In the present study we attempted to extend prior findings by testing whether differential rostral anterior cingulate activity predicts
degree of response to treatment, an important question in light of the substantial variability in response among depressed patients
(17). Higher activity in the rostral anterior cingulate was hypothesized to be associated with better response. Unlike the subjects in prior studies
(11–
14), most of the depressed participants investigated here were responders. Another distinguishing feature of this study was that it showcased a novel method for extracting localized sources from surface brain electrical activity recordings. The technique begins with EEG data, which are then analyzed with a new tomographic analysis procedure
(18–
20) that computes the three-dimensional intracerebral distributions of current density for specified EEG frequency bands without assumptions about any predefined numbers of generating sources. On the basis of EEG and magnetoencephalographic studies
(21–
23) implicating the anterior cingulate cortex as a likely neuronal generator for theta activity in the human brain, theta was hypothesized to be the only one of the seven conventional frequency bands analyzed to show this effect. In particular, despite prior literature implicating alpha activity in depression
(1,
3), an association between alpha activity and response to treatment was not expected because of no known relation between alpha activity and the anterior cingulate cortex
(24,
25).
Results
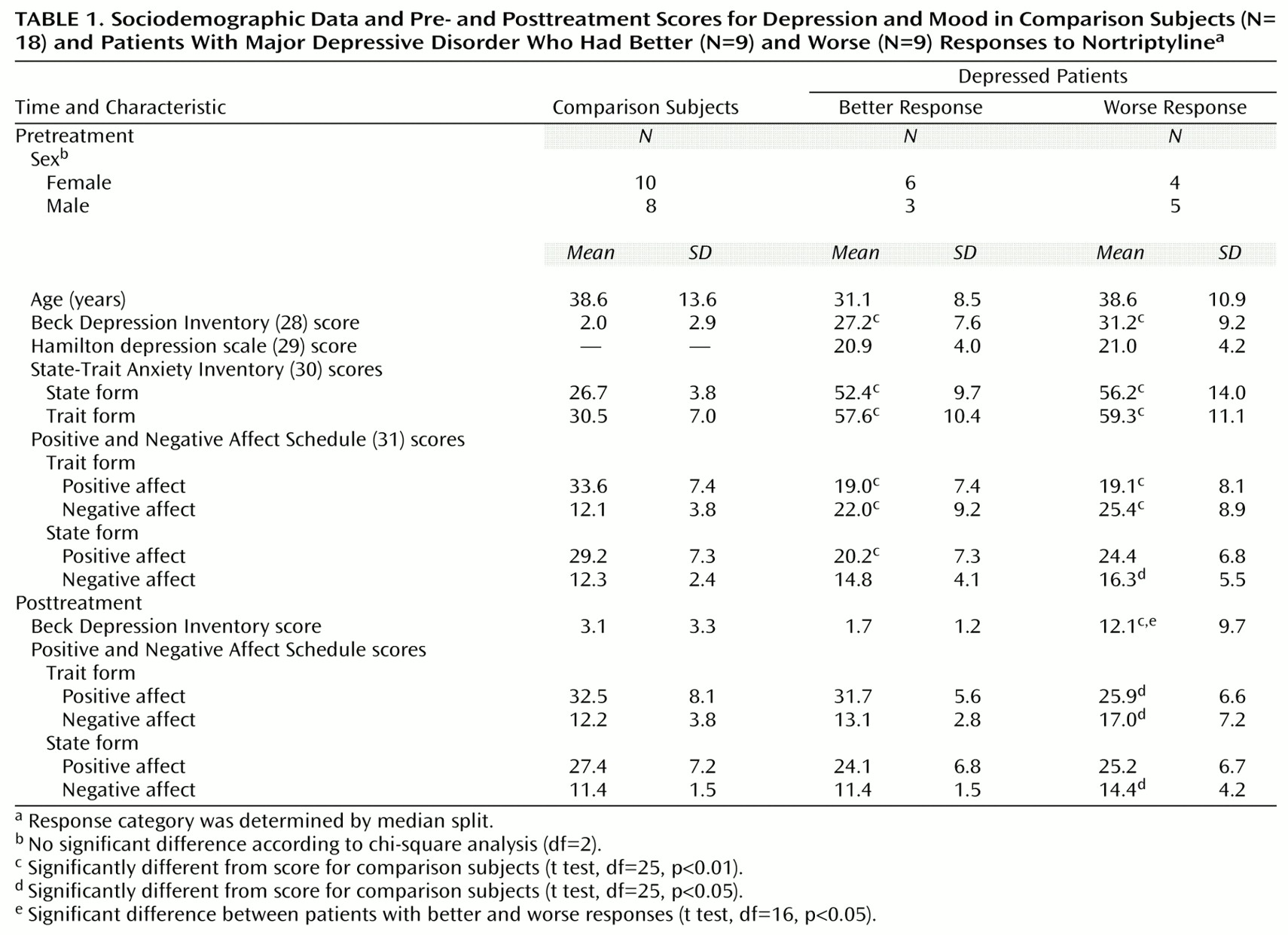
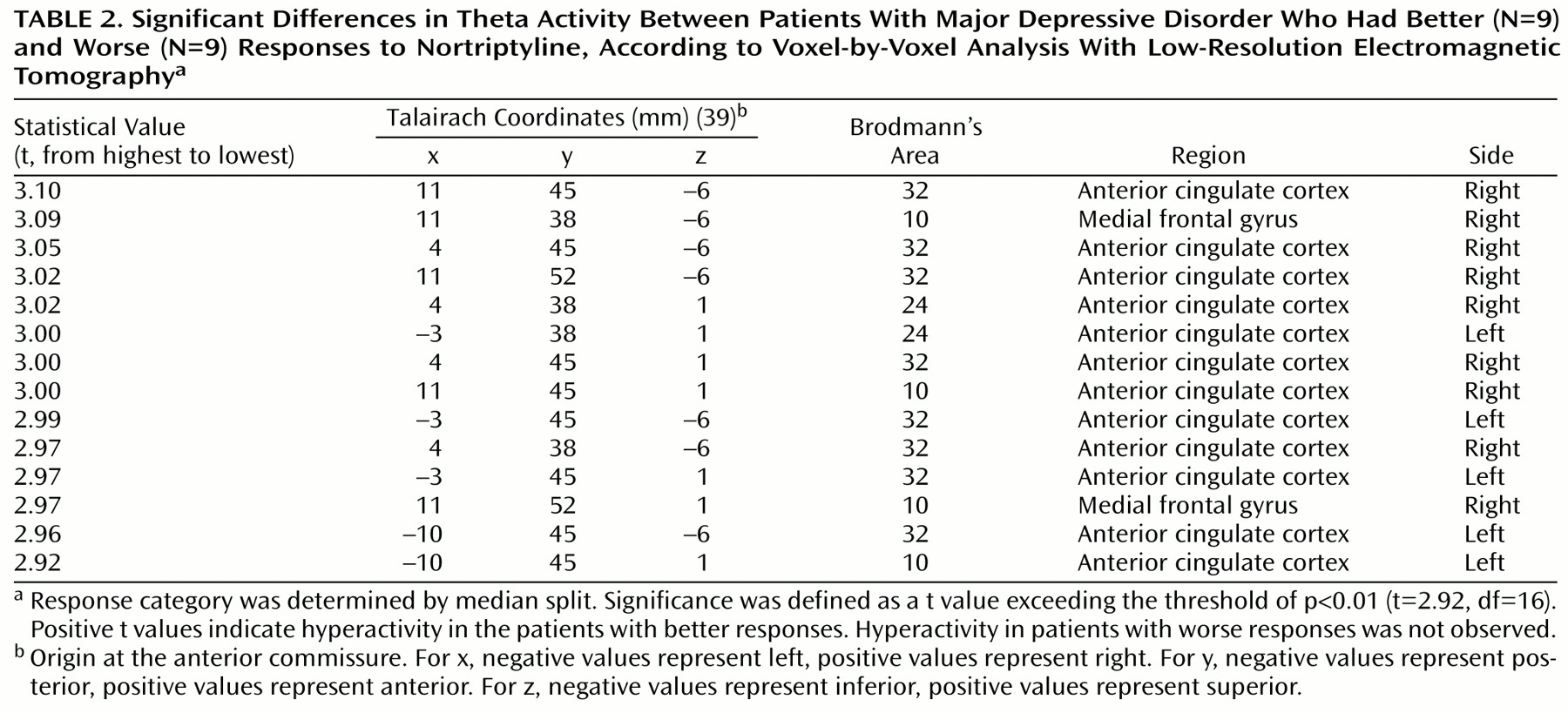
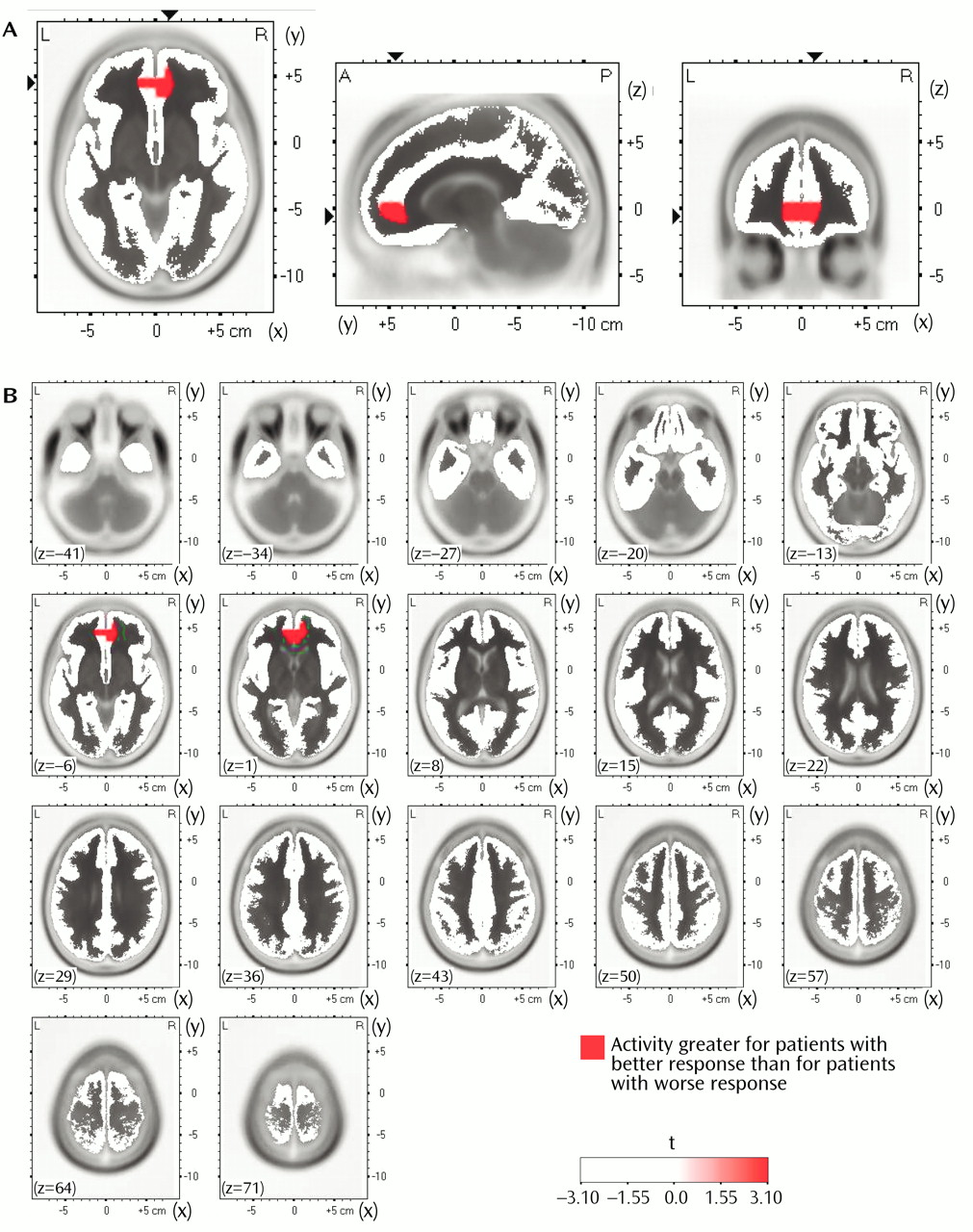
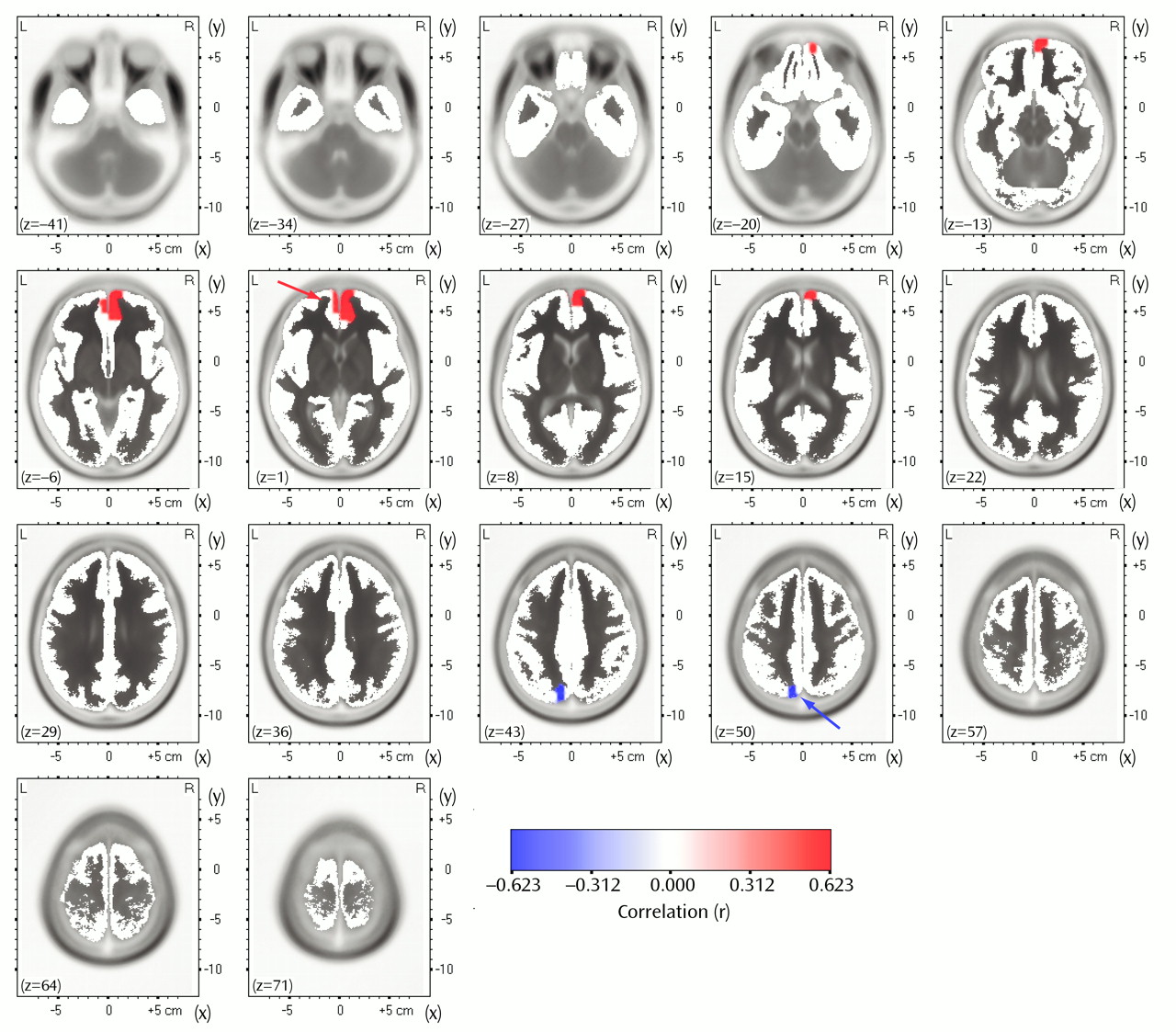
In the comparisons of depressed subjects with better and worse responses to treatment, the only result in any frequency band to survive the statistical threshold was theta activity (6.5–8.0 