Effectiveness and Tolerability
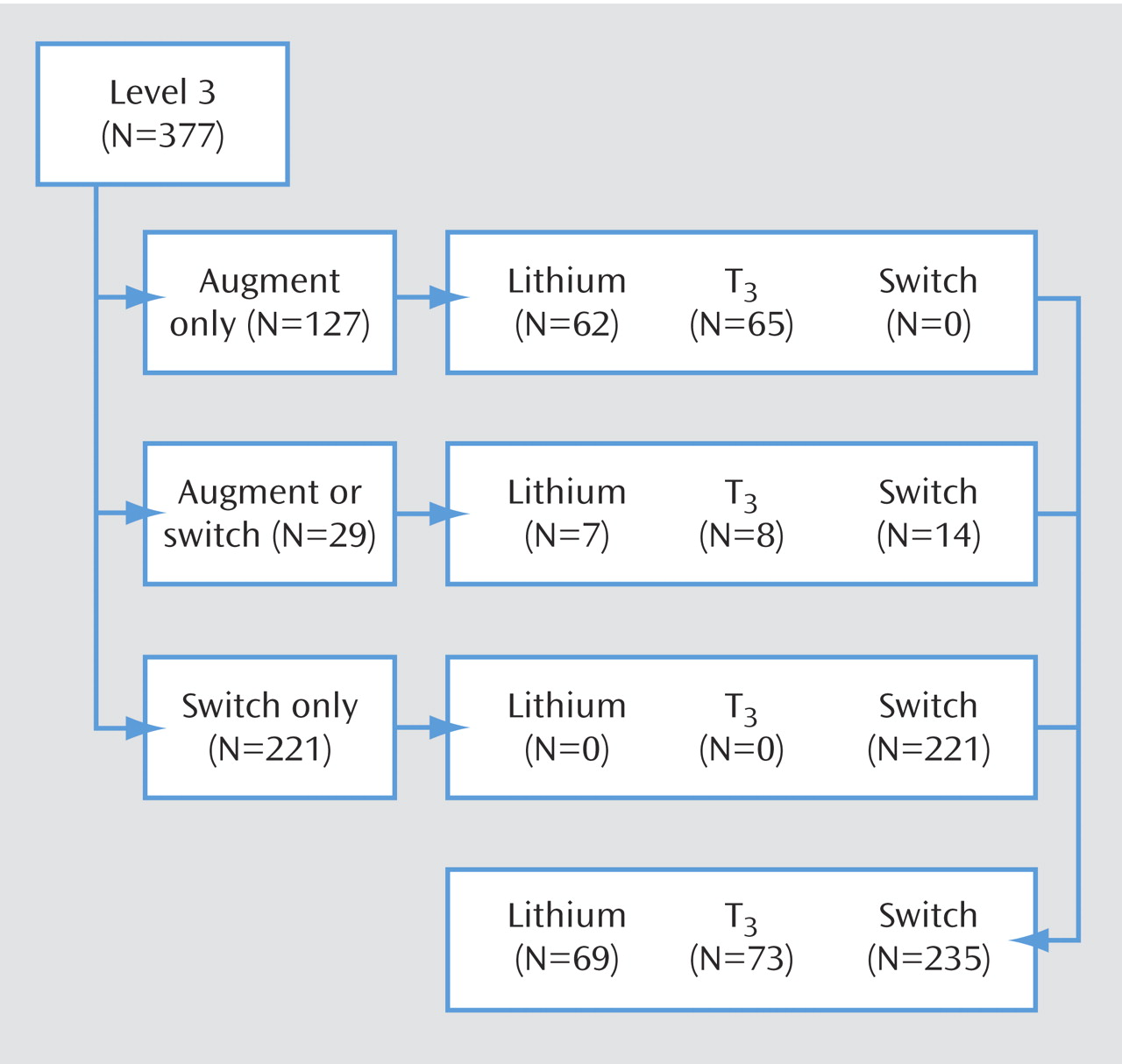
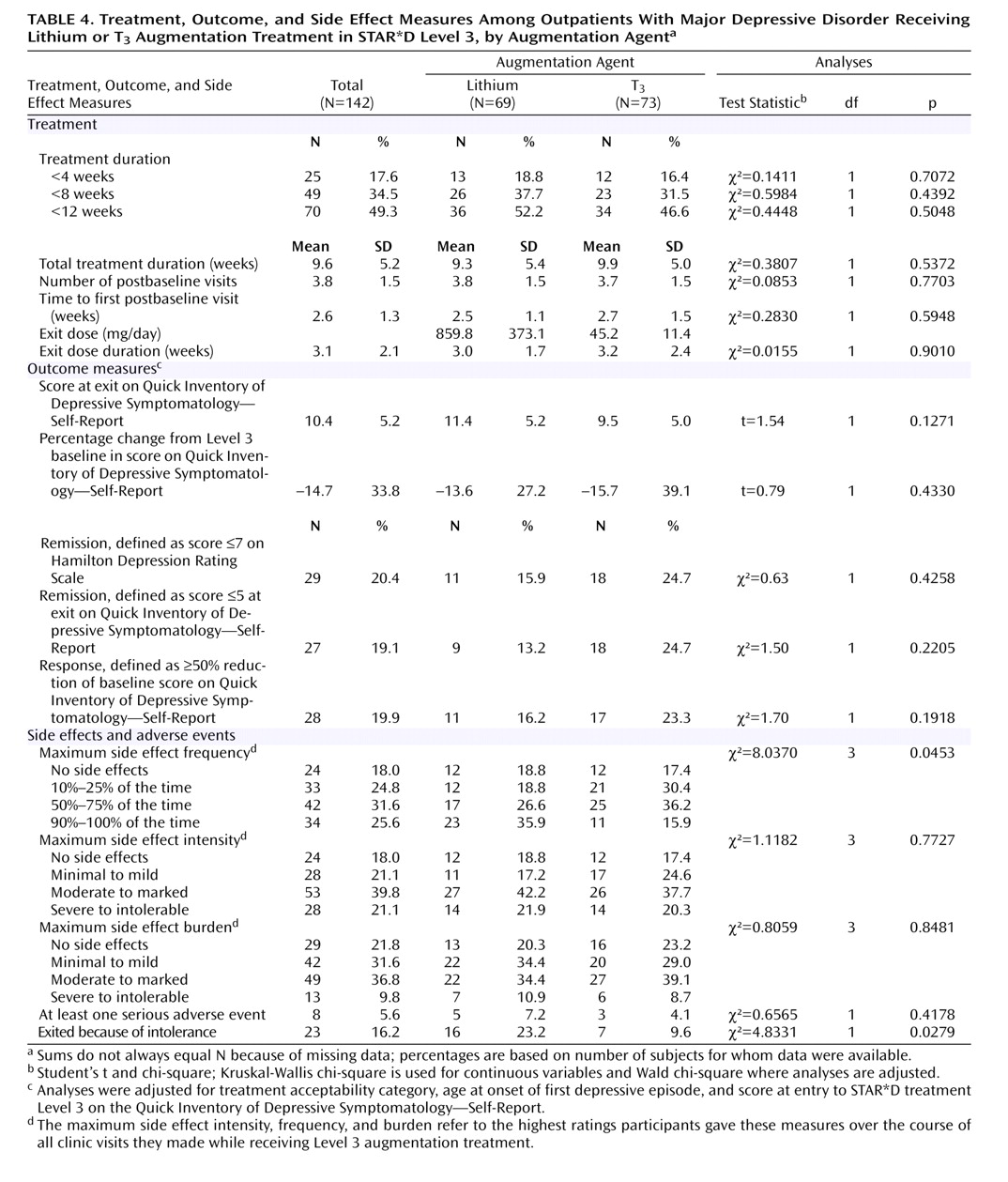
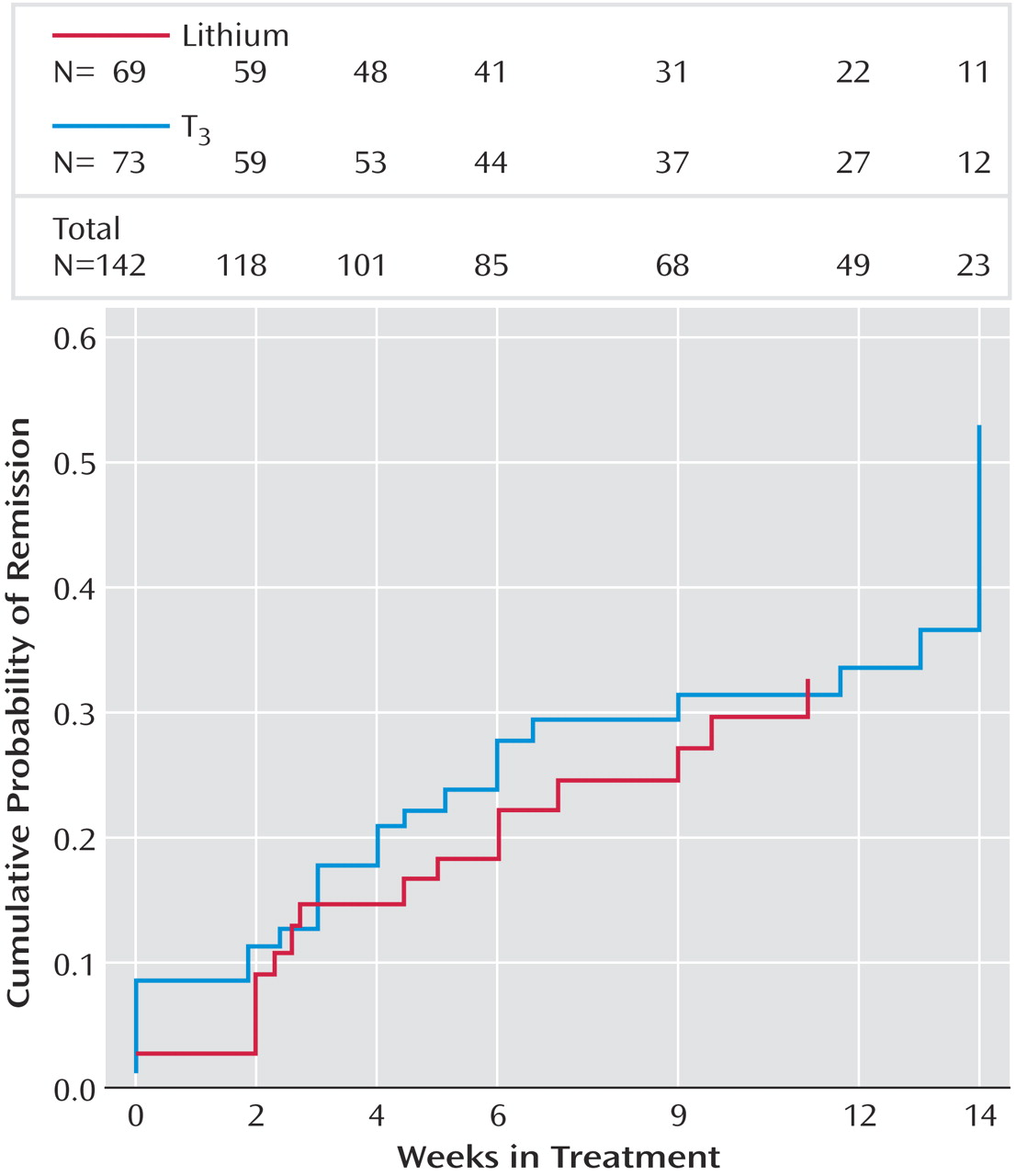
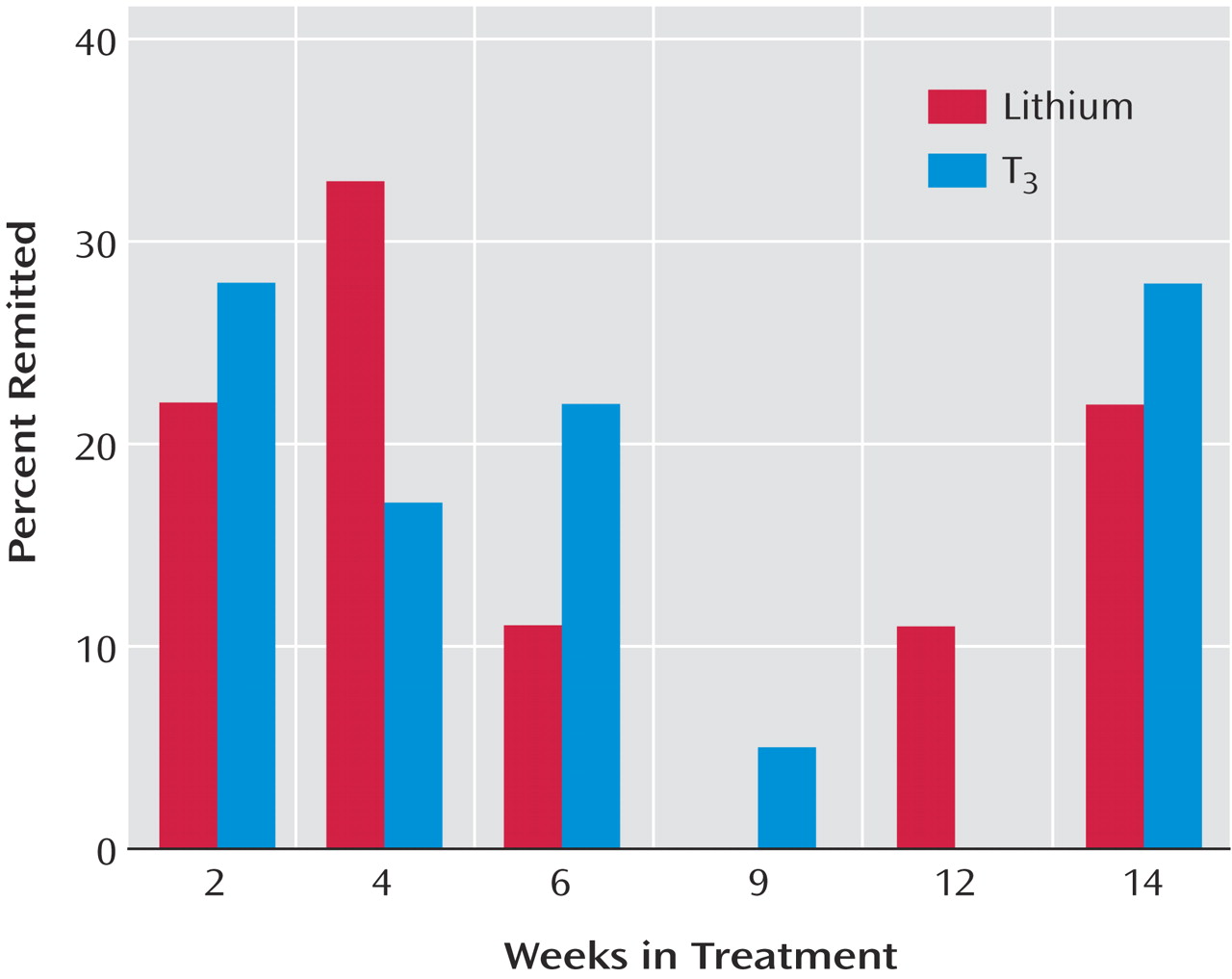
This is the first randomized, controlled effectiveness study to compare lithium and T 3 augmentation for outpatients with nonpsychotic major depressive disorder who had not obtained adequate benefit from two prior medication treatment attempts. Only a modest proportion of participants achieved remission. The two treatment groups did not differ significantly in the proportion who responded or remitted, in time to response or remission, or in exit measures of depressive symptom severity. While no significant differences were observed in the symptom-based outcome measures between the two groups, the group receiving T 3 augmentation consistently had greater proportions of responders and remitters, greater decreases in HAM-D and QIDS-SR scores, and greater toleration of treatment.
