Several indirect techniques provide evidence that serotonin (5-HT) physiology is disturbed in depression. Brain uptake of the 5-HT precursor 5-hydroxytryptophan is reduced, and the cerebrospinal fluid concentration of the 5-HT metabolite 5-hydroxyindoleacetic acid is usually low
(1,
2). Prolactin response to fenfluramine, a 5-HT releasing agent, is decreased
(3,
4). In treated depressed subjects, depletion of the 5-HT precursor tryptophan induces a relapse into sad mood
(5). These findings represent generalized 5-HT disturbances but do not indicate the neurochemical level(s) at which they may occur. Such changes could reflect alterations in 5-HT synthesis, 5-HT metabolism, postsynaptic receptors, or linked second messenger systems.
Postmortem and platelet receptor binding studies suggest that 5-HT abnormalities exist at the postsynaptic receptor level in depressed subjects who are suicidal. Postmortem studies of antidepressant-free depressed suicide victims report either significant
(6,
7) or nonsignificant
(8) increases in prefrontal cortex 5-HT
2 receptor binding. Cortex 5-HT
2 receptor binding may be reflected by platelet receptors, and elevated platelet 5-HT
2 receptor binding is commonly found in depression and suicidal states
(9–
18). These findings do not exclude the possibility that changes occur at other neurochemical levels; however, the presence of increased prefrontal 5-HT
2 receptor binding in depressed suicide victims has considerable support.
Depressed suicide victims usually have increased prefrontal cortex 5-HT
2 receptor binding
(6–
8). However, this finding may not be true for depressed subjects who have not made recent suicide attempts. Since increased 5-HT
2 receptor binding is present in undiagnosed suicide victims, it could represent the presence of suicidality rather than depression
(19–
21). In postmortem studies, it is difficult to investigate the effects of depression independent of suicide. McKeith et al.
(22) chose a group of subjects with major depressive disorder who had not been victims of suicide and showed a trend toward increased prefrontal cortex 5-HT
2 receptor binding. However, this study group was heterogeneous and included patients with histories of medication use, psychotic symptoms, and euthymia at the time of death.
Use of medication is a major confounding factor with respect to postmortem studies of 5-HT
2 receptor binding. In the one postmortem study that examined the effects of antidepressants, Yates et al.
(7) found an increase in prefrontal cortex 5-HT
2 receptor binding in unmedicated depressed subjects; however, those receiving chronic antidepressant treatment did not differ from the control group. In animal cortex, postmortem 5-HT
2 receptor binding changes with several types of chronic antidepressant treatment
(23–
26). Additional variance in the results of postmortem studies may be attributable to cause of death, delay in tissue processing after death, and choice of ligand
(27).
Two neuroimaging studies investigated 5-HT
2 receptors in primary depression. Using single photon emission computed tomography (SPECT), D’haenen et al.
(28) reported an increase in left-to-right prefrontal cortex 2-[
123I]ketanserin uptake. Using positron emission tomography (PET), Biver et al.
(29) found a significant decrease in [
18F]altanserin uptake in the left inferofrontal cortex. Unfortunately, both findings were confounded by recent psychotropic drug use, and it is not clear whether the subjects participating in these studies had made recent suicide attempts. In both studies one-half of the subjects had used psychotropic medication as recently as 10 days before scanning.
