Because of comorbid medical conditions, poor tolerance of psychotropic medications, and psychotic features or marked disability associated with depression, ECT is often the treatment of choice in older patients with severe depression
(1). As a result, the elderly constitute more than one-half of American patients who receive ECT for the treatment of depression
(2). Several studies have addressed the risks and benefits of ECT for older patients
(1). However, there is a paucity of data specifically addressing the outcome of ECT for persons over 75 years of age, often referred to as the “old-old”
(3–
5). Data relevant to these patients are usually combined with data on patients who may be 10 or 20 years younger, even though there is great variability in physiological, clinical, economic, and social characteristics among persons 65 and over
(3,
4). For instance, suicide risk doubles between ages 65 and 85
(6, p. 105).
To our knowledge, five studies have examined the outcome of ECT in the old-old
(7–
11). In each study ECT was found to be safe and effective. The design of four of these studies was limited by the use of retrospective chart review
(7,
9–
11), lack of a comparison group
(7,
9–
11), or small study group size
(9). The only prospective study of which we are aware used a standardized rating scale to examine clinical outcome among 78 patients of varying ages
(8). This study showed a trend toward greater improvement in the 20 old-old patients than in younger patients.
In the context of a large prospective, multisite study with standardized ECT administration and clinical assessment, we compared the outcome of ECT in adult (59 years of age and younger), young-old (60 to 74 years), and old-old (75 years and older) patients with major depression. We hypothesized that the old-old patients receiving ECT would have a higher burden of physical illness, would respond as well as younger adults and young-old patients, but would be more likely to experience cognitive impairment.
METHOD
The subjects participated between June 1992 and March 1998 in a multisite study that prospectively examined the relationship of medication resistance to ECT response and subsequent relapse. We have previously reported detailed study methods and preliminary results on a subgroup of subjects
(12,
13). Patients referred for ECT at four hospitals (Carrier Foundation, Belle Mead, N.J.; New York State Psychiatric Institute, New York; University of Iowa Hospitals and Clinics, Iowa City; and the Western Psychiatric Institute and Clinic, University of Pittsburgh Medical Center) were invited to participate if they met the Research Diagnostic Criteria (RDC)
(14) for an episode of primary, unipolar major depression and if they had a score of at least 20 on the 24-item Hamilton Depression Rating Scale
(15). Written informed consent was obtained from all study patients after the procedures had been fully explained.
The patients were interviewed before ECT with the Schedule for Affective Disorders and Schizophrenia
(16). Diagnoses were then made according to both the RDC and DSM-III-R. If psychotic features were present at any time during the index episode, the episode was classified as psychotic. In almost all cases the RDC and DSM diagnoses were identical, and DSM diagnoses are presented here. Patients were excluded if they had a history of schizophrenia, schizoaffective disorder, or other functional psychosis, had a history of neurological injury or disorder (including a dementing disorder), had abused substances other than nicotine within the past year, or had received a course of ECT within the past 6 months.
Before treatment, the physical status of each patient was assessed with a modified version of the Cumulative Illness Rating Scale—Geriatrics
(17,
18). Medical burden was rated for each of 14 organ systems on a 5-point scale (0=no problem, 1=mild, 2=moderate, 3=severe, 4=extremely severe).
Before starting and within 3 days of completing ECT, patients were rated with the 24-item version of the Hamilton depression scale, the Global Assessment Scale (GAS)
(19), and the Mini-Mental State
(20). To ensure adequate interrater reliability, the raters participated in annual training sessions and research interviews were videotaped and independently re-rated by an off-site rater. Intraclass correlation coefficients for the site and off-site ratings exceeded 0.90 for the Hamilton depression scale and GAS. Resistance to antidepressant treatment before ECT was classified by using the Antidepressant Treatment History Form
(12,
13), with the duration, oral dose, blood levels, compliance, and clinical outcome for each medication trial received during the index episode taken into account.
