82) were considered to have bipolar disorder not otherwise specified.
Patients also were categorized according to their apparent illness complexity, on the basis of selected clinical factors and presence of higher-severity bipolar disorder indicators or comorbid conditions that may complicate treatment, all as detailed by Solz and Gilbert (
26 ) and applied to other illnesses (
26,
27 ). We consulted with clinical and coding experts to identify
ICD-9-CM codes associated with relatively high-severity bipolar disorders and complex illness management. Indicators of higher illness complexity included diagnostic indicators for moderate or severe bipolar disorder (296.xx with fifth digit classification as "moderate" or "severe" or with psychotic features), an indication of self-harm (E950.x-E959) or overdose (969.xx), or presence of certain comorbid disorders (including bulimia nervosa [307.51], impulse-control disorder [312.30], or chronic fatigue syndrome [780.71]). Remaining patients with bipolar disorder were considered to represent relatively lower levels of illness complexity, and substance use comorbidity was considered separately.
We also calculated comorbidity scores at baseline and during follow-up by using an algorithm developed by Charlson and collaborators (
28 ) and adapted by Deyo and colleagues (
29 ) for use with administrative claims databases. To better identify patients with specific clinical conditions used to score comorbidity, required diagnosis codes were updated in consultation with clinical and coding experts to reflect recent changes or additions to the codes, without change in the number or identity of clinical conditions used to calculate comorbidity scores. These included diagnosis of the clinical state (depressed, manic or hypomanic, mixed, or unspecified) closest in time to the initial prescription fill was determined.
Treatment regimens
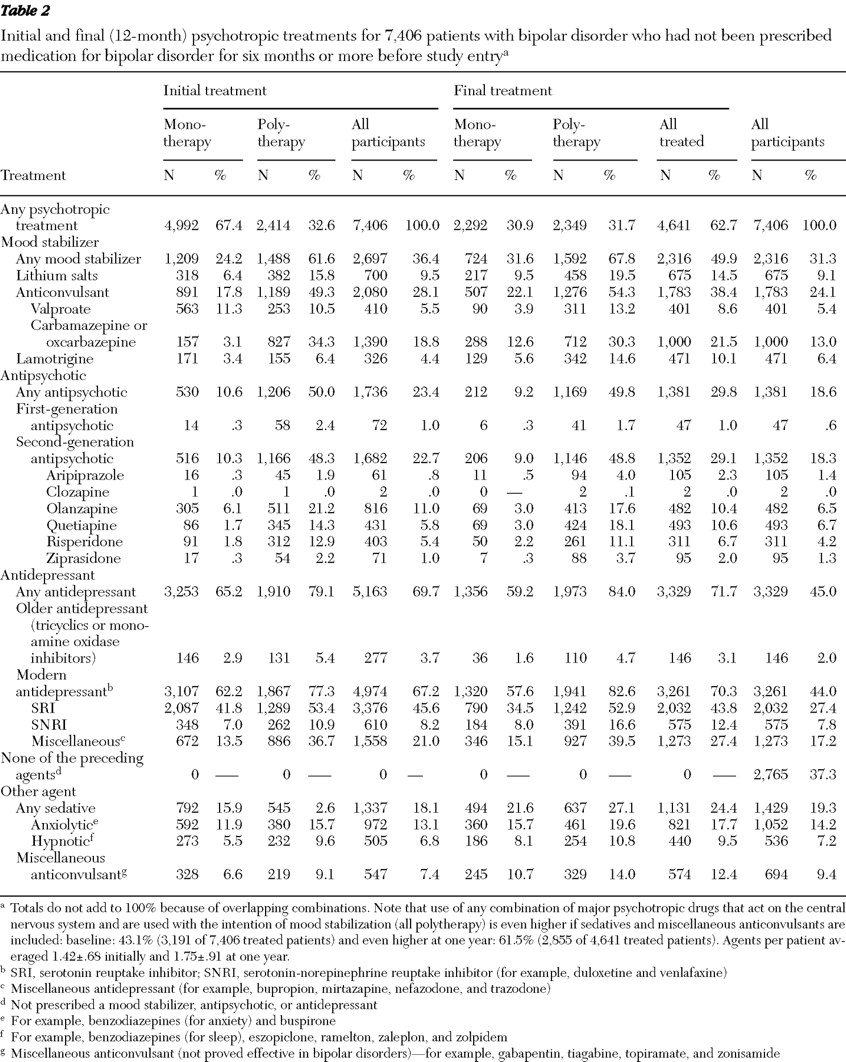
Psychotropic medicines provided were categorized as mood stabilizers, including lithium and anticonvulsants (carbamazepine or oxcarbazepine, valproate [any salt of valproic acid, including divalproex], and lamotrigine); antipsychotics, including modern antipsychotics (aripiprazole, clozapine, olanzapine, quetiapine, risperidone, and ziprasidone) and older neuroleptics (chlorpromazine, fluphenazine, haloperidol, loxapine, mesoridazine, molindone, perphenazine, pimozide, promazine, thioridazine, thiothixene, trifluoperazine, and triflupromazine); antidepressants, including serotonin reuptake inhibitors (SRIs) (citalopram as its racemate or R-isomer, fluoxetine, fluvoxamine, paroxetine, and sertraline) and serotonin-norepinephrine reuptake inhibitors (SNRIs) (duloxetine and venlafaxine), tricyclic antidepressants (including amitriptyline, amoxapine, clomipramine, desipramine, doxepin, imipramine, maprotiline, protriptyline, nortriptyline, and trimipramine), monoamine oxidase (MAO) inhibitors (isocarboxazid, phenelzine, and tranylcypromine), and modern antidepressants (including bupropion, mirtazapine, nefazodone, and trazodone). We also identified concomitant use of anxiolytic-sedatives, or hypnotics, including benzodiazepines, as well as miscellaneous anticonvulsants of unproved efficacy in bipolar disorders (gabapentin, levetiracetam, tiagabine, topiramate, and zonisamide). Initial treatments and combinations were assigned as psychotropic prescriptions dispensed during the first 30 days of treatment. Final regimens were assigned by psychotropic prescriptions dispensed within the final 90 days of follow-up.