The purpose of this study was to investigate the effects of depression on 5-HT
2 receptors in patients without recent suicide attempts by means of [
18F]setoperone PET. [
18F]Setoperone has a number of properties that make it favorable for imaging. Setoperone is an antagonist with high affinity and specificity for 5-HT
2 receptors. It has 100 times greater affinity for 5-HT
2A than 5-HT
2C receptors, thus making it relatively specific for the 5-HT
2A receptor
(30). Setoperone has only a 10- to 50-fold higher affinity for 5-HT
2 as compared to D
2 receptors, but D
2 receptors have very low density in the cortex
(31). 5-HT
2 and D
2 antagonists displace [
18F]setoperone in the striatum. Only 5-HT
2 antagonists displace [
18F]setoperone in the cortex; hence, [
18F]setoperone is still a suitable ligand for 5-HT
2 receptor imaging in the cortex
(32,
33). In fact, D
2 antagonists have no effect on cortical [
18F]setoperone signal
(33). [
18F]Setoperone has high brain uptake and a sufficiently high specific-to-nonspecific binding ratio
(32–
34). Metabolites of [
18F]setoperone do not appear to have significant brain uptake
(35). Since the cerebellum is practically devoid of 5-HT
2 and D
2 receptors, it may be used as a reference region to simplify image quantification
(31–
36). At our center, PET imaging with [
18F]setoperone has been shown to be reliable (test-retest reliability within 7%)
(37), and at another center, it has successfully confirmed an in vivo decrease in 5-HT
2 receptors in patients with Alzheimer’s dementia
(38).
On the basis of the foregoing evidence that unmedicated depressed suicide victims had increased 5-HT
2 receptor binding
(6,
7), and that in the only postmortem study of unmedicated depressed subjects who died of natural causes, a trend for increased 5-HT
2 receptor binding was also found
(22), it was hypothesized that increased frontal cortex 5-HT
2 receptor binding would be demonstrated in unmedicated depressed subjects who do not have a recent history of suicide attempts.
METHOD
This study was approved by the University of Toronto Human Subjects Review Committee. Fourteen depressed subjects, aged 18–40 years, and 19 healthy subjects, aged 18–45 years, were recruited by advertisement. All subjects had been free of any psychotropic drug use for more than 6 months, and no subject had taken any nonpsychotropic medication in the past 6 weeks. All were physically healthy and right-handed. Written informed consent was obtained from each subject after the procedures had been fully explained.
The 19 healthy subjects, who have been previously reported (37, 39), were screened with the Structured Clinical Interview for DSM-III-R—Non-Patient Edition
(40).
The patients’ diagnoses of a major depressive episode secondary to major depressive disorder were confirmed by the Structured Clinical Interview for DSM-IV—Patient Edition (SCID-P)
(41), which was administered by a trained research assistant. Each patient received a psychiatric consultation (with J.H.M. or S.H.K.) to verify the SCID-P diagnosis. A score higher than 17 on the Hamilton Depression Rating Scale
(42) was required for entry into the study. Patients with psychotic symptoms, bipolar disorder (type I or type II), or comorbid axis I diagnoses were excluded from the study. Potential subjects with a history of alcohol or drug abuse or dependence were also excluded, and any and all past drug use was recorded. Those with a history of substance use that did not meet criteria for the SCID-P diagnosis of substance abuse received a urine drug screen and then were included in the study only if the result was negative. Four subjects had previously completed a 6-week trial of an antidepressant, but none had received any antidepressant or psychotropic medication/substance within the past 6 months. The exclusion criteria also included a history of a suicide attempt during the previous year. Three subjects had a history of past suicide attempts, but no attempt had occurred within the past 5 years. Each depressed subject had routine tests (thyroid function, electrolytes, CBC) to rule out common medical causes of depression.
[
18F]Setoperone was prepared by [
18F]fluoride displacement on the nitroderivative precursor of setoperone on the basis of the procedure described by Crouzel et al.
(43). [
18F]setoperone was of high radiochemical purity (>99%) and high specific activity (mean=46 GBq/µmol, SD=20, at the time of injection). Imaging was based on the approach described by Blin et al. (32, 33). An intravenous bolus of 185 MBq of [
18F]setoperone was injected. PET images were obtained with the use of a GEMS 2048-15B camera (General Electric Medical Systems, Milwaukee, Wis.). Images were obtained in five 1-minute frames followed by 17 5-minute frames. The images were corrected for attenuation with a
68Ge transmission scan and reconstructed by filtered back projection (Hanning filter, 5 mm full width at half maximum).