All psychotropic medications (with the exception of lorazepam, up to 3 mg/day) were tapered before ECT was started. The patients received either right unilateral ECT with the d’Elia placement or bilateral ECT with the bifrontotemporal placement. At all sites the same titration schedule—a “methods of limits” procedure—was used to empirically quantify seizure threshold at the first treatment with either right unilateral or bilateral ECT
(21,
22). For a small number of patients treated at one site (N=36), initial electrode placement and stimulus intensity were selected randomly. For all patients treated at the three other sites (N=232), initial electrode placement was decided according to the following guidelines: treatment started with suprathreshold right unilateral ECT unless the patient had not responded to suprathreshold right unilateral ECT in the past or the patient’s depression was so severe that the patient’s psychiatrist requested bilateral ECT. For right unilateral treatment, stimulus intensity was increased to a minimum of 150% above the empirically determined right unilateral seizure threshold (i.e., 2.5 times seizure threshold). At all sites, electrode placement was switched to bilateral ECT if the patient did not show significant improvement after at least five suprathreshold right unilateral treatments. For bilateral ECT, stimulus intensity was set 150% above the empirically determined bilateral seizure threshold (i.e., 2.5 times seizure threshold).
There was no preset maximum number of treatments for either modality. All patients were encouraged to continue ECT until they had fully responded (i.e., they had experienced complete or almost complete resolution of their depressive symptoms) or until they had been treated with at least 8–12 bilateral treatments and they had showed no further improvement during the past 2–3 bilateral treatments. Some patients (or their attending psychiatrists) refused to switch from right unilateral to bilateral ECT and/or decided to discontinue ECT before they received the recommended complete course.
At all sites, ECT was administered by using a custom-modified brief-pulse, constant-current MECTA SR1 device (Lake Oswego, Ore.). The four devices were calibrated and modified similarly to deliver a maximal energy of up to 200 J at an impedance of 200 Ω (or 1250 mC). Thirteen (4.6%) of the patients in this study had right unilateral seizure thresholds higher than 240 mC and would not have been able to receive a stimulus intensity equal to 2.5 times the right unilateral seizure threshold if they had been treated with one of the commercial devices currently available in the United States. For virtually all patients, the medications given at the time of ECT included atropine (0.4 mg i.v.), methohexital (0.75–1.0 mg/kg), and succinylcholine (0.75–1.0 mg/kg).
The patients were divided into three groups on the basis of age: 59 years and younger, 60 to 74 years, and 75 years and older. The demographic, clinical, and treatment characteristics of these three groups were compared with chi-square analyses and analyses of variance (ANOVAs), as indicated; when statistical differences were found, the Tukey post hoc test was used for pairwise comparisons. Patients with Hamilton depression scores of 10 or less after completion of ECT were classified as responders, and the response rates of the three groups were compared by using chi-square analyses.
To further explore whether there was an interaction between study site, age, physical burden, and pre-ECT Hamilton depression score in predicting response, we also performed a logistic regression using these variables as predictors and response as the dependent variable (with partial responders included in the nonresponder group).
A small number of patients did not have post-ECT scores, either because they discontinued ECT before they received at least five treatments or because their diagnoses were changed once more information became available. There was no statistical difference in the proportions of these “dropouts” across the three age groups: 15.0% of the patients 59 years and younger (20 of 133), 12.7% of those 60 to 74 years old (eight of 63), and 8.3% of those 75 years and older (six of 72) (χ2=1.90, df=2, p=0.39).