We also examined overall health care utilization during follow-up and recorded office or emergency service visits, hospitalizations, and hospital days per patient, based on services rendered, as identified in the claims database.
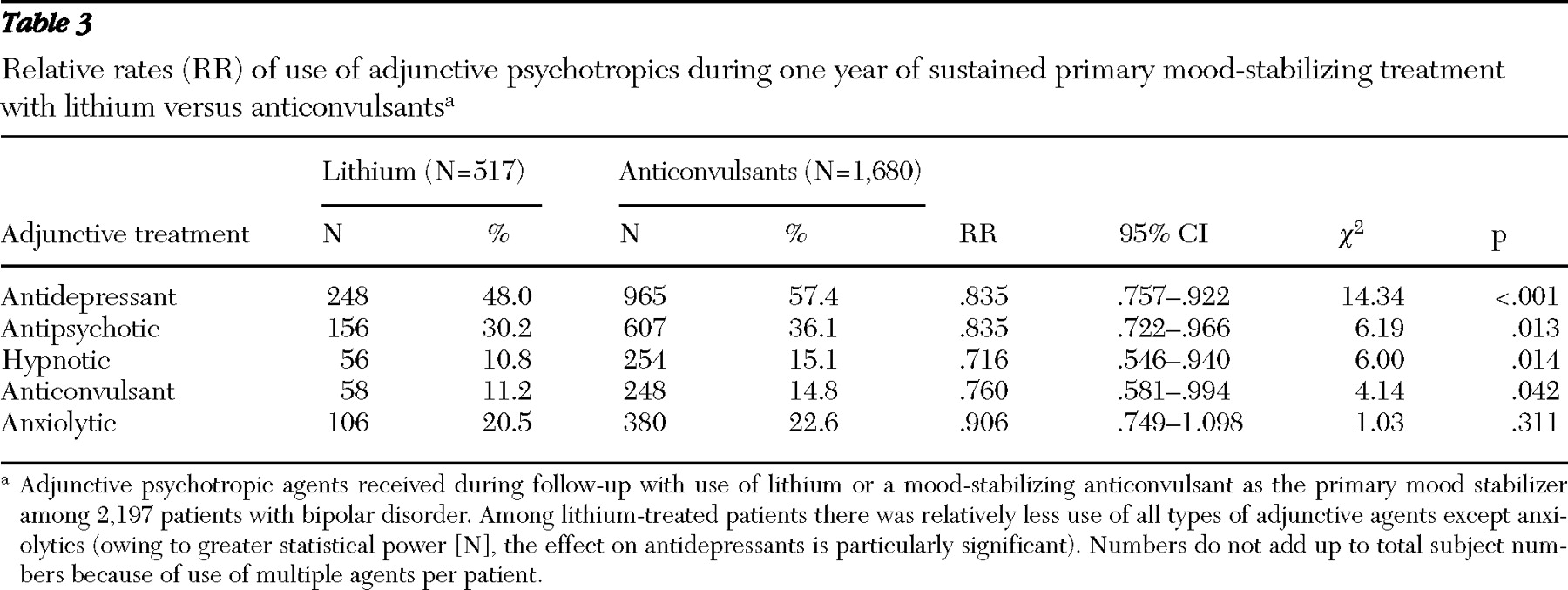
To evaluate mood stabilizer adherence, specifically, we identified a subgroup of patients who took a single mood stabilizer and initiated treatment with lithium or a mood-stabilizing anticonvulsant (carbamazepine, lamotrigine, oxcarbazepine, or valproate) and neither switched nor augmented therapy with any other mood stabilizer during follow-up. We measured mood stabilizer adherence by using a medication possession ratio (MPR). MPR is the percentage of the past 365 days with apparent access to an initial mood stabilizer. Also, a lower MPR would capture both treatment nonadherence and interruptions in treatment.
