The hepatitis C virus is a major public health problem. Although the incidence of new infection is declining, a large asymptomatic population with chronic hepatitis C exists. Over the next 10–20 years many of these patients will develop clinical symptoms. It is estimated that 4 million Americans are currently infected by the virus, and in 2000 more people will die from hepatitis C than from AIDS. Management of chronic hepatitis C infection involves monitoring liver function tests, liver biopsy, and education regarding the transmission and progression of the disease. Treatment with interferon alpha (IFN-α) is reserved for the patients who are likely to progress to cirrhosis.
Neuropsychiatric symptoms are widely reported in association with both hepatitis C and IFN-α treatment. Furthermore, hepatitis C disproportionately infects psychiatric patients. Fearing psychiatric vulnerability, practitioners may withhold IFN-α inappropriately. It is important that psychiatrists become familiar with hepatitis C infection and the psychiatric management of its complications.
In this article we examine the available studies regarding neuropsychiatric symptoms in chronic hepatitis C infection and IFN-α treatment. Studies were identified by computerized searches, and further references were obtained from bibliographies of the reviewed articles. We first describe the epidemiology and natural history of the hepatitis C virus, treatment with IFN-α, and the associated neuropsychiatric symptoms. We then review the mechanisms of interferon-associated neuropsychiatric symptoms, with particular attention to depressive symptoms. Finally, we discuss treatment and future research directions.
CLINICAL SPECTRUM AND NATURAL HISTORY OF HEPATITIS C
Most acute infections with the hepatitis C virus are asymptomatic, despite elevations in liver enzyme levels. Approximately 30% of the people with newly acquired hepatitis C infection develop symptoms, such as malaise, loss of appetite, and jaundice. Only rarely does acute hepatitis C cause fulminant liver failure. The acute episode resolves in approximately 15% of patients, while the rest develop chronic hepatitis
(1).
Chronicity is defined as evidence of persistent infection for at least 6 months after exposure to the virus. In the chronic phase of hepatitis C, 20% of infected persons have clinical liver disease and complain of fatigue and malaise, 50% are asymptomatic but have increased liver enzyme levels, and 30% are asymptomatic and have normal liver enzyme levels
(1). The progression of chronic hepatitis C is slow; often the first clinical indication of chronic infection is liver failure, hepatic encephalopathy, jaundice, or ascites. In the United States, hepatitis C currently is the leading cause of chronic liver disease and the most common indication for liver transplantation
(8).
Cirrhosis develops in approximately 20% of chronic carriers, and subsequent liver failure, portal hypertension, and hepatocellular carcinoma may develop. Once cirrhosis develops, the risk of hepatocellular carcinoma increases, occurring at a rate of approximately 1%–4% per year
(9).
A variety of extrahepatic manifestations have been associated with hepatitis C, including a mixed cryoglobulinemia and glomerulonephritis. Other associated syndromes include porphyria cutanea tarda, Sjogren’s syndrome, and hypo- and hyperthyroidism
(10). It is not clear how hepatitis C is related to the pathogenesis of these conditions or how frequently they occur. Clinicians should be aware of the varied symptomatic complaints and possible clinical presentations of hepatitis C, so that the diagnosis is not overlooked.
Several studies have shown that heavy drinking of alcohol worsens hepatic injury and may accelerate progression of hepatitis C infection
(11). However, the threshold for injury from alcohol is not determined, so it is unclear whether light or moderate drinking accelerates liver damage. Unfortunately, excessive drinking appears to be common among individuals infected with hepatitis C. Lee et al.
(12) found that 29% of individuals with hepatitis C reported heavy alcohol use and an additional 34% consumed alcohol regularly. Alcohol not only increases the risk of cirrhosis but also enhances the risk of hepatocellular carcinoma.
