Vitamin B
12 (cobalamin) and folate are essential in several metabolic pathways in the central nervous system, and their metabolism is intimately connected
(1,
2). Both are involved in single-carbon transfer (methylation) reactions necessary for the production of monoamine neurotransmitters, phospholipids, and nucleotides. A deficiency of either vitamin may cause an impaired methylation in the central nervous system and may result in neurological and/or psychiatric disease that becomes irreversible if not treated properly
(3,
4). This connection is supported by findings that psychiatric patients, especially depressed patients, frequently are found to have abnormalities in vitamin B
12 and folate status
(5–
7). Furthermore, both folate and vitamin B
12 replacement therapy in patients with major depression appear to produce substantial affective improvements
(8–
12). However, since the studies thus far have been restricted to psychiatric patients, it is unknown whether vitamin B
12 and folate deficiencies affect depressed mood in the general, community-dwelling population.
Low serum vitamin B
12 and folate levels are not very specific and have low sensitivity in diagnosing tissue deficiency
(13,
14). Therefore, in the present study the definitions of vitamin B
12 and folate deficiency were based on both low levels of these vitamins and high levels of the specific metabolites methylmalonic acid and total homocysteine, respectively
(15–
17). In the present study we examined whether these assessments of vitamin B
12 and folate deficiency were associated with depression in a community-dwelling, representative sample of disabled older women.
Results
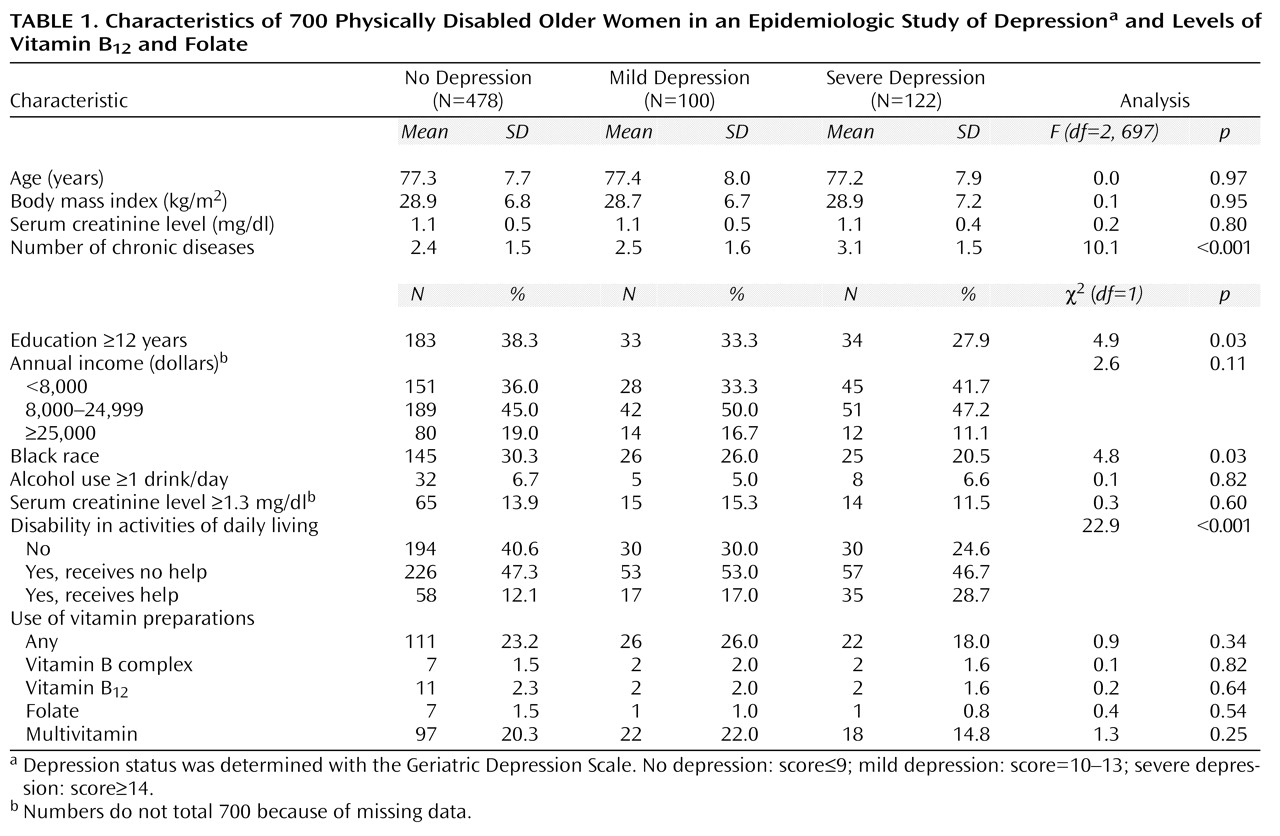
The mean age of the study sample was 77.3 years (range=65–100, SD=7.8), 28.0% were black, and 64.2% had less than 12 years of education. Of the 700 participants, 478 (68.3%) were not depressed, 100 (14.3%) were mildly depressed, and 122 (17.4%) were severely depressed.