To obtain a semiquantitative measure of 5-HT
2 receptor binding potential, we used the pseudoequilibrium prefrontal cortex-to-cerebellum ratio
(37). The cerebellum has no displaceable [
18F]setoperone binding
(32,
33). Between 65 and 90 minutes, there is no significant change in the prefrontal cortex-to-cerebellum ratio
(37). If the cerebellar concentration of [
18F]setoperone is used as an index for free and nonspecifically bound ligand in the cortex, then it can be shown that the cortex-to-cerebellum ratio between 65 and 90 minutes is linearly proportional to k
3/k
4 and f
2 × B
max/K
d (34) (k
3 and k
4 are rate constants for ligand transfer between free and specific binding compartments; f
2 represents the free fraction of ligand; B
max represents the total number of receptors available to a ligand; K
d is the dissociation constant for the ligand at the receptor; and pseudoequilibrium refers to a period of time in which the k
3/k
4 ratio is stable). Petite-Taboue et al.
(34) empirically demonstrated that the pseudoequilibrium cortex-to-cerebellum ratio is highly correlated with the k
3/k
4 ratio (r=0.91–0.97). Use of the cortex-to-cerebellum ratio avoids the need for an arterial puncture, making this approach less invasive
(34).
The 5-HT
2 binding potential does not estimate B
max or K
d separately. However, K
d is almost always unchanged in postmortem studies of depression and/or suicide, whereas changes are observed in B
max (6–
8,
19–
22).
For image analysis, each subject had a magnetic resonance imaging (MRI) scan (GE Sigma 1.5-T scanner; spin-echo sequence T
2-weighted image; x, y, z voxel dimensions=0.78, 0.78, and 3 mm, respectively). MRI scans were coregistered to each PET image by using the surface matching function in ANALYZE (CNS Software, Rochester, Minn.). For region-of-interest analysis, the regions of interest were drawn on the [
18F]setoperone scans with reference to the coregistered MRI by a rater blind to the identity of the subjects of the scans. The prefrontal cortex region of interest was drawn on five adjacent slices for each hemisphere, as described previously
(37). This region included Brodmann’s area 9 and parts of areas 8, 10, 46, and 32. The cerebellum region of interest was drawn bilaterally on two adjacent slices. Decay-corrected time-activity curves were obtained for each of these regions of interest.
The technique as described above is well standardized in our laboratory
(37,
39,
44). Pseudoequilibrium occurs between 65 and 90 minutes, since no significant change was found in the cortex-to-cerebellum ratio during this time in 20 healthy subjects
(37). To test the reliability of this measure, 11 normal subjects were scanned twice, and their data showed a test-retest standard deviation of 6%–9% and a mean scan-rescan difference of 5%–7%
(37). The intraclass correlation coefficient was greater than 0.98. Furthermore, this method of region-of-interest analysis has been applied to the same data with high interrater and intrarater reliability (37). We have already demonstrated that this technique is sensitive to age-related and medication-induced changes in 5-HT
2 receptors
(39,
44).