NEUROPSYCHIATRIC SYMPTOMS ASSOCIATED WITH INTERFERON IN HEPATITIS C PATIENTS
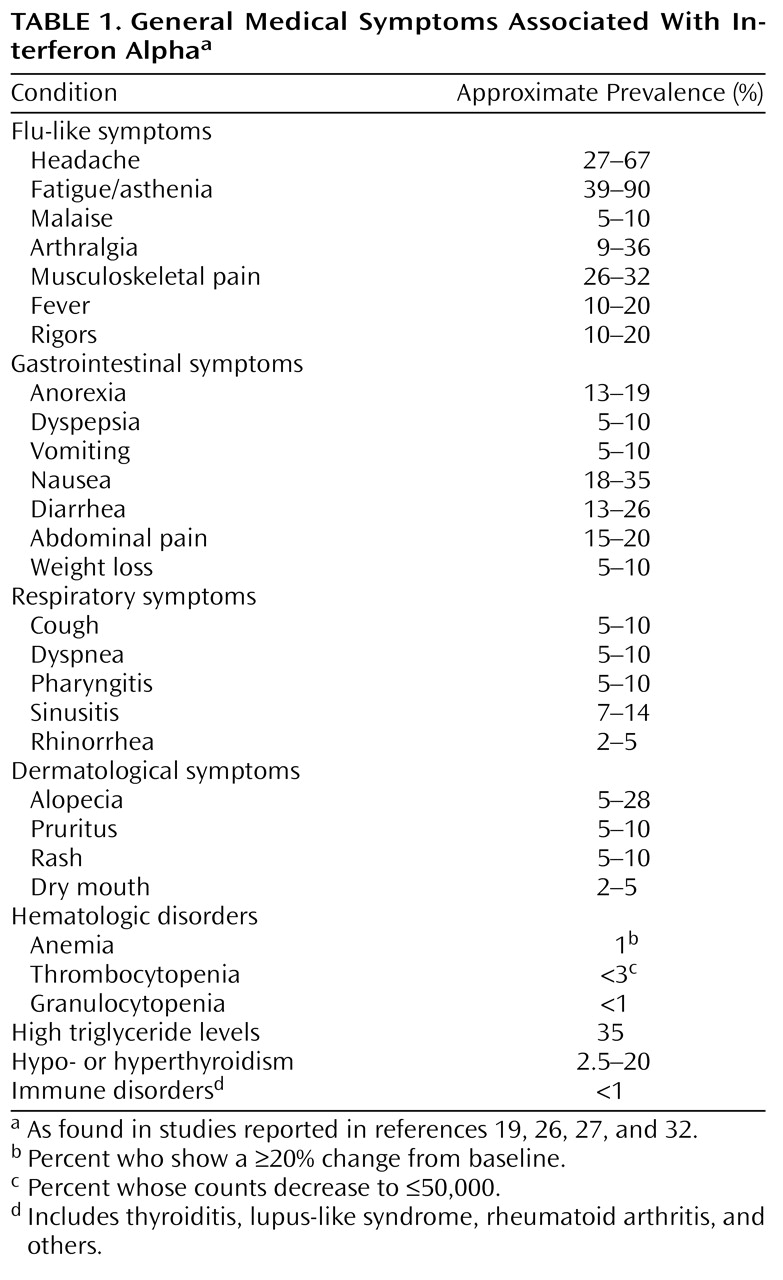
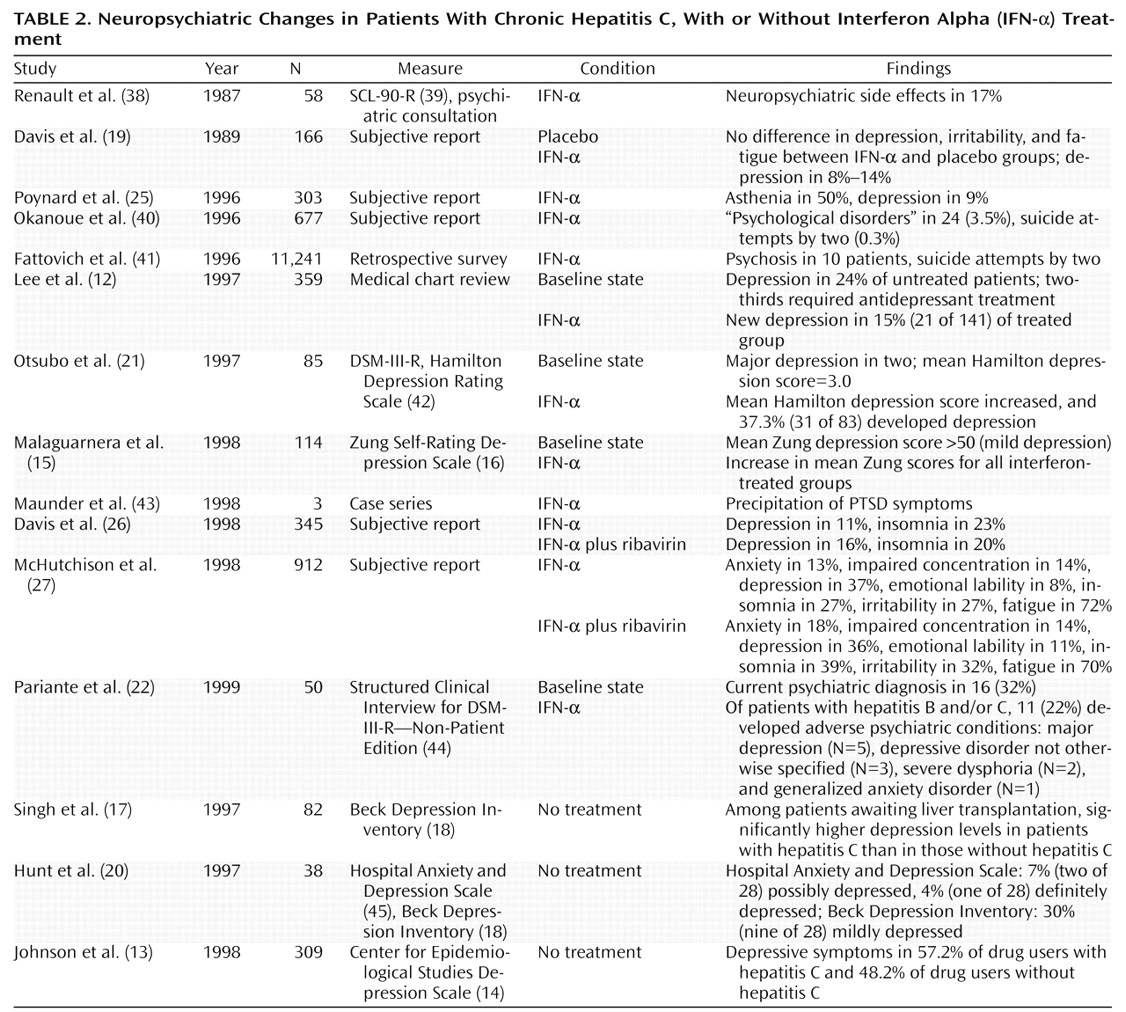
In addition to the neurological symptoms, more subtle neuropsychiatric symptoms frequently occur in hepatitis C patients treated with IFN-α (
Table 2). These side effects are troublesome and frequently account for dose reduction or treatment discontinuation. In one study of 58 patients with chronic viral hepatitis treated with IFN-α
(38), psychiatric symptoms were the most frequent reason for discontinuing therapy. These symptoms include cognitive, affective, and behavioral components that are not easily distinguished from each other or from depression. In addition, fatigue and insomnia, which affect both mental and physical functioning, are prominent side effects of interferon therapy.
The cognitive changes associated with IFN-α include impaired concentration, decreased alertness, deficits in verbal memory, and mental slowing. Reduced alertness and slowed reactions have been reported after only a single dose of interferon in healthy volunteers
(46).