Table 1 shows the demographic characteristics, disease status, and multivitamin use of the study sample according to depression status. Compared to the nondepressed subjects, the depressed women had less education, were more likely to be white, and had more chronic diseases and disability in activities of daily living. No differences were found with respect to age, income, body mass index, alcohol use, and use of vitamin preparations.
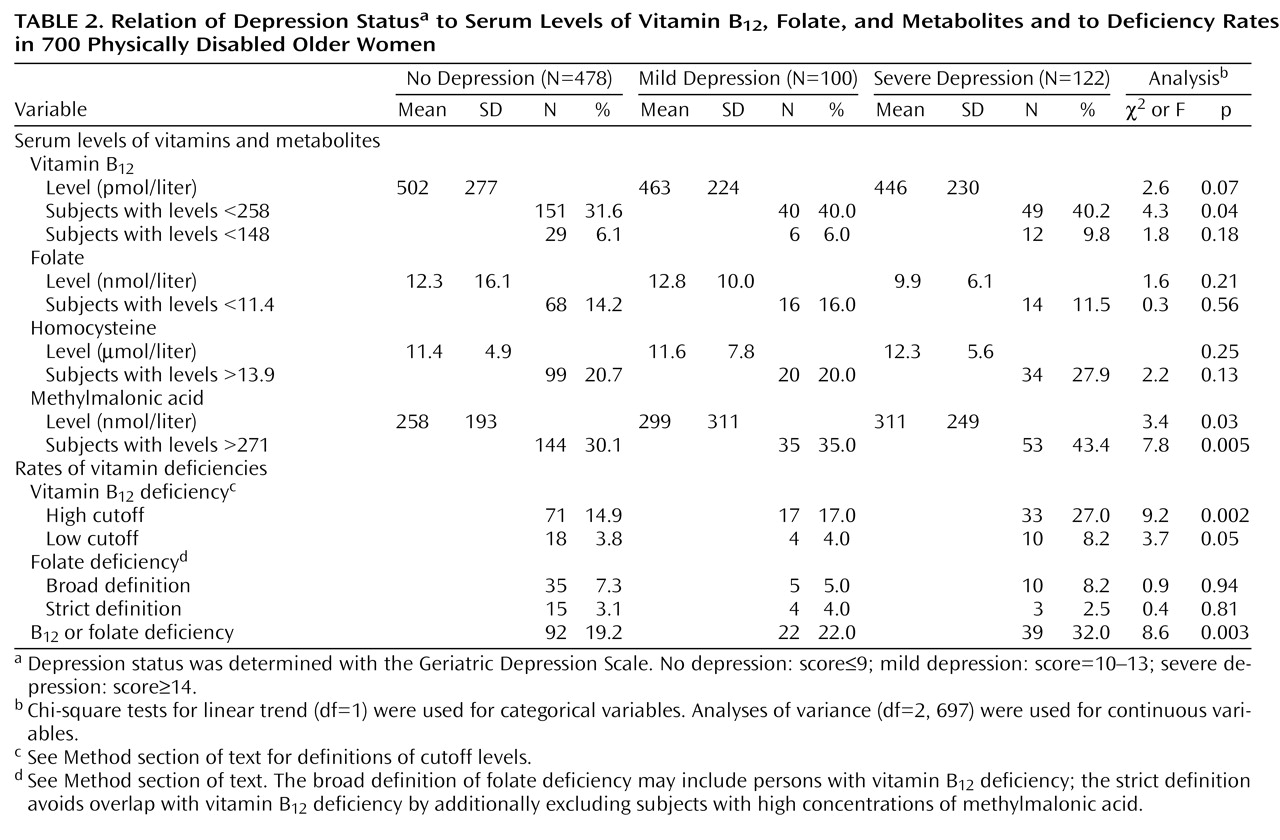
The prevalence of vitamin B12 deficiency defined according to the high cutoff (i.e., serum vitamin B12 level less than 258 pmol/liter plus an elevated serum methylmalonic acid level) was 17.3% (N=121). Of these women, 32 (4.6% of total sample) qualified for the lower, conventional cutoff for vitamin B12 deficiency, that is, a serum vitamin B12 level below 148 pmol/liter plus an elevated serum methylmalonic acid level. Folate deficiency was found in 7.1% (N=50) of the participants. According to the more specific definition (which avoids overlap with vitamin B12 deficiency), 3.1% (N=22) had folate deficiency. Only 18 (2.6 %) of the women had both vitamin B12 and folate deficiency.