RESULTS
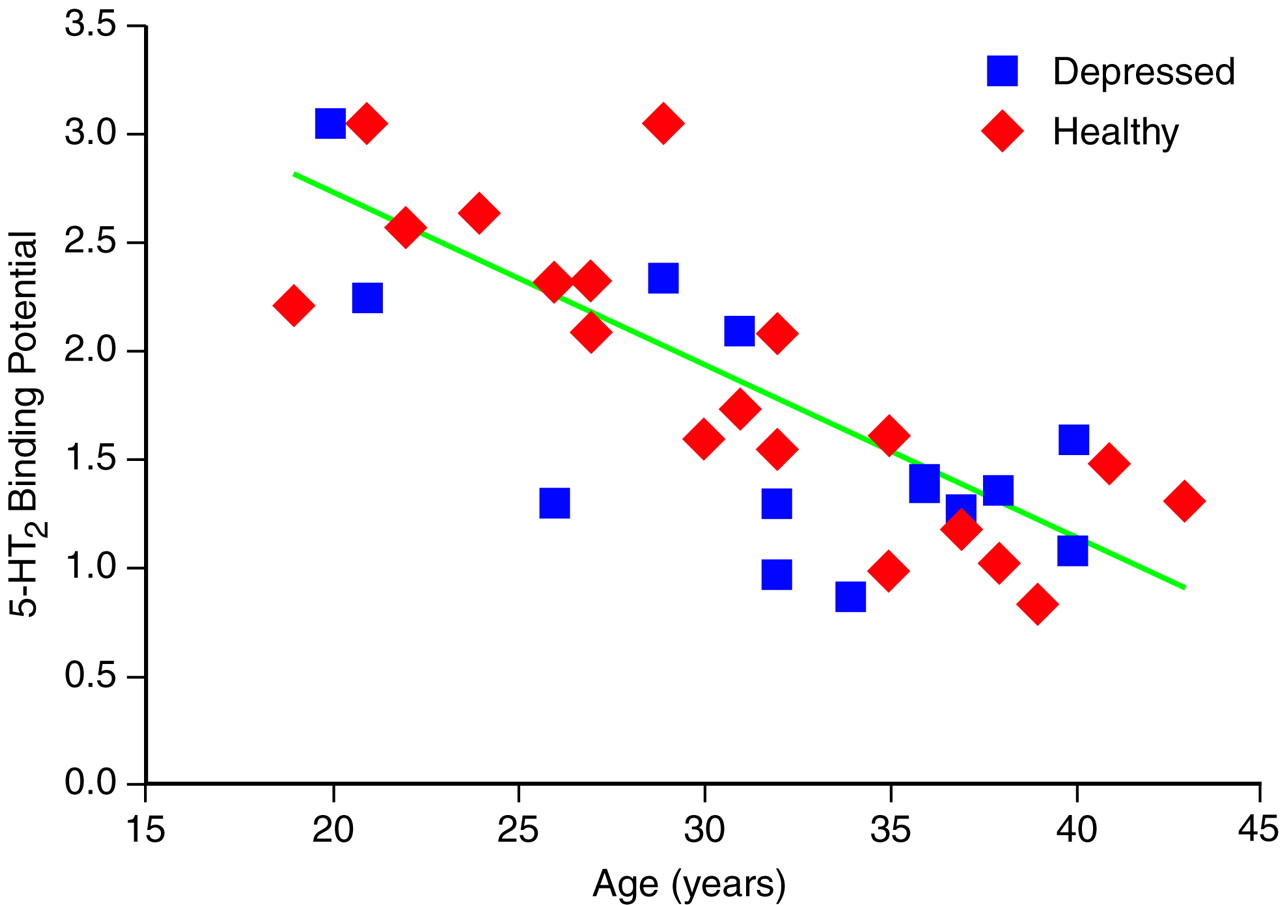
Fourteen patients (12 male and two female; mean age=32.3 years, SD=6.4) were compared with 19 healthy subjects (eight male and 11 female; mean age=31.8 years, SD=6.9). There was no significant difference in age between the patients and the healthy subjects (t=0.26, df=31, p=0.80). There were significantly more women in the comparison group (p=0.01, Fisher’s exact test). The mean baseline score of the patients on the Hamilton depression scale was 22.5 (SD=3.7). On the suicide subscale, five subjects scored 0, one subject scored 1, seven subjects scored 2, and two subjects scored 3. The mean prefrontal cortex binding potential was 1.6 (SD=0.6) in the depressed patients and 1.8 (SD=0.7) in the healthy subjects.
The effects of age and gender on binding potential were assessed. Age was associated with a significant decrease in 5-HT2 binding potential for all subjects (analysis of covariance [ANCOVA] with age as a covariant, F=42.3, df=1, 28, p<0.001), but gender was not (F=0.1, df=1, 28, p=0.70). The effects of age were also significant within the healthy group (F=31.9, df=1, 17, p<0.001) and the depressed group (F=11.2, df=1, 12, p=0.006); therefore, age was entered as a covariate in all further analyses. Gender was not significant within the healthy group (F=1.9, df=1, 16, p=0.20) or the depressed group (F=1.2, df=1, 11, p=0.30); therefore, gender was not considered a necessary covariate.
