Early identification of nonresponders to antipsychotic treatment could prevent unnecessary persistence with ineffectual agents, thereby diminishing risk of adverse events. Reduced risk of adverse events could in turn reduce duration of hospitalization, level of care required, amounts of concomitant medication, and costs of care. Although most studies have examined putative response predictors before initiation of treatment, examination of symptom changes shortly after commencement of treatment may be a more reliable way of predicting response. Findings from a meta-analysis have challenged the belief that onset of action of antipsychotics is delayed by providing evidence for a robust early onset (13.8% reduction in symptom scores after 1 week)
(1) . This result suggests that response during the first week of treatment might provide an indication of how likely patients are to respond later in treatment. In keeping with earlier findings that early symptom changes could be a useful predictor of outcome
(2), Correll et al.
(3) investigated the predictive value of symptom changes 1 week after initiation of treatment. They found that early nonimprovement (<20% reduction in Brief Psychiatric Rating Scale total score at 1 week) predicted nonresponse at 4 weeks in 100% of cases, suggesting that treatment refractoriness may already be identifiable after 1 week.
Using data from a large multinational, randomized, double-blind trial comparing risperidone and haloperidol, we determined time to antipsychotic response in first-episode schizophrenia. We also examined as possible predictors of response previous antipsychotic exposure, duration of untreated psychosis, age, and sex.
Results
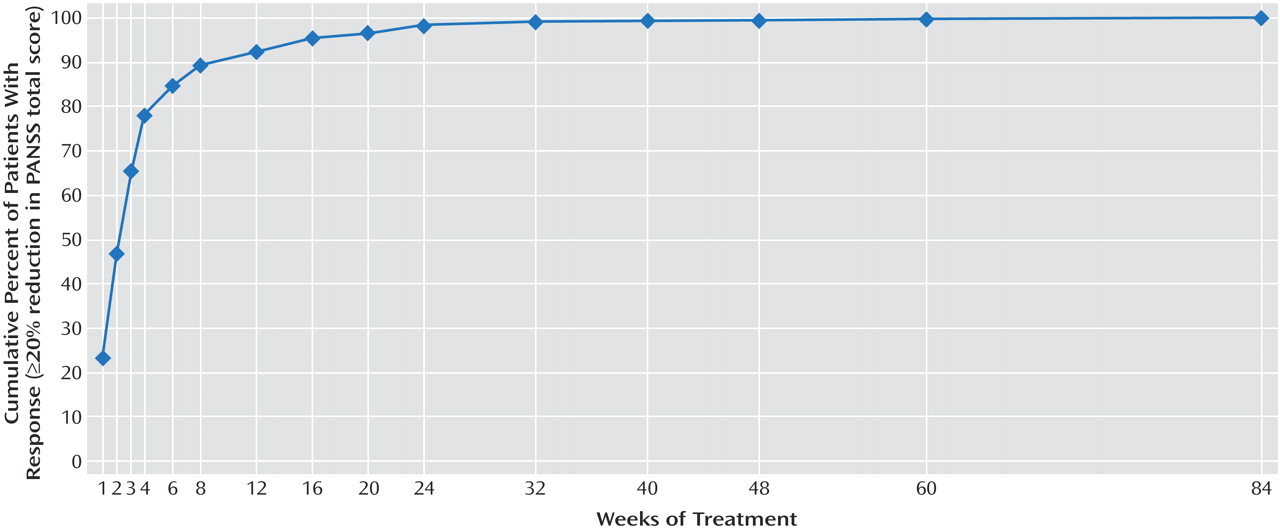
Of the 522 patients, 400 (76.6%) achieved a ≥20% reduction in the PANSS total score. However, time to response varied considerably (
Figure 1 ), with 93 (23.3%) patients achieving response during the first week of treatment, 93 (23.3%) at week 2, 74 (18.5%) at week 3, and 50 (12.5%) at week 4. Ninety (22.5%) of the patients did not respond until
after the fourth week, and of those, 45 (11.5% of the 400 patients with a clinical response) did not respond until
after the eighth week. The doses at times of response were 1 mg/day for 15.5% (N=62) of patients, 2 mg/day for 29.8% (N=119), 3 mg/day for 27.3% (N=109), 4 mg/day for 16.8% (N=67), and more than 4 mg/day for 10.8% (N=43).