Hz). (To test whether significant results emerged only from the theta band because of the lower threshold, p<0.01 versus p<0.005, analyses were repeated for the other bands at a two-tailed p<0.01, but again no significant results were observed.) The significant result for theta activity was found in the rostral anterior cingulate cortex (area 24), which extended rostrally to mesial regions of area 32 and to some voxels in the medial frontal gyrus (area 10;
Figure 1). As shown in
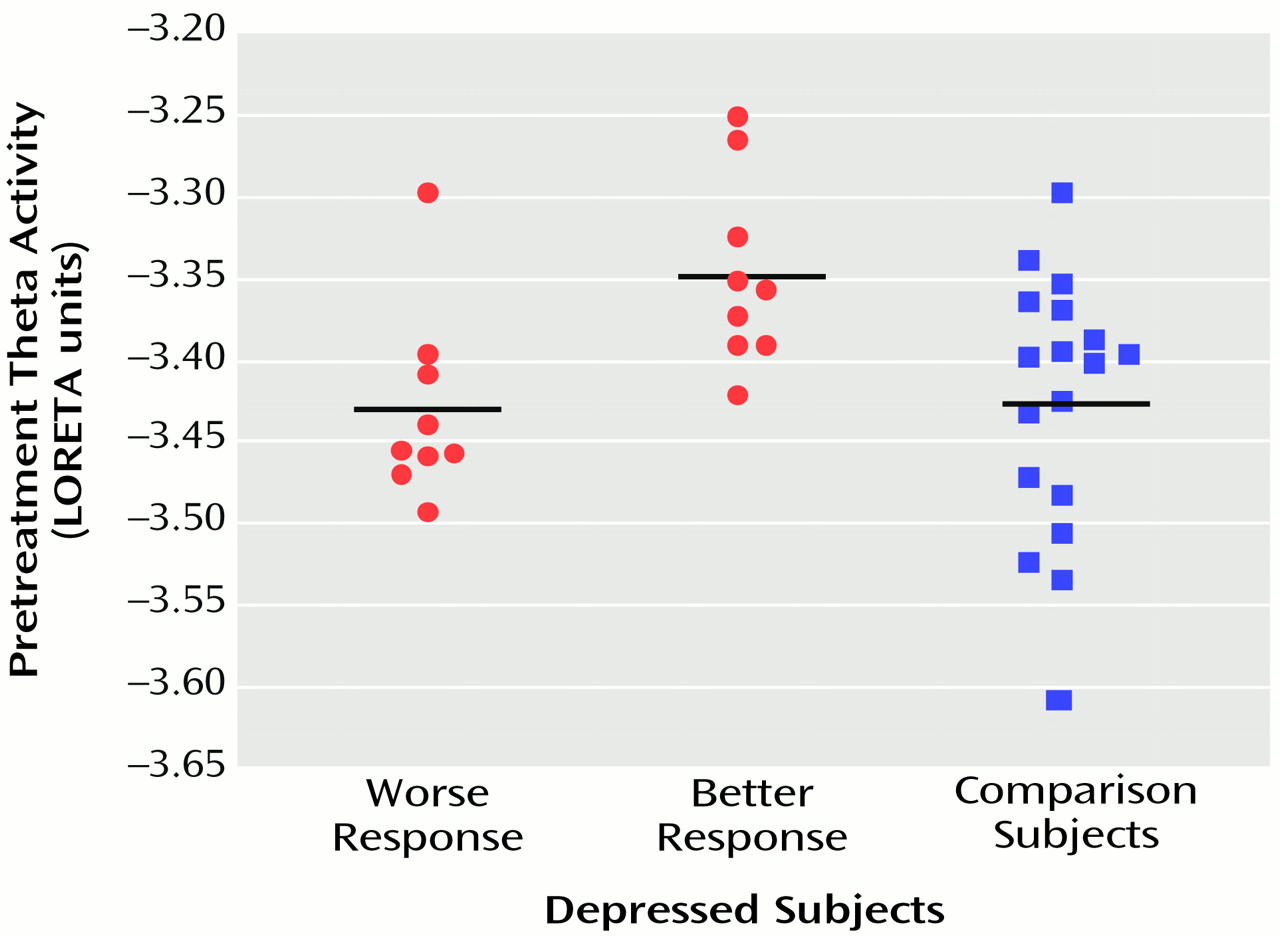
Figure 2, LORETA theta activity was higher in the patients with better responses (mean=–3.35, SD=0.06) than in the patients with worse responses (mean=–3.43, SD=0.06) (t=3.10, df=16, p<0.008). The difference between the patients with better responses and the comparison subjects (mean=–3.43, SD=0.08) did not pass the statistical threshold (t=2.65, df=25, p<0.02), whereas the patients with worse responses and the comparison subjects showed virtually identical pretreatment levels of anterior cingulate activity. Significant differences between the patients with better and worse responses were grouped in an uninterrupted cluster (volume=4.8 cm
3) involving 14 voxels (nine in the right and five in the left hemisphere) (
Table 2).