Table 2 shows the serum vitamin and metabolite concentrations and the vitamin deficiency prevalences by depression status. The mildly and severely depressed women tended to have lower vitamin B
12 levels than the nondepressed women, but this difference was not statistically significant. Forty percent of the depressed women scored below the vitamin B
12 cutoff of 258 pmol/liter, which was significantly higher than the 31.6% among the nondepressed. Even more profound differences between the nondepressed and depressed subjects were found for the serum methylmalonic acid concentration. Of the severely depressed women, 43.4% had elevated methylmalonic acid levels, whereas the rates were 35.0% and 30.1% among the mildly depressed and nondepressed subjects, respectively. Serum folate and homocysteine concentrations did not differ according to depression status.
Vitamin B
12 deficiency was present significantly more often among the severely depressed and mildly depressed subjects than among the nondepressed women (
Table 2). This pattern was also observed when the low cutoff was used to define vitamin B
12 deficiency. Folate deficiency, defined by either the broad or the strict criteria, was not associated with depression status. Other serum metabolites related to vitamin B
12 and/or folate metabolism were assayed, and there were no significant differences in methionine, methylglycine, dimethylglycine, cystathionine, and 2-methylcitric acid across depression status.
The mean corpuscular volume did not significantly differ between the subjects with a high-cutoff vitamin B12 deficiency (93.5 fl, SD=7.5) and those without a deficiency (93.3 fl, SD=6.5). Likewise, serum hematocrit levels did not significantly differ between the subjects with vitamin B12 deficiency (mean=39.9 mg/dl, SD=4.7) and nondeficient persons (mean=39.9 mg/dl, SD=4.2). Anemia (defined as a serum hematocrit less than 35.0 mg/dl) was somewhat more common in the subjects with vitamin B12 deficiency (17.4%) than in nondeficient subjects (11.0%) (c2=3.9, df=1, p=0.05). Anemia was not associated with depression status (c2=1.7, df=1, p=0.40).
As compared to those without a high-cutoff vitamin B12 deficiency, the deficient subjects were more likely to have congestive heart failure (16.5% versus 9.3%) (c2=5.5, df=1, p=0.02) or cancer (21.5% versus 14.2%) (c2=4.1, df=1, p=0.04) but less likely to have diabetes (8.3% versus 19.2%) (c2=8.3, df=1, p=0.004). Multivariate analyses were subsequently adjusted for the presence of these three diseases. The prevalences of other diseases (e.g., disc disease, spinal stenosis, arthritis, myocardial infarction, angina, pulmonary disease, and stroke) were not significantly associated with vitamin B12 deficiency. Also, there was no association between vitamin B12 deficiency and cognitive function. The percentage of persons with scores on the Mini-Mental State between 18 (study inclusion criterion) and 23 (indicative of mild cognitive impairment) did not significantly differ (c2=0.0, df=1, p=0.84) between those with vitamin B12 deficiency (17.4%) and nondeficient persons (16.6%).