RESULTS
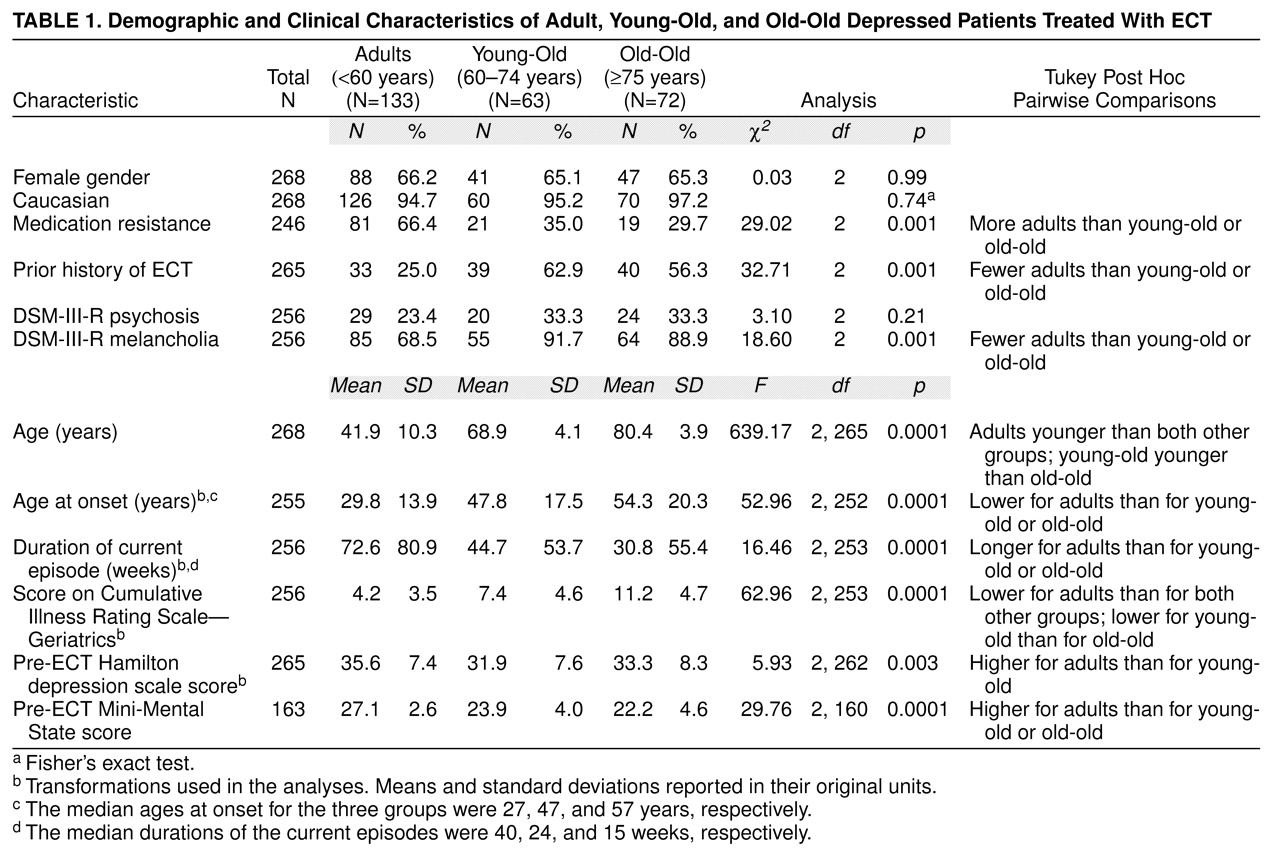
Demographic and clinical characteristics of the three study groups are contrasted in
table 1. When compared to both groups of older patients, the patients aged 59 years and younger had a significantly lower medical burden (total score on Cumulative Illness Rating Scale—Geriatrics and number of systems rated as having a severe or extremely severe problem), lower age at onset of major depression, smaller proportion of patients with melancholia, and higher mean baseline Mini-Mental State score. They also were less likely to have received ECT in the past, had longer current episodes, and were more likely to be medication resistant. The patients aged 60 to 74 years had a significantly lower medical burden than those aged 75 years and older. Otherwise, the two older age groups did not differ.