There were no discernible effects of diagnosis on 5-HT
2 binding potential (ANCOVA, F=1.6, df=1, 30, p=0.22) (
Figure 1). The interaction between age and diagnosis was not significant (F=0.1, df=1, 29, p=0.70).
The three subjects with a history of a suicide attempt did not have a 5-HT2 binding potential which was significantly different from that of the healthy subjects (ANCOVA, age as covariant, F=0.6, df=1, 19, p=0.46); however, this subgroup was small.
Given the previous report of an increased right-to-left ratio of prefrontal cortex 2-[
123I]ketanserin uptake in depression
(28), the right-to-left ratio of prefrontal cortex 5-HT
2 binding potential was calculated for each scan. The mean ratio for the depressed subjects was 1.02 (SD=0.03). For the healthy subjects, the ratio was 1.01 (SD=0.02). Depression was not associated with right-to-left prefrontal cortex asymmetry (analysis of variance, F=0.2, df=1, 30, p=0.69).
DISCUSSION
To our knowledge, this is the first investigation of 5-HT
2 receptors in drug-free depressed subjects who have not recently attempted suicide, and we found no evidence for increased 5-HT
2 receptor binding in the prefrontal cortex. We had hypothesized an increase in 5-HT
2 binding potential, which would have been consistent with postmortem studies of depressed suicide victims
(6–
8), but our data do not support this theory.
Unlike postmortem studies of depressed subjects, most of whom ended their lives by suicide, this was a study of depressed patients who had not made recent suicide attempts
(6–
8). Given that increased 5-HT
2 receptor binding in undiagnosed suicide victims has been reported, it appears that increased 5-HT
2 receptor binding is linked to suicide attempts but not depression without suicide attempts
(19–
21).
Our findings replicate in part those reported by D’haenen et al.
(28). They imaged 5-HT
2 receptors in 19 depressed and 10 healthy subjects by means of 2-[
123I]ketanserin SPECT. All of their depressed subjects had been free of psychotropic drugs for 7 days, and 10 of their depressed subjects had been free of antidepressants for at least 3 weeks. As in our study, D’haenen and colleagues found that age was a significant covariate and that no changes in bilateral 5-HT
2 binding were present. In contrast to our results, D’haenen et al. also reported an increase in left-to-right prefrontal cortex 2-[
123I]ketanserin uptake. Their unexpected finding was not hypothesized a priori and is not supported by postmortem data. Neither the current study nor another recent study
(29) confirmed their finding. It is possible that their unreplicated finding represents a subgroup with treatment-refractory illness or the effects of medication withdrawal
(23–
26).
Biver et al.
(29) measured 5-HT
2 binding potential in 22 healthy and eight depressed subjects by means of [
18F]altanserin PET. Using statistical parametric mapping
(45,
46), they detected two marginally significant regions of approximately 4-cm
3 total size in bilateral inferofrontal cortex. These regions reflected decreased 5-HT
2 binding potential in the depressed group. Spatial normalization of statistical parametric mapping has not been validated for [
18F]altanserin
(47). Such localized findings may be diluted in our region of interest. Our results may also differ because of sampling differences: we selected untreated patients, whereas Biver et al. chose patients with depression who were withdrawn from treatment (and may have had treatment-refractory illness). Also, the effect of medication use in their study is not clear; of the eight depressed subjects studied, one took prednisone, four had taken benzodiazepines until 10 days before scanning, and four had taken antidepressants until 3 weeks prior to scanning. Steroids and antidepressants are known to affect 5-HT
2 receptor regulation
(7,
23–
26,
48), and γ-aminobutyric acid receptors influence serotonin release
(49). The mean age of their patient group was 48 years, 16 years older than the mean age of our patients. Six of their eight patients had recurrent major depression, as compared with three of 14 in our study. Their finding may reflect a vulnerability to recurrent major depression rather than the state of depression itself.