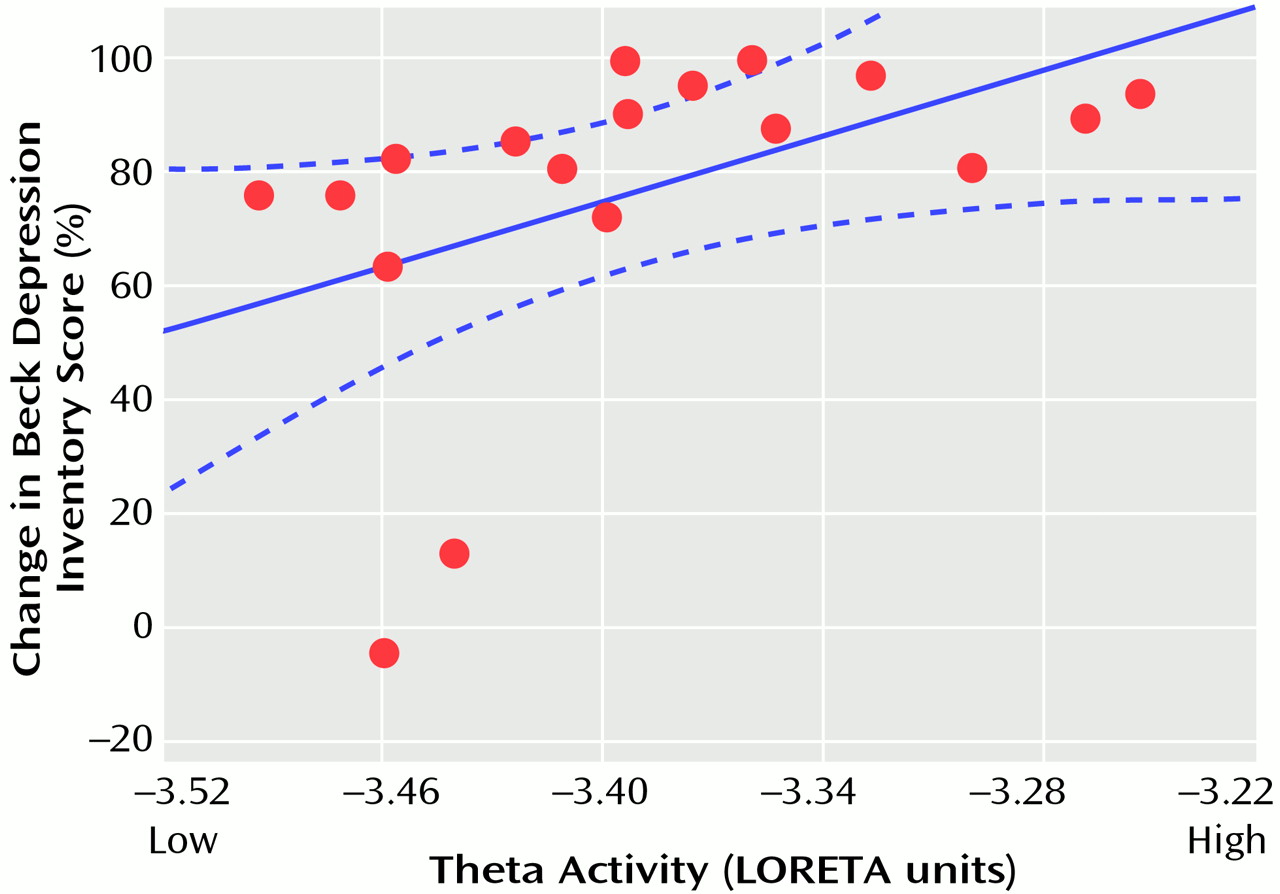
Follow-up analyses showed that these results were not driven by the two depressed patients who were classified as clinical nonresponders. The Pearson correlation between the percentage change in Beck Depression Inventory score and the theta activity in the entire cluster was 0.48 (df=16, p<0.05) with the two nonresponders and 0.56 (df=14, p<0.03) without them. The higher the LORETA theta activity in these rostral regions of the anterior cingulate cortex, the better the response to treatment (
Figure 3).
To test whether the association with the degree of response was specific to the rostral anterior cingulate cortex, we computed Pearson correlations between the percentage change in Beck depression score and the LORETA theta activity in the entire brain volume without the two nonresponders. In this analysis, only two regions showed significant correlations (p<0.05, uncorrected for multiple comparisons). As shown in
Figure 4, theta activity within a medial frontal region extending caudally to areas 24 and 32 of the anterior cingulate cortex (33 voxels; 27 in the right and six in the left hemisphere) was positively correlated (r=0.54, df=14, p<0.05) with percentage change in the Beck depression score, whereas theta activity within a much smaller region involving the left precuneus (five voxels) was negatively correlated (r=–0.62, df=14, p<0.03) with percentage change in Beck depression score.
Because the depressed patients with better responses were slightly (but not significantly) younger than those with worse responses and because anterior cingulate activity shows remarkable functional decrease with age
(44), a hierarchical regression analysis predicting theta activity within the cluster emerging from the correlational analysis was conducted by entering age as the first predictor and then entering percentage change in Beck depression score. Age was not related to theta activity in the anterior cingulate cortex (beta=–0.09, t=–0.32, df=14, p>0.75). Percentage change in Beck depression score remained a significant predictor of theta activity in the anterior cingulate cortex even after removal of the variance associated with age (beta=0.64; R
2 change=0.32, F=6.20, df=1, 13, p<0.03). Similar information was obtained from calculating the partial correlation between percentage change in Beck depression score and theta with age partialed out (r=0.57, df=13, p<0.03).