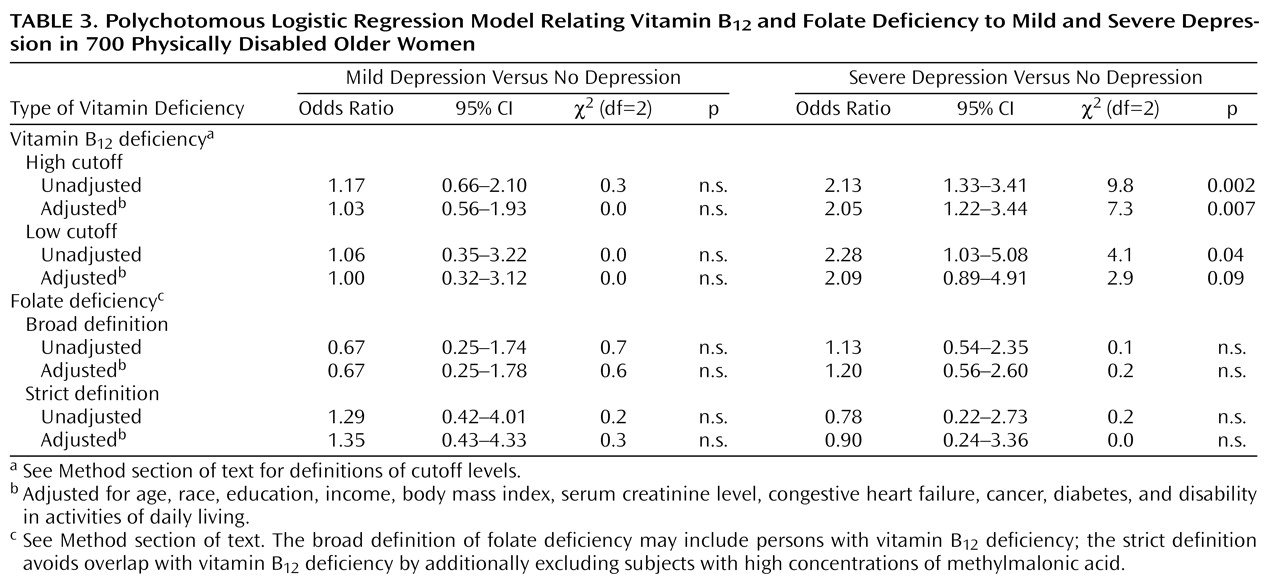
Polychotomous logistic regression models were calculated to assess the associations of vitamin B
12 deficiency and folate deficiency with the three-category depression variable (
Table 3). Adjustment was made for age, race, education, income, body mass index, serum creatinine level, disability in activities of daily living, and the presence of diabetes, cancer, and congestive heart failure. For mild depression, no associations with vitamin B
12 or folate deficiency were found. However, for severe depression, strong associations with vitamin B
12 deficiency were found. Women with a high-cutoff vitamin B
12 deficiency were 2.13 times as likely to be severely depressed. After adjustment for all covariates, the odds ratio became 2.05. When we used the more stringent, low-cutoff definition of vitamin B
12 deficiency, the odds ratios were somewhat higher (unadjusted odds ratio=2.28, adjusted odds ratio=2.09). Folate deficiency was not associated with severe depression.
Discussion
To our knowledge, this is the first study examining the relationship between vitamin B12 deficiency and depression in a community-based population. We found that community-dwelling older physically disabled women with metabolically significant vitamin B12 deficiency had a risk of depression that was more than twice as high as that of women without vitamin B12 deficiency. The higher risk was apparent for severe depression but not for mild depression. No association with depression was found for metabolically significant folate deficiency.
If vitamin B
12 deficiency is truly associated with depression, what are the potential mechanisms? Vitamin B
12 deficiency significantly reduces the reactions promoted by two B
12-dependent enzymes:
l-methylmalonyl-CoA mutase and methionine synthase. There is a resulting accumulation of methylmalonic acid and homocysteine, respectively. The serum vitamin B
12 levels in our sample were indeed strongly, inversely correlated with serum levels of homocysteine and methylmalonic acid
(21). Various neurotoxic mechanisms involving the homocysteine metabolism pathway have been suggested; these include a buildup of
S-adenosylhomocysteine and an increased metabolism of homocysteic acid that may become neurotoxic through activation of
N-methyl-
d-aspartate receptors
(26). However, the association between homocysteine and depression was not very strong in our study, whereas the association with the other metabolite, methylmalonic acid, was markedly stronger: the depressed subjects had significantly higher methylmalonic acid levels than the nondepressed subjects. Methylmalonic acid levels can be elevated in subjects with renal insufficiency, but this did not play a role in our study since the serum creatinine levels and the proportions with elevated creatinine levels were similar in the depressed and nondepressed subjects. To our knowledge, detrimental effects on mood due to actions in the methylmalonic acid metabolism pathway have not been extensively examined and described before. Our findings should encourage further research efforts in this area.