The modest remission rates observed with lithium augmentation may have been due to the low doses used as a result of limited toleration of side effects. Nevertheless, these results probably reflect what clinicians can expect with lithium augmentation in actual practice. The doses of T
3 used in this study approximate those used in placebo-controlled trials, and given the drug’s tolerability, they were easily reached. For both lithium and T
3 augmentation, the duration of treatment was not only sufficient, but it was substantially greater those of previously reported augmentation trials of these agents, none of which exceeded 6 weeks
(6,
20,
21) . In this study, the mean duration of augmentation treatment was more than 9 weeks, and hence participants very likely had an adequate exposure to the augmentation agents. The modest remission rates may reflect problems with tolerability of lithium augmentation and the difficult-to-treat nature of many cases of major depression in real-world settings.
The remission rates observed in this study are consistent with those reported in other recent augmentation trials, including lithium augmentation of nortriptyline
(21) and fluoxetine
(8) and T
3 augmentation of fluoxetine
(12) . Compared with results reported by Joffe et al.
(21) in the only prior study comparing augmentation with lithium and T
3 after unsatisfactory antidepressant monotherapy, our results show a substantially lower proportion of participants who experienced a response to treatment. Yet in the Joffe et al. study, both the duration of treatment with antidepressants before augmentation and the duration of augmentation treatment were substantially shorter than in our study. Joffe et al. studied 50 participants who received 2 weeks of augmentation with lithium (N=17), T