Discussion
In a group of individuals treated for major depression, baseline theta activity within a rostral region of the anterior cingulate cortex involving areas 24 and 32 predicted degree of response 4–6 months later. Depressed subjects showing a better response had higher theta activity than those responding less well, and they also tended to have higher activity than comparison subjects; this finding replicates that of Mayberg et al.
(11). In addition, theta activity in the left precuneus was inversely related to response after treatment. However, this unexpected effect involved a very small region (five voxels) and emerged only in the correlational analysis with less stringent statistical criteria.
Prior research suggests that higher theta activity within the anterior cingulate cortex is best interpreted as an electrophysiological manifestation of higher activation within this region. This interpretation relies on observations of increased theta activity during stimulus expectancy and focused attention
(45), higher working memory load
(22), successful encoding of new information
(46), and concentrated task performance
(47).
The present findings add to the emerging story of anterior cingulate involvement in depression. Dorsal anterior cingulate activity is low in depression
(4–
8), and remission has been associated with increased activity in the same region
(48–
50). Similarly, the high level of activity in the rostral anterior cingulate cortex (area 24a) and mesial regions of area 32 associated with response to treatment
(11–
14,
49) has been shown to normalize (i.e., decrease) in patients who respond to sleep deprivation
(14,
51). The fact that rostral anterior cingulate activation predicted the magnitude of response in the current study and did not simply differentiate between responders and nonresponders underscores the robustness of this effect and extends previous findings
(11–
14,
49) by demonstrating that the magnitude of anterior cingulate activation accounts for gradations of response (even for the analyses that excluded clinical nonresponders). (We previously reported a preliminary analysis of the PET data involving nine depressed subjects
[52]. In that analysis, patients with better and worse responses differed in a clearly more dorsal region of the anterior cingulate: x=–4, y=–14, z=46; areas 23, 24, 31. The fact that only six of these subjects had artifact-free EEG data and therefore could be included in our group of 18 depressed subjects may account for the discrepancy between the EEG and PET results.) The region involved is remarkably close to the ones reported previously in studies of response to treatment that used PET
(11,
14) and single photon emission computed tomography (SPECT)
(12). Taken together, these results suggest that hyperactivation of the rostral anterior cingulate cortex may represent an adaptive, compensatory reaction to the state of being depressed that increases the likelihood of remission and then normalizes after recovery.
In a model of depression that integrates some of these data, Mayberg and colleagues
(10,
50) considered distinctions between different areas of the anterior cingulate cortex to account for the formation and maintenance of depressive symptoms. They described a dorsal compartment that modulates attentional and cognitive symptoms (e.g., apathy, attentional and executive deficits) and that includes the dorsolateral prefrontal cortex, the dorsolateral anterior cingulate (area 24b), the inferior parietal lobe, and the striatum. A ventral compartment, on the other hand, is postulated to be involved in vegetative and somatic symptoms (e.g., sleep disturbance, loss of appetite and libido) and includes the hypothalamic-pituitary-adrenal axis, insula, subgenual cingulate (area 25), and brainstem. Their model posits that the rostral anterior cingulate (area 24a) plays a key role in response to treatment because of its putative regulatory role in integrating information of the dorsal and ventral compartments. While not furnishing direct empirical evidence for this formulation, the present results and those gathered from previous studies
(11–
14,
49) are consistent with it.
In order to advance possible mechanisms relating rostral anterior cingulate activity and response to treatment, the functions of the various anterior cingulate subdivisions already noted need to be considered. Devinsky et al.