Cancer patients have a high incidence of cognitive changes during interferon treatment. In a retrospective study of cancer patients who had received biologic response modifiers (IFN-α, interleukins, and others) Meyers and Abbruzzese
(47) found a 53% frequency of cognitive abnormalities on neuropsychological tests. That study and others
(48–
51) showed similar patterns of disturbances, including psychomotor slowing, visual-constructional deficiencies, motor perseveration, and difficulties with backward digit span, logical verbal tasks, calculation, and memory recall. However, other investigators
(36,
52,
53) have found no problems in the neuropsychological performance of cancer patients treated with interferon. The explanation for this discrepancy is not clear. However, duration of therapy is one factor that differs across studies. In the preceding studies, neuropsychological examination later in the course of treatment was associated with positive findings. In 25 patients with chronic myelogenous leukemia, neuropsychological tests showed poorer performance on tests of cognitive speed, verbal memory, and executive function in those treated with IFN-α than in patients with myelogenous leukemia not exposed to IFN-α
(54). In that study, duration of therapy accounted for 55% of the variance in cognitive and mood scores. Furthermore, Amato et al.
(55) showed that cognitive changes were related to the number of courses of treatment, with 100% of patients experiencing memory loss after more than six courses of treatment with interferon. Other possible reasons for disparate results are the small numbers of patients in many studies, missing data, and the variety of diseases treated.
It is not clear whether cognitive deficits persist after treatment discontinuation. In a retrospective study of 14 patients
(56), 12 of whom had discontinued treatment because of neurotoxicity, all had evidence of persistent neurotoxic deficits, and 10 showed symptoms consistent with frontal-subcortical dysfunction. Most authors have noted resolution of symptoms within 2–3 weeks after discontinuation of interferon
(57), but the study methods have been poor.
The cognitive changes in hepatitis C patients treated with IFN-α have not been well studied. McHutchison et al.
(27) found that 14% of hepatitis C patients treated with either IFN-α alone or IFN-α plus ribavirin experienced impaired concentration as measured by a self-report questionnaire. In a consensus interferon comparative trial
(32), 2%–5% of patients reported confusion or impaired concentration as adverse experiences. Although little is documented with respect to cognitive changes secondary to IFN-α treatment in patients with hepatitis C, they may parallel those seen in cancer patients.
The reported rates of depressive symptoms associated with IFN-α range from 3% to 57%, with most investigators finding proportions between 10% and 40% (
Table 2). However, many of these data were based on self-report questionnaires concerning general side effects, rather than validated measures of depression. In most studies, “depression” was considered as a single symptom or problem, and its various manifestations were not investigated individually. In a review of 1,019 patients receiving interferon for various malignancies
(58), 6.5% complained of depression. In a review of the safety of interferon, based on reports of adverse events to the Food and Drug Administration, depression, suicidal ideation, attempted suicide, and completed suicide constituted approximately 4% of all adverse events reported
(59). Studies investigating interferon treatment for chronic hepatitis C by using self-report questionnaires have yielded varying rates of depression. Davis et al.
(19) found no difference in the rate of reported depression between patients treated with IFN-α and a placebo group. However, in two recent large multicenter trials comparing IFN-α alone to IFN-α plus ribavirin for treatment of chronic hepatitis C
(26,
27), rates of depression between 11% and 37% were found. A large retrospective survey of 11,241 hepatitis C patients treated with interferon
(41) revealed psychosis in 10 patients and two suicide attempts. Okanoue et al.
(40) reported that only 3.5% of 677 patients had psychological disorders; however, two patients attempted suicide. The expected rates for these disorders are significantly higher than those reported, suggesting that these studies were at least insensitive, and perhaps had other methodological inadequacies.
Four prospective studies have evaluated mood symptoms in hepatitis C patients treated with IFN-α (
Table 2). Renault and colleagues
(38) found that three of 58 patients with chronic viral hepatitis developed an “organic affective syndrome” marked by depression, hopelessness, emotional lability, and tearfulness. In a prospective placebo-controlled study comparing the effects of four different types of interferon on the rate of depression in patients with chronic hepatitis C
(15), a sudden increase in the mean scores on the Zung Self-Rating Depression Scale for all interferon-treated groups was found. These scores increased from mild to moderate depression at 1 month for all groups and returned to baseline by 3 months of treatment. In a prospective study by Otsubo et al.