3 (N=17), or placebo (N=16) after 5 weeks of prospective treatment with either imipramine or desipramine. Although remission rates were not reported, response rates were around 60% for both active augmentation agents and 19% for placebo. One possible explanation for the greater percentage of responders in the Joffe et al. study is that the augmentation may have sped up response to a brief course of antidepressant therapy. It may be, too, that augmentation strategies are more efficacious in treatment with tricyclic antidepressants
(8) . A third possibility is that participants in our study, who had already undergone two prior medication trials without achieving remission, had more difficult-to-treat forms of depression. Joffe et al. examined the effects of augmentation after a single, relatively brief trial of antidepressant monotherapy. In an earlier STAR*D report
(3), for participants who had gone through one medication trial (treatment Level 1) without achieving remission and then underwent augmentation with bupropion or buspirone (Level 2), remission rates and response rates were both around 30%
(3) .
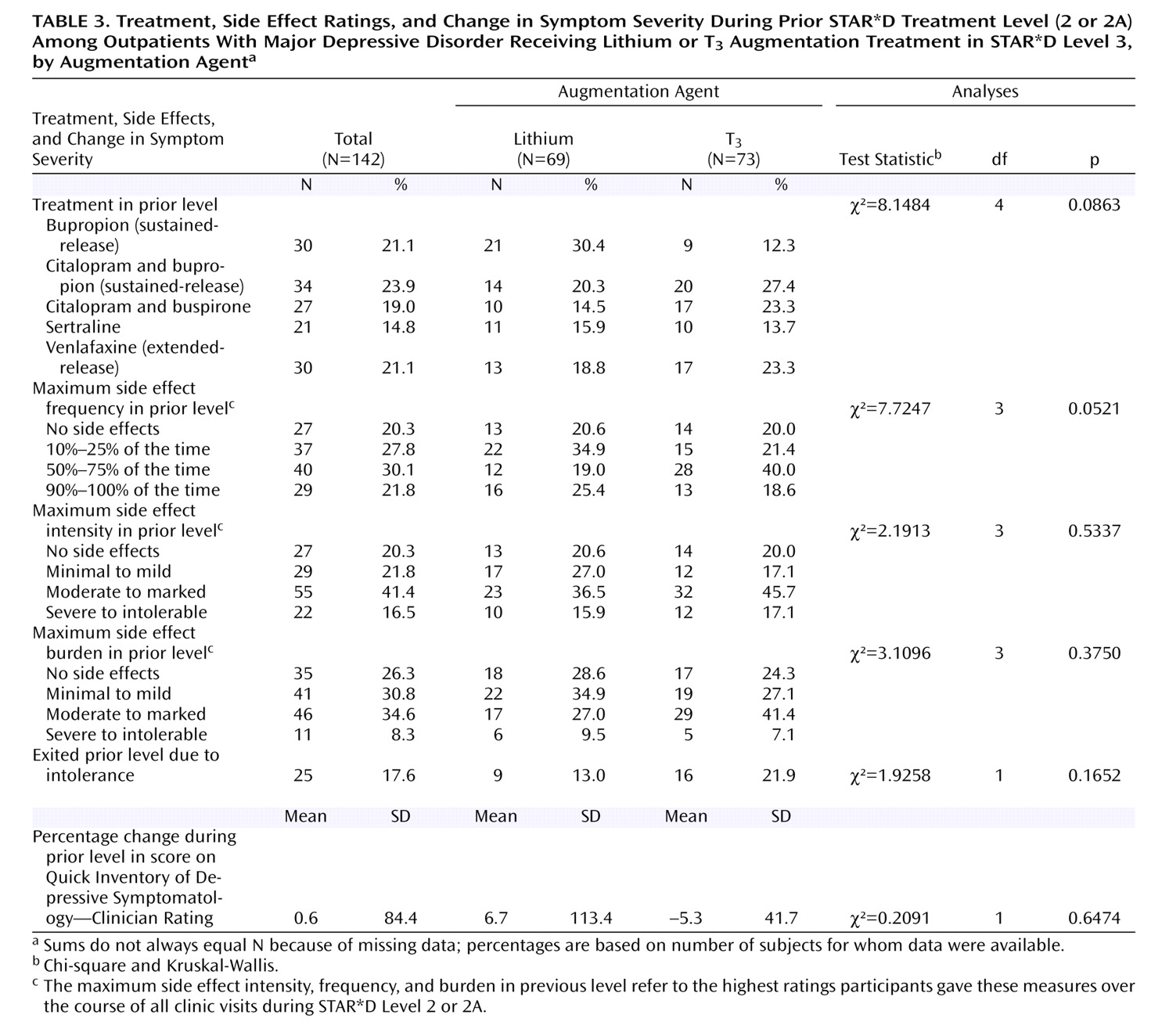
The T 3 group appeared to tolerate the augmentation better than the lithium group. Almost twice as many participants in the lithium group exited augmentation treatment because of side effects. Few in either group had serious adverse events. Given the greater volume of evidence for lithium augmentation and the paucity of randomized trials of T 3 augmentation with the newer generation of antidepressants, the results we observed with T 3 augmentation were better than expected. Overall, if a clinician has a choice between lithium and T 3 augmentation, these results suggest slight advantages with T 3, especially for patients who have already had two unsuccessful treatments.