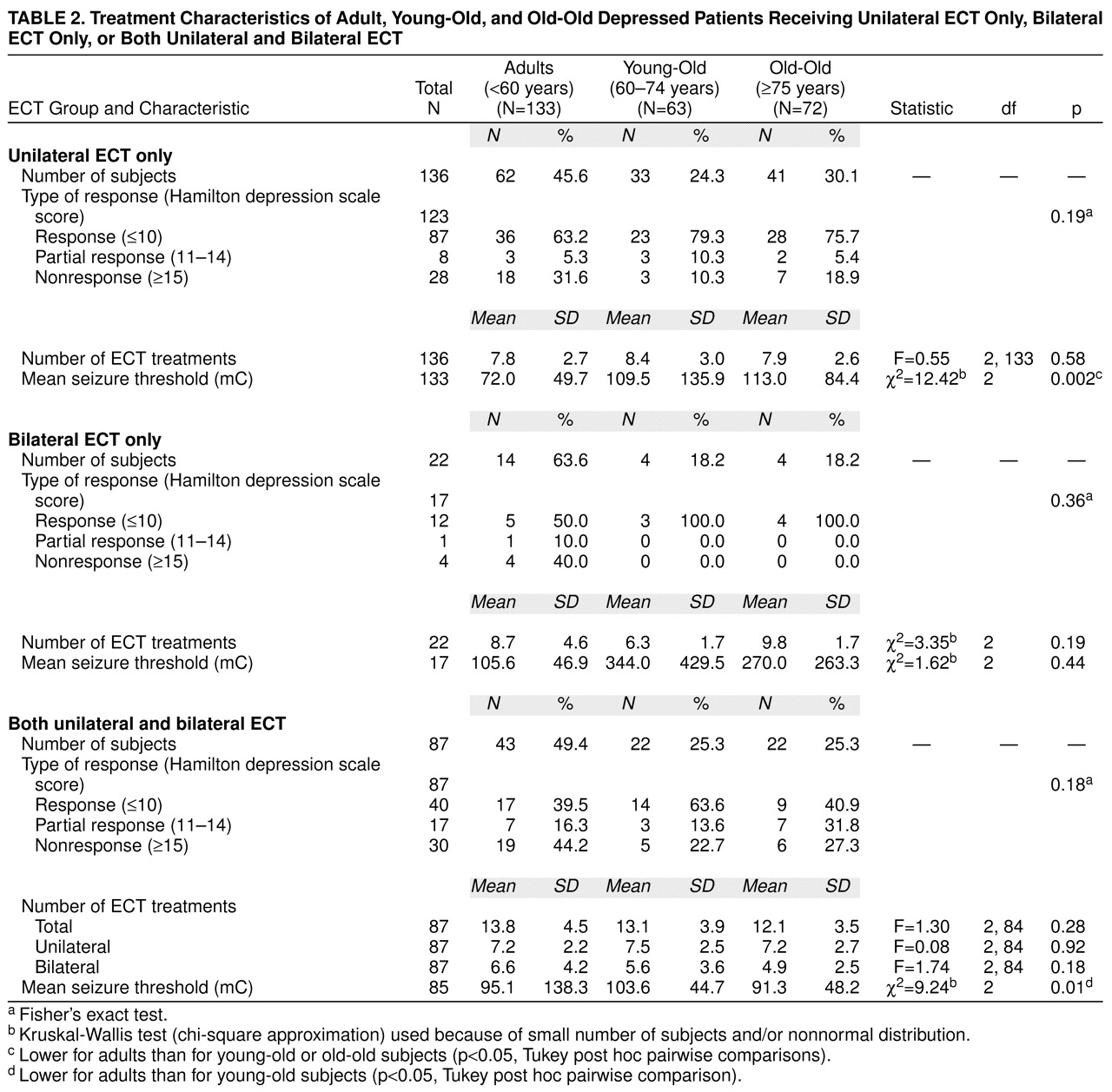
table 2 presents the treatment characteristics of the three study groups as a function of treatment type: unilateral ECT only, bilateral ECT only, or both unilateral and bilateral ECT (patients who were randomly assigned to initial treatment with right unilateral or bilateral ECT were excluded). With right unilateral ECT, the patients aged 59 years and younger had a significantly lower seizure threshold than each of the older groups. There were no significant differences among the age groups in total number of treatments or likelihood of switching from unilateral to bilateral ECT.