Analytic approach
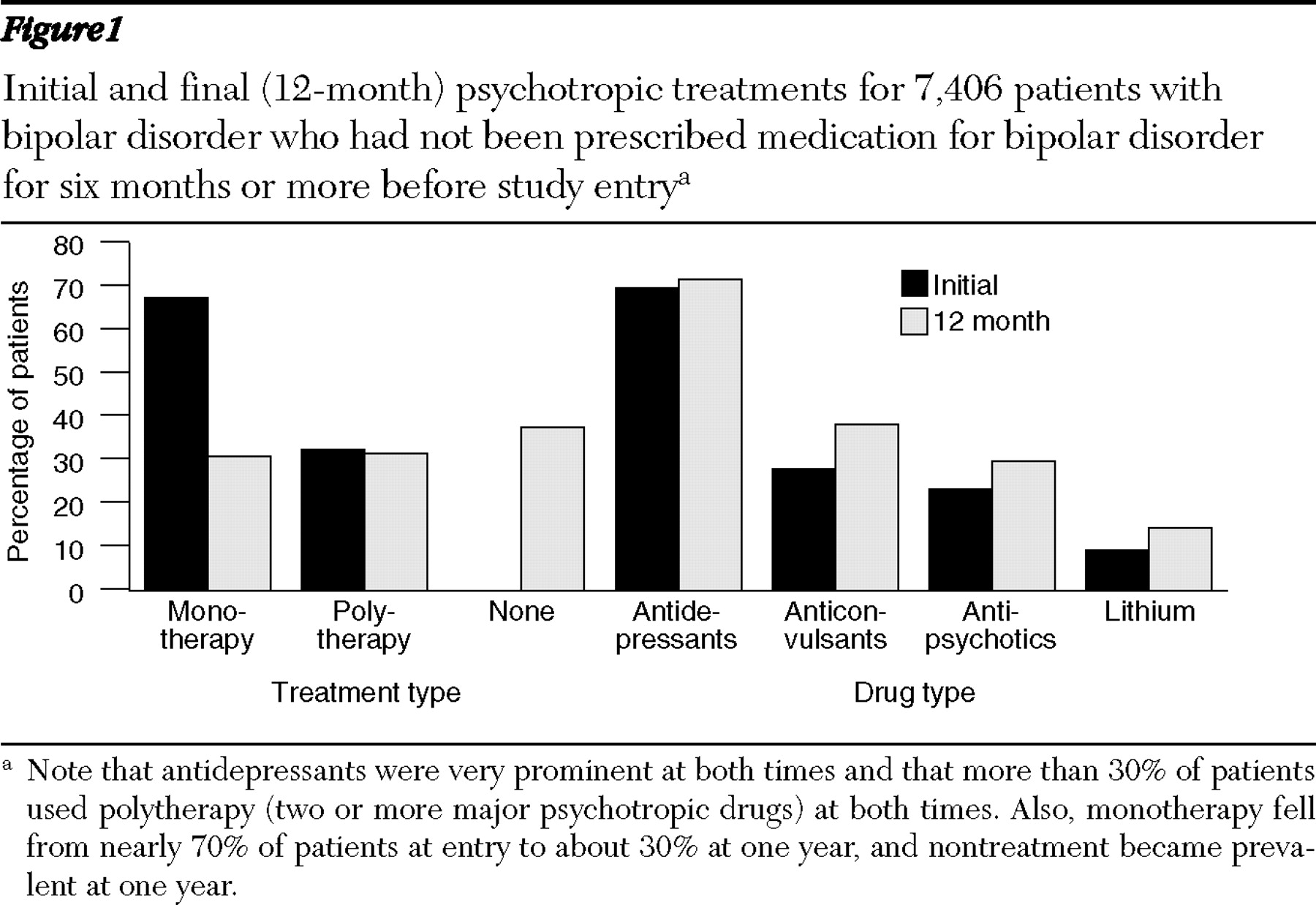
We analyzed patient demographic and clinical characteristics, treatment regimens, and health care utilization in the study sample. We considered initial treatment patterns and examined changes in treatment during a year of follow-up, comparing all single and multiple psychotropic prescriptions dispensed during the first 30 days (initial treatment) to those during the final 90 days of follow-up (final treatment).
To identify characteristics associated with treatment adherence in the subsample of patients treated with a single mood stabilizer for up to a year, we compared factors of potential interest (including sex, age, diagnostic type, comorbidities, health care service use, prescriber type, geographic regions, mood-stabilizer treatment, and psychotropic co-treatments), contrasting adherent patients (MPR ≥80%) and nonadherent patients (MPR <80%) in preliminary bivariate comparisons. Factors differing between these adherence subgroups were further evaluated by multivariate least-squares regression modeling. Modeling included days hospitalized per patient to control for potential effects of prolonged hospitalizations with incompletely recorded inpatient pharmacy claims. Index year of starting treatment was also included to adjust for possible changes in prescribing patterns over time. When multiple items were considered in some comparisons, one was selected as a comparator against which effects of other factors on treatment adherence were related.
Data are reported as means and standard deviations (SDs) unless stated otherwise, and statistical tests required two-tailed p<.05 for significance. Analyses used commercial statistical software (SAS 9.1.3).