Radiologic studies indicate that schizophrenic patients with the syndrome of polydipsia and intermittent hyponatremia have ventricle enlargement and smaller amygdala-hippocampus volumes than do patient and normal control populations
(1–
3). However, these studies have not controlled for fluid status despite documented changes in the volume of intracranial structures during fluid loading in animal studies and in human case reports. It remains unclear whether ventricle enlargement reflects physiologic condition at the time of assessment or true differences between study groups. The potential confounding effect of fluid status needs to be resolved, because there is evidence that patients with polydipsia and intermittent hyponatremia suffer from a neurobiologically distinct syndrome
(1,
4,
5).
In an exploratory study, Elkashef et al.,
6) examined the impact of incidental water loading on ventricle size in three patients with polydipsia and intermittent hyponatremia. They found that third ventricle volume decreased during water loading in all patients, whereas lateral ventricle volume decreased in two of the three patients.
The purpose of this study was to verify that variations in physiologic condition during water loading account for the alterations of intracranial structures observed on magnetic resonance imaging (MRI). Using serum sodium concentration, serum osmolality, and body weight, we examined how changes in fluid status influence brain and ventricle volumes on MRI during a prospectively defined water-loaded state versus normal hydration.
METHOD
We studied eight male patients with a DSM-IV diagnosis of schizophrenia (N=6) or schizoaffective disorder (N=2). Their mean age was 39.7 years (SD=6.1), and the mean length of psychiatric illness was 23.8 years (SD=3.9). In the prior 6 months, each patient had had, in association with polydipsia, 1) two episodes of acute hyponatremia, 2) two episodes of more than 5% normalized diurnal weight gain, and 3) two morning urine specific gravities less than 1.008. We ruled out common causes of hyponatremia. After complete description of the study to the subjects, written informed consent was obtained according to state and university hospital institutional review board guidelines.
The patients were first randomly assigned to one of two conditions: normal fluid intake or oral water loading. The experiment was then repeated under the alternative condition the following day. During water loading, patients were allowed to drink freely until 1) serum sodium concentration was ≤130 meq/liter, 2) serum osmolality was ≤280 mOsm, or 3) body weight increased by more than 5%.
Physiologic measures and body weight were ascertained each morning immediately before the MRI to confirm that the desired protocol condition was achieved. Psychological variables reflecting psychiatric symptoms (Brief Psychiatric Rating Scale [BPRS] score) and cognition (scores on the Controlled Oral Word Association test
[7] and the Mini-Mental State examination
[8]) were also assessed. MRI was performed with a 1.5-T unit with a standard head coil. Images were stored on magnetic tapes and transferred to a mensuration system, and target structures were anatomically defined with the use of previously described methods
(9).
Paired t tests with a Bonferroni-corrected alpha probability of p<0.01 were used to compare global volumetric measures across conditions. Multiple regression analyses were used to assess relationships between ventricle-brain ratio (VBR) change scores and physiologic change scores.
RESULTS
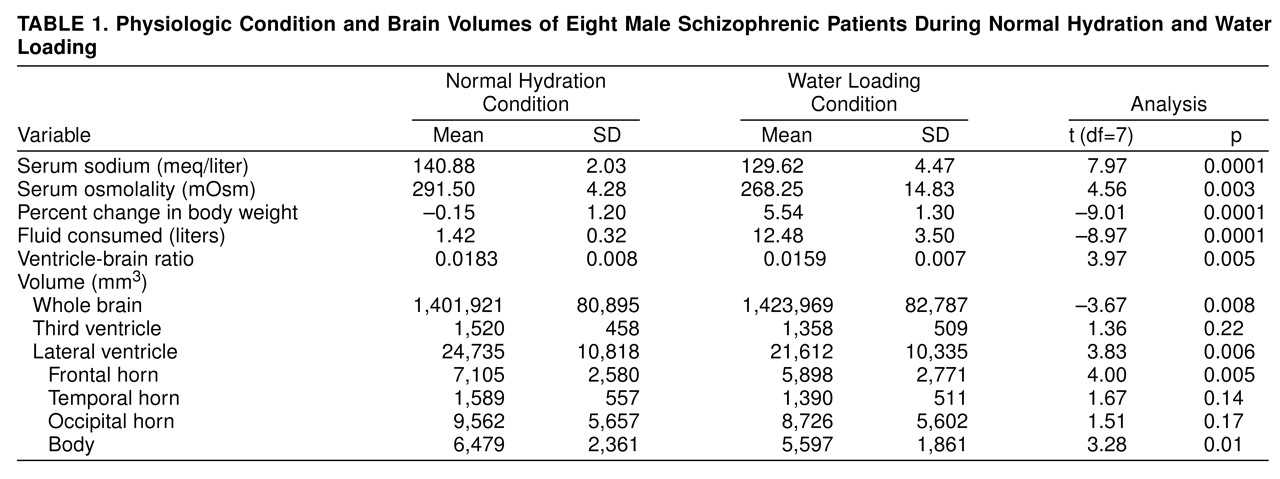
Table 1 presents physiologic measures during the water loading and normal hydration conditions. Patients successfully met the a priori criteria for each condition at the time of the MRIs. Typical of this syndrome, our patients consumed a mean of 12.48 liters of fluid during the 8-hour water loading period. All patients had normal serum sodium levels and osmolality and experienced insignificant weight change during the normal hydration study.