(21), reported in the Japanese literature and available in English only as an abstract, 31 (37.3%) of 83 patients were diagnosed as having a major depressive episode, according to DSM-III-R criteria, at least once during a 24-week course of interferon therapy for hepatitis C. The mean score on the Hamilton Depression Rating Scale
(42) was also higher than it had been at baseline. The same group of investigators recently presented similar results in a letter to the
Journal (60). Pariante et al.
(22) prospectively followed 50 patients with hepatitis B and/or C, using the Structured Clinical Interview for DSM-III-R—Non-Patient Edition
(44), and found that 22% developed psychiatric diagnoses during treatment; five patients developed major depression, three developed depressive disorder not otherwise specified, two developed severe dysphoria, and one developed generalized anxiety disorder.
It is not clear why the reported rates of depression vary so widely. The rates were higher in studies that used a specific tool to assess for depression and in prospective studies. Of course, the term “depression” has several meanings, ranging from transient sadness to the syndrome of major depression. Also, patients experiencing fatigue, malaise, or cognitive changes may complain of being “depressed.” At the same time, suicidal ideation has been reported, and completed suicide has occurred during the course of IFN-α therapy
(61,
62), so depressive symptoms must be taken seriously.
Depressive symptoms immediately before treatment may be more important than a history of psychiatric illness or treatment in predicting the intensity of depression that develops during interferon therapy. Renault et al.
(38) found that prior individual or family history of psychiatric illness did not predict the development of depressive symptoms. In the study by Pariante et al.
(22), previous or current psychiatric illness did not predict discontinuation of IFN-α therapy or the development of psychiatric adverse effects. However, higher baseline scores on the Montgomery-ijberg Depression Rating Scale
(63) predicted the intensity of depressive symptoms during treatment of 10 cancer patients
(64). When Otsubo and colleagues
(21) compared the patients who experienced depression during interferon treatment to those who did not, the depressed group showed significantly higher scores on the Eysenck Personality Questionnaire
(65), more severe depressed mood, and more severe sleep disturbances before the start of interferon therapy. One shortcoming of these studies is the lack of inclusion of patients with prior or recent diagnoses of substance abuse or dependence. Further studies are needed to find predictors of psychiatric morbidity during IFN-α treatment.
Irritable or labile mood may be another manifestation of depression. Irritability has been reported in up to 35% of patients receiving IFN-α
(27). Emotional lability is reported in approximately 10% of patients
(26). These side effects may be sufficiently distressing as to require dose reduction or termination of treatment, suggesting that this may be a focus of intervention.
Like irritability, fatigue may result in reductions of the IFN-α dose
(66,
67). Fatigue is a nearly ubiquitous side effect of IFN-α, reported in up to 90% of patients receiving such treatment
(66). Patients report fatigue after initial doses of IFN-α, and it may become more severe during treatment. Generally, tolerance to IFN-α–induced fatigue does not develop. Fatigue may be described in terms of weakness, tiredness, lack of desire to participate in normal activities, mental laziness, and poor motivation. Fatigue may be worse in patients receiving higher doses, those receiving doses daily (as opposed to every other day), and older patients. In a study using the Piper Fatigue Scale
(68), for 30 patients with malignant melanoma the most extreme fatigue scores were in the affective domain, which is the emotional meaning attributed to fatigue. In this study, fatigue peaked at 6 weeks and remained high throughout treatment
(67). As for other psychiatric disorders precipitated by treatment with IFN-α, occasional cases of mania and psychosis have been reported
(69–
71), as well as a single case report of symptoms related to posttraumatic stress disorder
(43). There have been reports of other anxiety symptoms, including phobias and obsessional thoughts and rituals. Finally, insomnia has been reported in up to 40% of patients and can be another dose-limiting symptom
(72).