Among the 522 patients, improvement of at least 20%, 30%, 40%, and 50%, respectively, on the PANSS total score was achieved by 76.6% (N=400), 63.0% (N=329), 44.8% (N=234), and 27.5% (N=144) of patients. Improvement of at least 20%, 30%, 40%, and 50%, respectively, was achieved by 72.9% (N=381), 59.6% (N=311), 44.6% (N=233), and 27.0% (N=141) of patients on the PANSS negative subscale, by 76.6% (N=400), 62.1% (N=324), 44.6% (N=233), and 28.9% (N=151) of patients on the PANSS general psychopathology subscale, and by 82.8% (N=432), 74.3% (N=388), 63.8% (N=333), and 48.5% (N=253) of patients (the most improvement) on the PANSS positive subscale.
Based on Cox regression analysis, which controlled for baseline PANSS score and study center, patients with previous antipsychotic exposure were less likely to respond (odds ratio=0.77, 95% confidence interval [CI]=0.61–0.97, p<0.03). Age, sex, and study medication were not significantly associated with response. For neuroleptic-naive patients (N=163, N=20 nonresponders), longer duration of untreated psychosis (log transformed) was associated with decreased likelihood of response (odds ratio=0.86, 95% CI=0.76–0.96, p=0.01).
Discussion
The majority of the subjects in the study responded to antipsychotic treatment, in keeping with previously reported favorable response in first-episode schizophrenia
(6) . Our findings provide further evidence that, at least in some patients, onset of antipsychotic action is early—i.e., in the first week of treatment
(1) . However, time to response varied widely, with some responders achieving ≥20% improvement in PANSS total score only after more than 10 weeks of treatment. The median time to response in this group of patients was almost 3 weeks.
Our findings differ from those in a study of multiepisode patients by Correll et al.
(3), as we did not find early nonresponse to be a reliable predictor of later nonresponse. Whereas Correll et al., using a somewhat different approach, found that all patients who failed to achieve 20% improvement after 1 week of treatment also failed to do so after 4 weeks of treatment, we found that 60% (N=256 of 424) of such patients (i.e., patients who had not improved after 1 week of treatment) were nonresponders at week 4. Moreover we found that of nonresponders at week 1 (N=424), 65.3% (N=277) went on to have a clinical response. Correll et al. found that 35% of those who had responded by week 1 were also responders at week 4; we found that 69.4% (N=68 of 98) of such patients were also responders at week 4.
If, as suggested in practice guidelines
(7), treatment trial periods of 1 month or even 6 weeks were used for the subjects in our study, many patients would incorrectly have been regarded as nonresponders. Additional research is indicated, and if similar results emerge for other treatments, then practice guidelines recommending shorter durations of treatment
(7) may need to be revised for first-episode psychosis. A possible explanation for the discrepant findings is that the study by Correll et al.
(3) was of short duration (4 weeks). Also, in our study, a low-dosing strategy was adopted, which could explain slow response in some patients. We consider this explanation unlikely, however, as low doses of both risperidone
(8) and haloperidol
(9) have been shown to be at least as effective as high doses in first-episode psychosis. Our finding that shorter duration of untreated psychosis was a predictor of response is in keeping with previous work
(10,
11) .
The strengths of this study include large number of subjects, the fact that the subjects were first-episode patients, use of standardized assessment and diagnostic criteria, the ability to test treatment response for both a typical and an atypical antipsychotic, and the long treatment period. Flexible dosing design was both an advantage, in that it allowed mimicking of clinical practice, and a limitation, because time to response for a specific dose could not be examined. Future studies should examine the relationship between early symptom reduction and later overall outcome.