Total VBR decreased by 13.1% during the water loading condition. Lateral ventricle volume decreased by 12.6%, while total brain volume increased by 1.6%. Examination of ventricular subsections showed that volume decreases ranged from 17.0% in the frontal horns to 8.7% in the occipital subsection. Scores on the BPRS, the Controlled Oral Word Association test, and the Mini-Mental State examination did not change significantly.
We found a powerful association between change in serum sodium concentration and change in VBR (r=0.82, df=6, p=0.006). This correlation was less strong for serum osmolality (r=0.63, df=6, p<0.05), weight change (r=0.42, n.s.), and amount of fluid consumed (r=0.23, n.s.). We found no association between age or length of illness and non-water-loaded ventricle volumes, VBR, or changes in VBR.
DISCUSSION
There was a significant decrease in VBR and ventricle size during hyponatremic conditions in patients with polydipsia and intermittent hyponatremia. At the same time, brain volume increased in a manner consistent with cerebral edema. These findings suggest that abnormalities in sodium or water metabolism did not confound findings of ventricle enlargement in previous studies of patients with polydipsia and intermittent hyponatremia
(1–
3). Specifically, Emsley et al.
(1) found larger frontal horns in such patients. If their patients had been hyponatremic at the time of examination (unlikely but not determined; reference 1, p. 502), the degree of enlargement would have been blunted. Similarly, the finding by Kirch et al.
(2) of smaller amygdala-hippocampal volumes would have been obscured by the effect of hyponatremia.
We have documented how alterations in sodium balance are associated with reversible, state-dependent changes in brain morphology, such that decreases in ventricle volume are strongly correlated with severity of hyponatremia. Similar physiologic influences have been previously reported in alcoholism and anorexia nervosa. Thus, transient metabolic or physiologic changes may be superimposed on more enduring disease-related changes.
We did not find a significant change in neuropsychiatric variables during hyponatremia. Since patients with polydipsia and intermittent hyponatremia are already cognitively compromised
(5), further impairment would require drastic physiologic intervention, whereas the hyponatremia induced was relatively mild for these patients. Our findings instead emphasize how sensitive intracranial structures are to hyponatremia, whereas neuropsychological functioning is more resistant to physiologic alterations.
This study adds to accumulating evidence that patients with polydipsia and intermittent hyponatremia suffer from a severe neurobiological syndrome that distinguishes them from schizophrenic patients without polydipsia and intermittent hyponatremia through radiologic, neurologic, behavioral, psychological, and endocrine measures
(5). Patients with polydipsia and intermittent hyponatremia may suffer from a distinct syndrome sharing psychiatric symptoms but including abnormal sodium and water metabolism. Alternatively, patients with polydipsia and intermittent hyponatremia may share the same disorder but manifest more profound neuropathology, as evidenced by common but exaggerated findings of ventriculomegaly and hippocampal abnormalities. Finally, perhaps these distinctions are due to repeated encephalopathy, seizures, and coma causing neuronal damage and an inordinately large VBR.
Our study has documented the importance of monitoring serum sodium levels during neuroimaging studies to control for influence on the volume of intracranial structures. The sensitivity of neurobiological variables to physiologic alterations needs careful attention when biological studies are being performed.