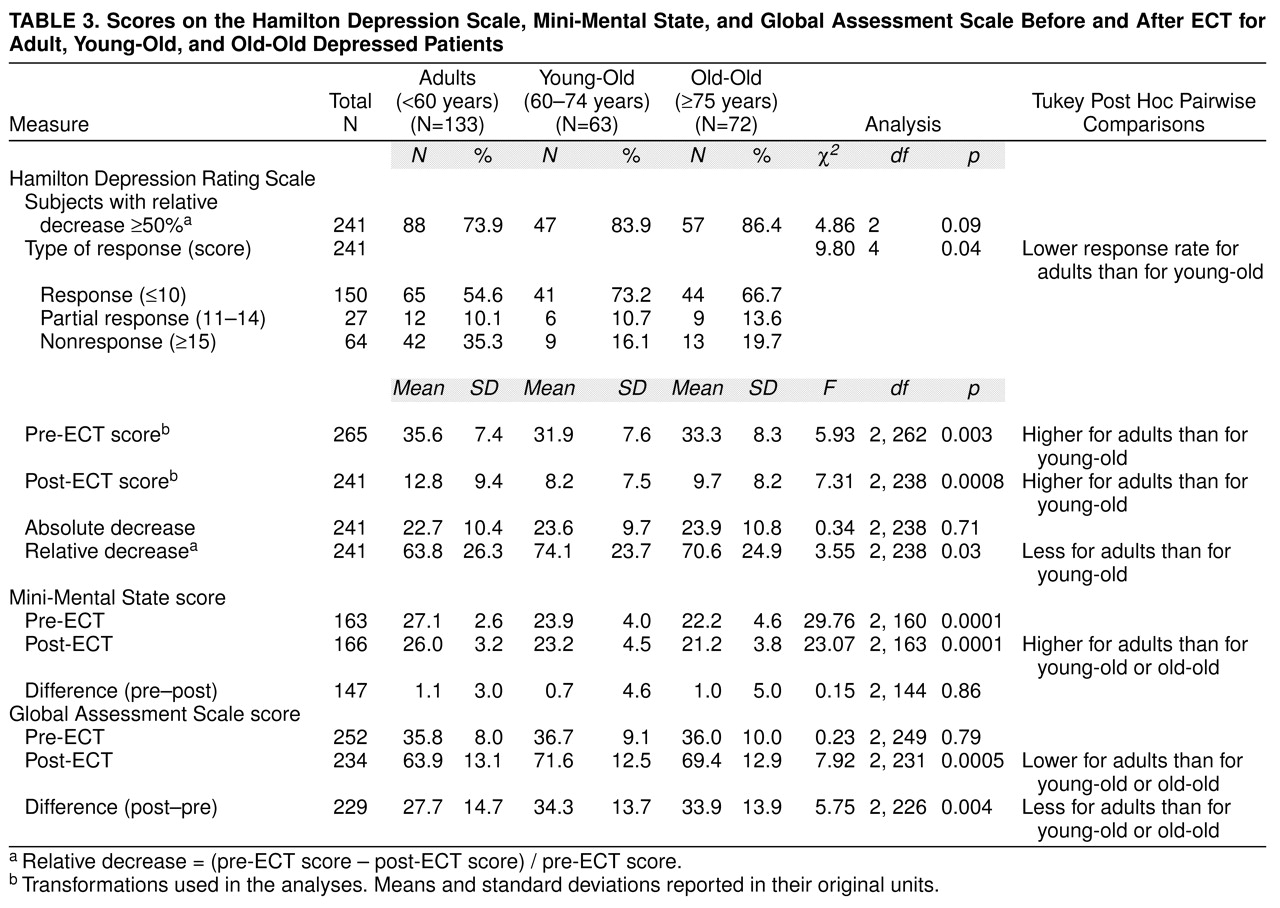
The therapeutic and global cognitive outcomes for the three groups are summarized in
table 3. Patients aged 59 years and younger had higher pre- and post-ECT Hamilton depression scale scores and a lower rate of response than patients aged 60 to 74 years. Similarly, they experienced a smaller improvement as measured by the GAS. A logistic regression failed to reveal a significant association between response and physical burden (score on the Cumulative Illness Rating Scale—Geriatrics) after age, study site, and pre-ECT Hamilton depression score were controlled for (χ
2=0.15, df=1, p=0.70). In the three age groups, there were small decreases in Mini-Mental State mean scores after ECT (
table 3); this decrease reached statistical significance only for the youngest patients (t=3.4, df=84, p=0.001; young-old: t=0.9, df=33, p=0.40; old-old: t=1.1, df=27, p=0.28).
DISCUSSION
To our knowledge, this report includes the largest number of patients aged 75 and over enrolled in a prospective study of ECT using standardized rating scales. Several findings deserve comment.
