Tardive dyskinesia consists of abnormal, involuntary, irregular choreoathetoid movements of the muscles of the head, limbs, and trunk and often is irreversible
(1). Several different suppressive agents have been tried with limited success
(2). Serotonin (5-HT) modulates striatal dopamine release and could influence dyskinetic movements
(2,
3). Excess dopamine may amplify 5-HT release in the brain. Improvement of tardive dyskinesia with cyproheptadine, a 5-HT antagonist, has been noted
(4). Because ondansetron is a selective 5-HT
3 antagonist used to prevent nausea and vomiting in cancer patients
(5), we examined its efficacy, safety, and tolerability in a short-term, open-label trial in patients with tardive dyskinesia.
METHOD
We studied 20 inpatients with DSM-IV-diagnosed schizophrenia. Five of the patients were men, and 15 were women; their mean age was 69.8 years (range=42–81, SD=10.1). All of the patients met Research Diagnostic Criteria for at least mild tardive dyskinesia, and all had been experiencing the dyskinesia for at least 6 months. The mean duration of tardive dyskinesia was 9.8 years (range=1–30 years). All patients had stable psychopathology and had been receiving a stable psychotropic drug regimen for at least 6 months before study entry. Tardive dyskinesia had been stable for at least 3 months before entry to the study as measured by Abnormal Involuntary Movement Scale (AIMS)
(6) scores. All patients had no serious medical or neurological disease, and all provided written informed consent.
Oral ondansetron was administered in a dose of 8 mg/day at 8:00 a.m. in the first week of the study; a dose of 12 mg/day (8 mg at 8:00 a.m. and 4 mg at 8:00 p.m.) was administered during weeks 2–12. Doses of all psychotropic medications were kept unchanged throughout the study.
Tardive dyskinesia was assessed with the AIMS. Patients were rated at baseline and after 4, 8, and 12 weeks. Severity of movements was rated on a scale of 0 to 4. Assessments were videotaped and then reviewed in random order at the completion of the study by four blind raters (P.S., T.M., H.S., and N.G.) who scored each session; their scores were then averaged for each subject and session. The intraclass correlation coefficient (ICC)
(7) for total AIMS scores, based on 25 randomly selected ratings, was 0.83 (df=2, 15, p=0.001).
Psychosis was assessed by two trained raters using the Positive and Negative Syndrome Scale
(8) and the severity subscale of the Clinical Global Impression scale (CGI)
(9) at baseline and after 12 weeks. For the Positive and Negative Syndrome Scale, ICC=0.79 (df=2, 19, p=0.002); for the CGI severity subscale, ICC=0.86 (df=3, 18, p<0.001).
Total AIMS scores at baseline and after 4, 8, and 12 weeks of treatment were analyzed by using one-way analysis of variance (ANOVA) with repeated measures. Total Positive and Negative Syndrome Scale as well as CGI scores at baseline and after 12 weeks were compared by using ANOVA with repeated measures.
RESULTS
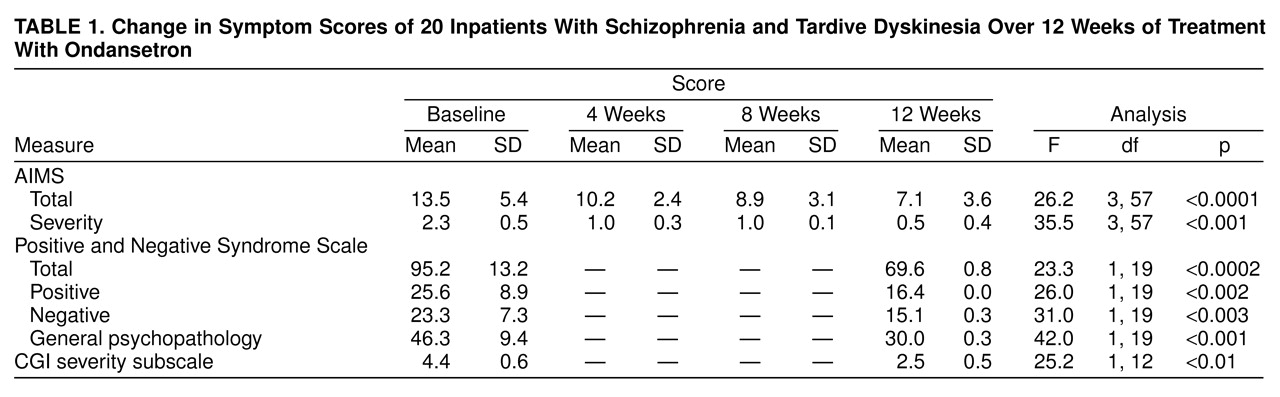
A significant overall reduction of AIMS total scores after treatment with ondansetron was noted (
table 1). The mean severity score decreased significantly during the study (
table 1). A clinically significant improvement in the severity of tardive dyskinesia, defined as a reduction of 75% or greater, occurred in 13 patients. The mean reduction in severity score was 78%.
The total AIMS scores of the 10 patients who had experienced tardive dyskinesia for 10 years or less (mean=9.6, SD=4.0) was 25% lower than the ratings of 10 patients who had experienced tardive dyskinesia for more than 10 years (mean=12.8, SD=4.2) (F=8.2, df=3, 54, p<0.01). AIMS scores were not influenced by the patients’ age (F=0.17, df=3, 54, p=0.91) or sex (F=0.15, df=3, 54, p=0.93).
The patients’ total scores on the Positive and Negative Syndrome Scale showed a significant decrease from baseline to week 12 (
table 1). There were significant improvements in scores on the Positive and Negative Syndrome Scale subscales as well as on the CGI severity subscale from baseline to week 12 (
table 1).
DISCUSSION
These findings show that ondansetron is beneficial in treating tardive dyskinesia as well as psychotic symptoms in patients with schizophrenia. The drug was generally very well tolerated and caused no major side effects. A literature search did not reveal any effects of ondansetron on the metabolism of the other medications.
The involvement of the serotonin system in modulating extrapyramidal motor effects is suggested by the absence of extrapyramidal symptoms with the use of clozapine, which has a strong affinity to the 5-HT
1c, 5-HT
2 receptor
(10,
11), a high ratio of 5-HT
2-to-D
2 blockade, and 5-HT
3 antagonism
(12). The serotonin system interacts directly with dopaminergic neurons in the substantia nigra and ventral tegmental area
(13).
Ondansetron and other 5-HT
3 antagonists have been shown to block motor hyperactivity induced by central administration of amphetamine and dopamine
(14). The ability of dopamine to inhibit the firing of medial prefrontal neurons can be potentiated by 5-HT
3 agonists and is blocked by 5-HT
3 antagonists
(15). It has been proposed that the 5-HT
3 antagonist action of clozapine could contribute to its antipsychotic action by means of this mechanism
(16). These observations suggest that 5-HT
3 antagonists can restore and maintain “normal” dopamine function in patients with schizophrenia and may produce an antipsychotic effect without inducing sedation or extrapyramidal symptoms.
Ondansetron appears to have an antipsychotic effect in acute schizophrenia at lower doses (4–12 mg/day)
(17); a double-blind, placebo-controlled study (unpublished 1991 paper by H.Y. Meltzer), however, indicated no clinical improvement. An improvement in psychotic symptoms in patients with Parkinson’s disease has been reported
(18).
In conclusion, our results suggest that ondansetron may be beneficial for tardive dyskinesia and for psychosis. Controlled double-blind studies for extended periods are required to verify our findings.