Some other psychobiological explanations for the link between vitamin B
12 deficiency and depression have been suggested as well. Vitamin B
12 is required for the synthesis of
S-adenosylmethionine, which is the major methyl donor in many important methylation reactions in the brain. Since
S-adenosylmethionine has antidepressant properties
(27), inhibited synthesis may cause depression. However, in our study,
N-methylglycine and methionine concentrations, which should be low in the case of
S-adenosylmethionine deficiency, were normal in women who were severely depressed (mean=1.5 mmol/liter, SD=5.9, and mean=20.8 mmol/liter, SD=0.6, respectively) and were not significantly different from the levels in the subjects with no or mild depression. Consequently, it is not very likely that the effect of vitamin B
12 deficiency on depression operates through inhibited
S-adenosylmethionine synthesis. Another explanation may be that vitamin B
12 deficiency affects serotonin and catecholamine synthesis, which may result in depressive illnesses
(27,
28). Finally, vitamin B
12 deficiency has been shown to cause demyelination of the spinal cord and the brain
(4,
11) and consequently may result in neuropsychiatric disorders.
Some alternative, nonpsychobiological explanations should be given as well. Depression itself could cause low vitamin B
12 levels through decreased appetite and resultant decreased food intake
(29). This is unlikely in our sample, as body mass index and the percentage of subjects who reported weight loss during the previous year (23.8%) did not differ across depression status. Also, serum folate level, which is sensitive to dietary intake, did not differ across depression groups. However, we do not have information on specific nutrient intake in this study. In addition, malabsorption of vitamins and increased utilization of vitamin B
12 among depressed patients have been suggested to play roles
(30). These possibilities cannot be directly addressed by our data and require further examination. It has also been suggested that estrogen use has an effect on vitamin B
12 level. However, the relation of estrogen use and vitamin B
12 level was examined in a study of elderly women by Carmel et al.
(31), and no link was found. Also, in our sample, estrogen use (by 9.3% of the subjects) was not associated with vitamin B
12 level or with depression status, indicating that estrogen use did not play any role in the link between vitamin B
12 deficiency and depression. Finally, our results could be explained by serious undiagnosed illnesses leading to both malnutrition and depression. This is unlikely, however, since the Women’s Health and Aging Study used a very rich source of information concerning diseases, disability, and health status. Adjustment for these variables did not affect the findings.
We did not find an association between depression and folate deficiency. This result is inconsistent with findings from some earlier studies of depressed psychiatric inpatients, in which there was an inverse relationship between folate status and severity of depression: depressed patients with folate in the deficient range had more severe depression than those with folate in the normal range
(5,
6). The discrepancy between our and others’ results may be due to the fact that the earlier studies used only serum folate level to diagnose folate deficiency and did not use serum metabolites to confirm the true presence. Serum folate levels are rapidly affected by fluctuations in daily dietary intake and may obscure the true folate status. Consequently, associations between depression and serum folate level that are not metabolically confirmed may be more likely to be due to decreased appetite and lower resultant food intake in the most severely depressed patients. Further, the subjects of our study were members of the community-dwelling older population, whereas earlier studies all focused on depressed patients in institutions. The prevalence of folate deficiency in our community-dwelling sample was lower than prevalences found in studies of depressed psychiatric inpatients, even though those studies used lower cutoffs for serum folate to define deficiency. Although the severity of depression appears to be associated with abnormally low folate levels among patients with a psychiatric diagnosis of depression, our study suggests that an association between folate deficiency and depression is absent in the population of community-dwelling older disabled women.
Our study had some limitations. The analyses presented here are cross-sectional; thus, cause-and-effect relationships cannot be proven. Longitudinal data are necessary to further examine the causal pathway in the link between vitamin B12 deficiency and depression. Other factors—such as the duration of the vitamin deficiency, the duration of the depressive symptoms, associated metabolic disorders, and genetic predisposition—may also determine whether an affective disorder will occur. We did not have data about these aspects. Finally, our results apply to disabled women only. Exploration of possible sex differences and generalization of our findings to healthier populations will be necessary in further studies.
The prevalence of depression in our sample was high (31.7%) and higher than in similarly aged samples of (less disabled) women (around 20%)
(32). Vitamin B
12 deficiency is rather common among older persons. In our study sample of physically disabled older women, 17.3% had a confirmed tissue deficiency of vitamin B
12, which is slightly higher than the 12% to 15% reported in other (less disabled) older populations (33, 34). In line with findings in other studies
(8,
21), our data showed that only a small proportion (17.4%) of the B
12-deficient subjects had megaloblastic anemia. Clinicians and other health care providers need to be aware of the high prevalence of vitamin B
12 deficiency in disabled older women, and they need to screen and treat appropriately, irrespective of the presence of anemia. Our findings show, apparently for the first time, that vitamin B
12 deficiency and depression are associated in a community-based population of disabled older women.