The demographic and clinical characteristics of the old-old patients (75 years and older) who received ECT were similar to those of the young-old patients (60 to 74 years). In contrast, both groups differed along several dimensions from the patients aged 59 years and younger. The older patients had significantly shorter index depressive episodes and were less likely to have had inadequate responses to adequate medication trials before ECT. This suggests that in many cases, the degree of impairment associated with severe geriatric depression warranted rapid intervention. Because of the faster response with ECT than with pharmacotherapy, the relative absence of drug interactions, and the benefits of early intervention for treatment response, ECT is often selected as a first-line treatment for elderly patients with severe major depression
(5,
23,
24).
The safety and efficacy of ECT have been demonstrated previously for depressed patients with cardiovascular disease
(25) or stroke
(26) and for physically ill elderly patients
(27). The results of the present study confirm that despite a higher burden of physical illness, as indicated by higher scores on the Cumulative Illness Rating Scale—Geriatrics, even the oldest depressed patients were able to complete and benefit from ECT courses in a manner similar to that for younger, physically healthier patients. This finding is congruent with the results of four previous studies that showed older physically ill patients respond in a manner similar to that of younger and physically healthier patients
(25,
28–
30).
The higher seizure threshold of the oldest patients is consistent with the repeated observation that seizure threshold increases with age
(21,
31,
32). Since the stimulus intensity during the ECT course was based on individual seizure threshold, despite their higher initial thresholds, the patients in the oldest age group were no more likely to be switched from suprathreshold right unilateral to suprathreshold bilateral ECT or to receive more treatments. Of note, the stimulus intensity was set 150% above these empirically determined seizure thresholds (i.e., 2.5 times threshold) since right unilateral ECT with a stimulus intensity below this level (e.g., just above threshold) has been shown to have minimal therapeutic efficacy
(22). The favorable response of many of these older patients to suprathreshold right unilateral ECT is of particular importance since they may be more susceptible to persistent confusion when treated with bilateral ECT
(24,
29,
33).
In a previous study, Wilkinson et al.
(8) observed a significant improvement in Mini-Mental State scores in all age groups after ECT. We did not observe such an improvement in any of our age groups. The reasons for this discrepancy could be related to when the post-ECT Mini-Mental State was administered. In the study of Wilkinson et al., the Mini-Mental State was administered 72 hours to 1 week after the final treatment, whereas in the present study, Mini-Mental State scores were usually obtained the day after, and no more than 72 hours after, completion of ECT. In two studies on the time course of cognitive side effects of ECT in elderly inpatients
(34,
35), Mini-Mental State scores decreased during a course of ECT but improved between completion of ECT and discharge.
Younger patients (under 60 years of age) showed a significantly smaller improvement in scores on the Hamilton depression scale and GAS, and they had a lower rate of categorical response than the young-old patients, while the old-old patients had intermediate values. Of note, their relatively low response rate (54.6%) was the same as the response rate of younger patients (54%) in the only other prospective study to compare acute ECT response in older and younger patients
(8). This response rate appears to be lower than the high rate of response historically associated with ECT (e.g., 70%–90%). However, these “historical” response rates should no longer be expected
(12,
36). As in this study, many patients currently treated with ECT have previously had unsuccessful medication trials, and these treatment-resistant patients are less likely to respond to ECT
(12,
36). In a subgroup (100 patients) of the subjects in this report, we showed
(12) that greater age is associated with a higher rate of response, but this association disappeared once statistical adjustment was made for the relationship between age and medication resistance. Thus, the differences in magnitude of improvement and response rate among the different age groups in this study might be attributed to the higher proportion of younger patients who were medication resistant (i.e., had not responded to at least one antidepressant medication trial before treatment with ECT).
In conclusion, this study suggests that despite a higher burden of physical illness and cognitive impairment, even the oldest patients with severe major depression tolerate ECT as well as younger patients and that they demonstrate similar or better rates of acute response. However, the present study has several limitations. Specific indications for ECT and transient complications some patients may have experienced during treatment were not reported. Our outcome data are limited to acute response, and thus we did not assess rates of relapse and recurrence during continuation or maintenance treatment after ECT. Future studies of ECT in the oldest old should address these important issues.