MECHANISMS OF INTERFERON-ASSOCIATED NEUROPSYCHIATRIC CHANGES
Studies of mechanisms are preliminary, so theories about mechanisms remain speculative. IFN-α may directly affect the central nervous system (CNS). For example, it has been shown to enhance the excitability of cultured neurons from rats and cats
(73), and it may gain entry to the CNS in areas where the blood-brain barrier is lacking, such as the median eminence, area postrema, and infundibular recess.
IFN-α affects neuroendocrine, cytokine, and neurotransmitter pathways. IFN-α and ACTH have been reported to be antigenically and structurally related
(74), and IFN-α induces increased plasma cortisol levels acutely
(75,
76). This suggests one possible mechanism for IFN-α induction of depression, since chronically increased hypothalamic-pituitary-adrenal (HPA) activity is associated with depression
(77,
78). IFN-α also alters thyroid function. Hypothyroidism or hyperthyroidism develops in 2.5%–20% of patients
(79,
80), and this may cause or exacerbate neuropsychiatric symptoms.
IFN-α induction of secondary cytokines, such as interleukin-1 (IL-1), IL-2, IL-6, and tumor necrosis factor alpha (TNF-α), may also contribute to neuropsychiatric symptoms. Fever, sleep disturbance, cachexia, anorexia, neurodegeneration, and neuroendocrine dysfunction are associated with IL-1 and TNF-α secretion
(81). In experimental animals, IL-1 may damage the blood-brain barrier and thus contribute to IFN-α–related neurotoxicity. IL-6 levels have been shown to be elevated in patients with depression
(82).
Investigators are beginning to elucidate possible mechanisms linking secondary cytokines and neuroendocrine systems. Secondary cytokines may activate the HPA axis, which may in turn cause depressive symptoms. Cytokines can stimulate neurons containing corticotropin-releasing factor (CRF) in the paraventricular nucleus of the hypothalamus
(83). Although the exact mechanism for this stimulation is unclear, both direct CNS and peripheral action by cytokines have been proposed. Cytokines that enter the brain in areas lacking a blood-brain barrier may directly activate neuroendocrine systems through prostaglandin and nitrous oxide pathways
(83,
84). IL-6 directly affects CRF-containing nerve terminals in the median eminence. Secondary cytokines may signal the brain, via the vagus nerve, by stimulating peripheral visceral afferent neurons and ultimately modulate CRF secretion via several interconnected neuronal circuits
(83,
84).
Neurotransmitter levels are also acutely increased by IFN-α. Norepinephrine and epinephrine increase with administration of IFN-α in healthy volunteers
(75). Also, IFN-α may act as a central dopamine agonist through an opioid-associated mechanism
(85). Persistent elevation of amines in patients treated with IFN-α is a possible mechanism of depression, as it is thought that long-term administration of amine agonists may result in down-regulation and subsensitivity of receptors
(86).
Levels of the neurotransmitter serotonin may be reduced because of reductions in serum tryptophan levels. Tryptophan is an amino acid precursor of serotonin. Evidence implicating the importance of tryptophan in depression comes from both treatment
(87,
88) and tryptophan-depletion
(89–
91) studies. Depletion of brain tryptophan has temporarily reversed the antidepressant effects of selective serotonin reuptake inhibitors (SSRIs) and has produced dysphoric symptoms in several patient populations. Reduction in serum tryptophan levels, by as much as 50%, has been observed in patients treated with IFN-α. A possible mechanism for IFN-α–mediated tryptophan depletion involves induction of indoleamine 2,3-dioxydenase. Induction of this enzyme effectively shunts tryptophan along the kynurenine pathway and away from the serotonin pathway. Alterations in the serotonin transporter may also play a role in IFN-α–associated depression. The serotonin transporter plays a crucial role in the termination of serotonergic neurotransmission, is known to be a main target of antidepressants
(92,
93), and is implicated in the pathogenesis of depression
(94). Recently Morikawa et al.