Mayberg and colleagues
(50), using PET with [
11C]
N-methylspiperone, also found no increases in 5-HT
2 binding potential in a group of subjects with poststroke depression or dysthymia, as compared with age-matched healthy volunteers. Unlike our study, the theoretical basis for investigating up-regulation of 5-HT
2 receptors was that localized decreases of 5-HT and norepinephrine near an infarct location could cause changes in 5-HT
2 binding potential.
Whenever negative results are found, the sensitivity of the approach to detecting a difference and the expected magnitude of such a difference have to be considered. Our approach is reasonably sensitive to changes in 5-HT
2 receptor binding because it has detected the effects of age
(37,
39,
44), atypical neuroleptic occupancy
(44), and Alzheimer’s disease
(38). Even in the present study group, robust age effects were observed. Postmortem studies of depressed suicide victims report increases in 5-HT
2 receptor binding of approximately 70% within the prefrontal cortex
(6,
7). Thus, if differences in vivo were similar in magnitude to those reported in postmortem studies, we should have had 99% power to detect such change
(51). In fact, our study had 80% power to detect a difference as small as 20%. The present study cannot rule out a change of less than 10%, since it had only limited power (27%) to detect such a small change. A study group of 70 depressed and 70 healthy subjects would be required to detect a 10% change.
The second limitation of our approach is that we measured 5-HT
2 binding potential in a large volume of the prefrontal cortex that included Brodmann’s area 9 and parts of areas 8, 10, 46, and 32. It is conceivable that highly localized regional changes within the prefrontal cortex may have been undetected. Voxel-by-voxel image analysis is available for regional cerebral blood flow data
(45–
47); however, we are not aware of any spatially validated voxel analyses for 5-HT
2 binding potential images.
Other limitations also need to be considered. Rather than determining B
max and K
d individually, we measured the 5-HT
2 binding potential, which is linearly proportional to B
max/K
d. If our patients had an increase in receptor number (i.e., increased B
max) and a decrease in affinity (i.e., increased K
d), then these two changes could have cancelled each other out in 5-HT
2 binding potential. Such an event is highly unlikely because B
max and K
d would have to change in the same direction and by the same quantity. Furthermore, this possibility is not supported by previous reports of elevated 5-HT
2 B
max in drug-free depressed suicide victims
(6,
7), in whom changes in K
d were not found.
Our findings do not rule out a role for prefrontal 5-HT
2 receptors in the treatment of depression. Using [
18F]setoperone PET, Massou et al.
(52) found increased 5-HT
2 binding potential in six depressed subjects who were treated with selective serotonin reuptake inhibitors (SSRIs), as compared with eight untreated depressed subjects. Many tricyclic antidepressants bind to 5-HT
2 receptors, and both tricyclic antidepressants and SSRIs are known to affect 5-HT
2 receptor binding in animal models
(23–
26,
53). It is proposed that 5-HT
2 receptors may mediate antidepressant action through postreceptor cellular targets such as the cAMP response element-binding protein via the stimulation of Ca
2+-dependent protein kinases
(54).
We made a special effort to exclude subjects with a recent history of suicide attempts because it might have confounded the effects of depression. On the other hand, it may be of scientific interest and clinical importance to study patients who have a prominent suicidal history or who have made recent suicide attempts. Elevated 5-HT
2 receptor binding has been well replicated in suicide victims; hence, it is likely that suicidal depressed subjects are different from depressed subjects with regard to 5-HT
2 receptor binding
(6,
7,
19–
21).
In conclusion, we found no increase in 5-HT
2 receptor binding in the prefrontal cortex in unmedicated patients with major depression. This result is in contrast to postmortem studies that showed an increase of 65%–75%
(6,
7). It is still possible that vulnerability to suicide attempts is associated with increased 5-HT
2 receptor binding and that 5-HT
2 receptors may have an important role in the therapeutics of depression, even though a primary role for increased 5-HT
2 receptor density in depression seems unlikely.