Strengths and Limitations
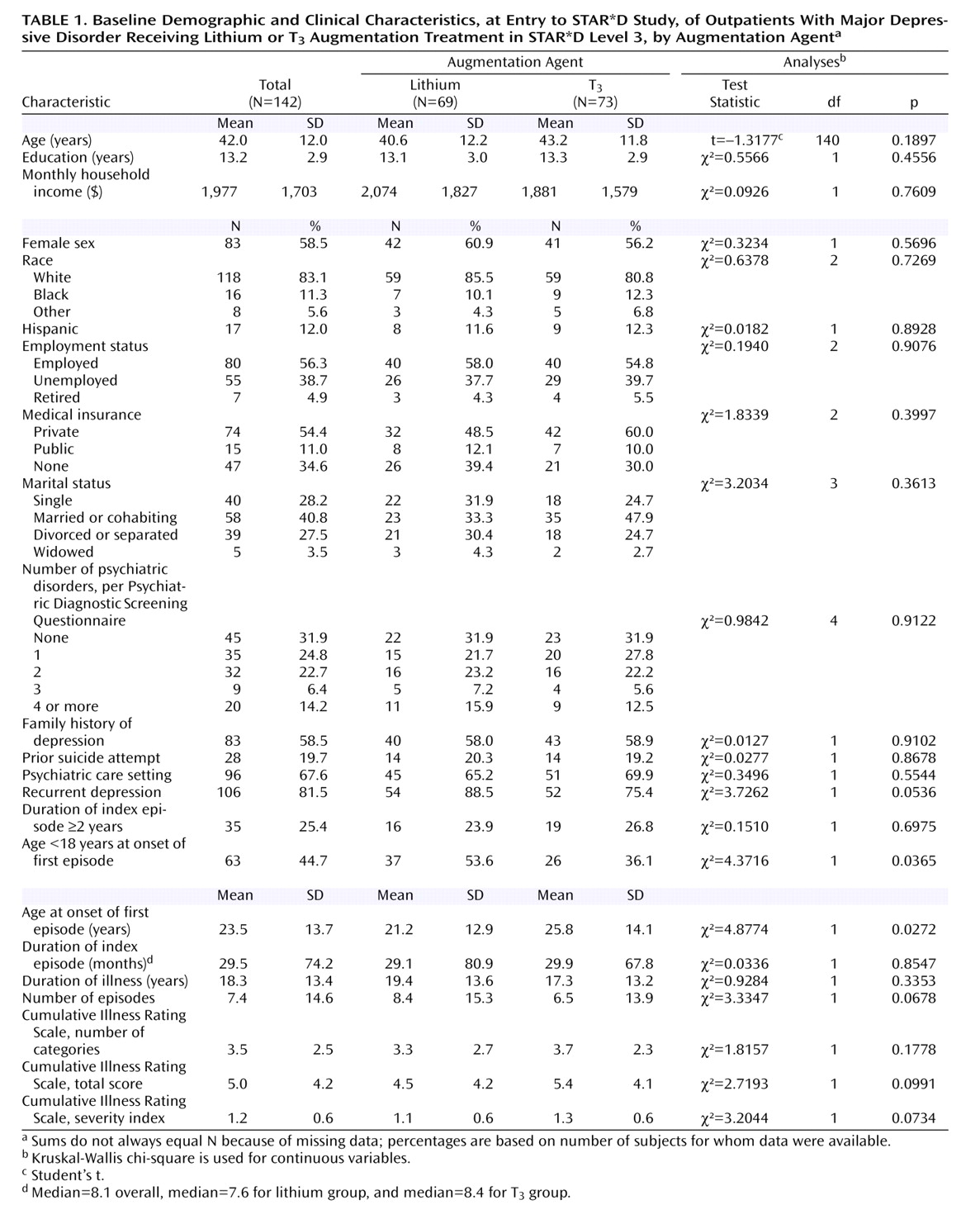
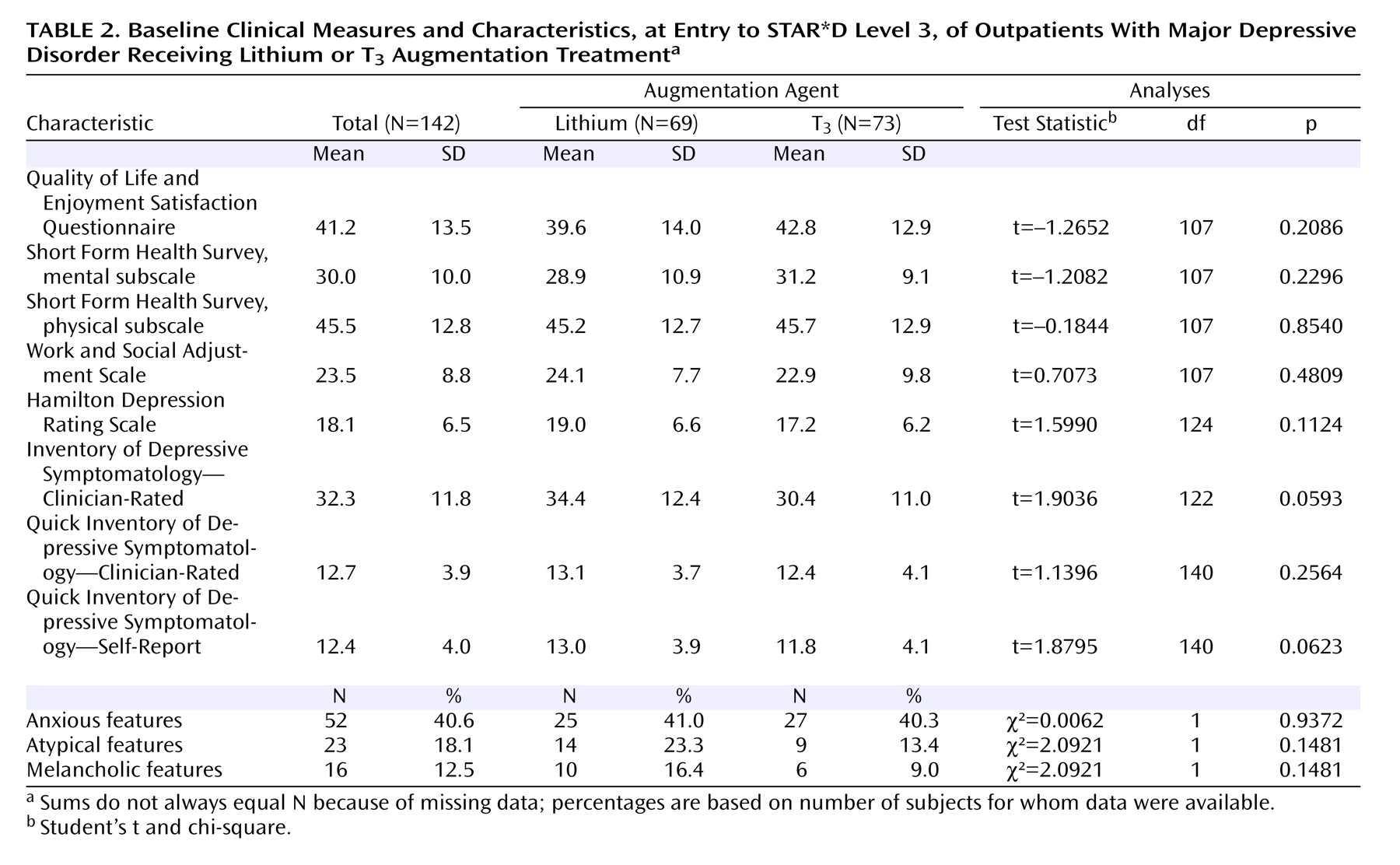
Among the strengths of this study are that it was conducted in representative real-world practices with patients who presented for care—they were not recruited through advertising. Participants had undergone two prospectively administered medication trials that had failed to bring them to remission. This effectiveness design enhances the ecological validity and the generalizability of the study’s results. Medication treatment was open-label, and clinicians used evidence-based guidelines to optimize dose and duration of treatment. The primary outcome measures were collected by assessors who were blind to participants’ treatments.
This study also had several limitations. First, it did not have the statistical power to reliably detect small differences in remission rates between the augmentation therapies. Second, we did not systematically assess laboratory indices, including pretreatment assessment of thyroid function and serial monitoring of lithium levels. Third, we used open-label administration of the augmentation therapies. Fourth, the study design did not include a placebo control group—a particularly noteworthy limitation, given the low remission rates: it is not possible to confirm that either augmentation therapy was more effective than supportive clinical management along with ongoing antidepressant therapy. Finally, participants in the lithium augmentation group took relatively low doses because of intolerable side effects, and as a result they had minimal blood lithium levels. This limitation leaves open the question of whether keeping the doses of lithium small limits its effectiveness for augmentation
(6,
7) . Yet, as noted earlier, patients in this study took the highest tolerable doses, reflecting the reality of prescribing lithium to patients with major depressive disorder who present for care in everyday practice.