(95) showed that IFN-α alters levels of serotonin transporter messenger RNA, suggesting a possible mechanism of IFN-α–induced depression.
DISCUSSION
Hepatitis C infection is common, and although the incidence of new infection is declining, a large population has been exposed to this virus and will receive clinical attention in the next 10–20 years. Furthermore, identification and treatment of chronic hepatitis C infection are likely to become more aggressive with the improved prognosis offered by combination therapy. For example, the Department of Veterans Affairs has made it a priority to diagnose and treat veterans with hepatitis C. One preliminary estimate projects that this initiative will cost $670 million
(101). Undoubtedly, psychiatrists will increasingly be involved in the care of such patients.
Patients are likely both to seek treatment from psychiatrists directly and to be referred by primary care providers and hepatologists. Psychiatrists are often involved in the care of intravenous drug users and alcoholics, both of whom have high rates of hepatitis C infection. These patients should be routinely tested for hepatitis C virus, even in the absence of elevations in liver function measures. In addition, alcohol-dependent patients in particular should be warned about the additive or possibly synergistic effects of heavy drinking and hepatitis C on the development of serious liver disease. Hepatitis C may also be manifested in less obvious ways. For example, in the absence of abnormal liver enzyme levels, symptoms of fatigue and malaise may not be investigated or may be dismissed as a component of psychiatric illness or as medication side effects. Even in patients with known risk factors for hepatitis C, these symptoms may attract little or no attention. Depression may be the presenting syndrome as well. The threshold for hepatitis C testing should be very low in these patients. As the treatment of chronic hepatitis C improves, early detection of hepatitis C might lead to secondary prevention and improved outcomes.
Increasing numbers of patients undergoing treatment with IFN-α are almost certain to be referred for evaluation and treatment of depression and possibly for other neuropsychiatric symptoms. Recognition of depression is important, as treatment has the potential to improve adherence to IFN-α treatment. Mohr and colleagues
(102) found that treating the interferon-associated depressive symptoms in multiple sclerosis patients reduced the number of treatment dropouts. Identifying patients who are likely to respond to treatment of depression is critical, as recently it has become apparent that patients with depressive symptoms who are medically ill and who do not meet criteria for DSM-IV major depression are at risk for the same problems as those diagnosed with dysthymia and major depression. These patients suffer from diminished quality of life, less participation in life and medical care, increased disability, increased utilization of health care services, and increased morbidity and mortality
(103). Unfortunately, there are almost no data available concerning efficacy of antidepressant treatment for chronic hepatitis C and IFN-α–associated neuropsychiatric conditions. In clinical practice, SSRIs appear to be used commonly and to be helpful. However, controlled trials are needed to determine whether this is true, and for whom.
Even more basic descriptive research is needed in order to adequately design an intervention trial. The existing studies of depressive symptoms associated with IFN-α treatment are seriously flawed and difficult to compare. At this point, it is not possible to determine how many hepatitis C patients undergoing IFN-α treatment develop depressive symptoms that might warrant treatment, how long they last, or whether they can be distinguished clearly from nonspecific malaise and fatigue. A thorough prospective study using multiple testing points and having adequate sample size is needed before treatment studies are begun. Additional research into the mechanisms underlying the development of neuropsychiatric symptoms with hepatitis C and with IFN-α treatment are also needed. The possibility that stimulation or hypersensitivity of the HPA axis might be implicated is particularly intriguing and should be pursued. Serotonin dysfunction is also a possible mechanism of depression in hepatitis C patients and provides some rationale for treating this population with SSRIs. Tryptophan supplementation as a way to increase serotonin also deserves study.
Research may not only shed light on general mechanisms of depression but also could further the understanding of the complex interaction between endocrine and immunologic processes and neuropsychiatric symptoms. Most important, improved understanding of the natural history and treatment of these symptoms may help patients with chronic hepatitis C sustain treatment, which ultimately may prolong life and improve their quality of life. Research should include patients with psychiatric problems so that they are not excluded from effective treatments for hepatitis C.