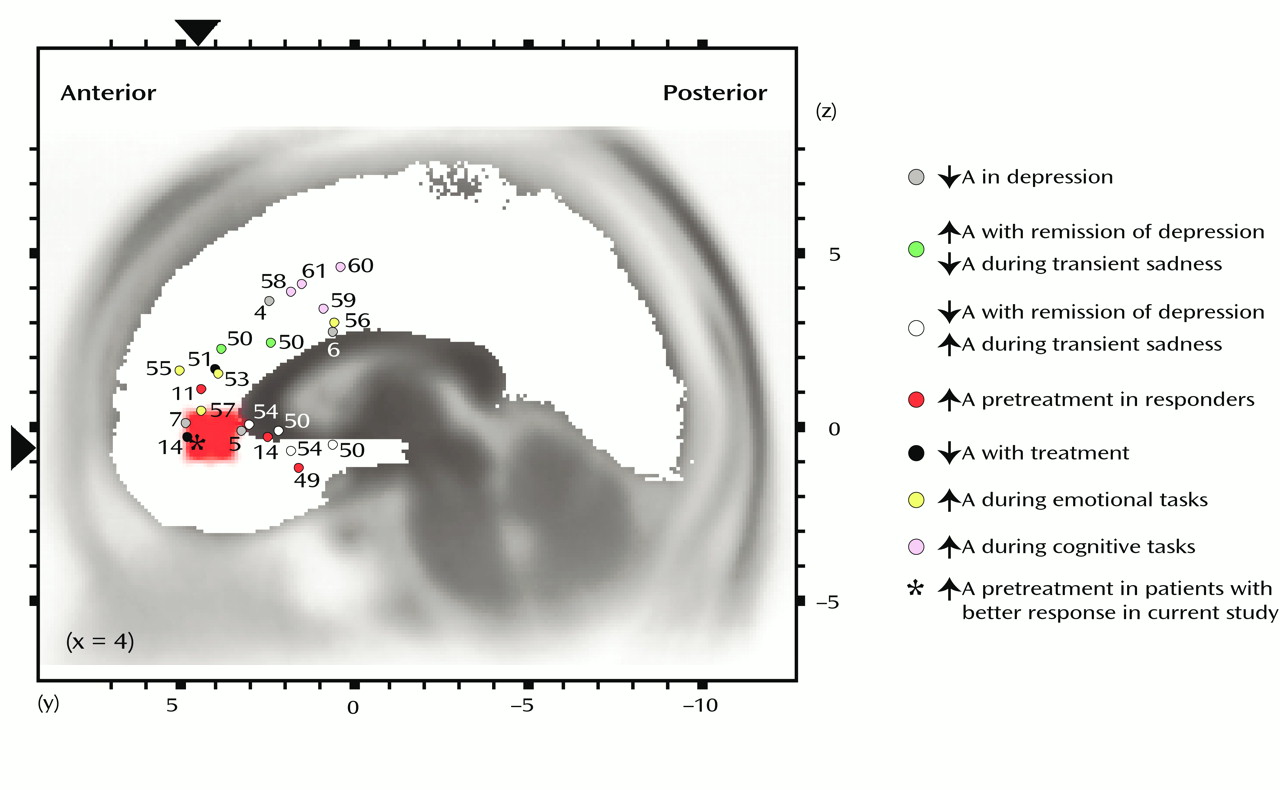
(15) argued for distinguishing between an “affect subdivision” encompassing rostral and ventral areas of the anterior cingulate cortex (areas 25, 32, 33, and rostral area 24) and a dorsal “cognitive subdivision” of the anterior cingulate (caudal area 24′ and 32′, cingulate motor area). The affect subdivision has extensive connections with limbic and paralimbic structures (amygdala, nucleus accumbens, orbitofrontal cortex, periaqueductal gray, and autonomic brainstem motor nuclei) and is involved in regulating visceral and autonomic responses to stressful behavioral and emotional events, emotional expression, and social behavior. On the other hand, the cognitive subdivision of the anterior cingulate cortex has important connections with the spinal cord and the parietal cortex, and it is involved in response selection and processing of cognitively demanding information. Functional neuroimaging studies have provided compelling evidence distinguishing the two anterior cingulate subdivisions (
Figure 5) (for reviews, see references
53 and
59). Whereas several theories include the proposition that the anterior cingulate in general acts as a bridge between attention and emotion
(9,
15,
16), we agree with Mayberg that the rostral anterior cingulate in particular may serve this purpose (see also reference
62). Functionally, the rostral anterior cingulate may integrate salient affective and cognitive information, such as that derived from environmental stimuli or task demands, and subsequently modulate attentional processes within the cognitive subdivision accordingly. With regard to good response to treatment, hyperactivation of the rostral anterior cingulate in depression might reflect an amplification of the disposition to perform these functions that in turn regulate the dysfunctional cognitive and affective patterns associated with depression. Of anatomical relevance, dorsal anterior and posterior cingulate pathways devoted to attentional processes and amygdalar pathways devoted to affective processing converge within area 24
(62), further pointing to the utility of distinguishing between the rostral and ventral areas that Devinsky et al.
(15) aggregated into the affect subdivision.
Other research further implicates the rostral anterior cingulate in monitoring and evaluating the significance of environmental stimuli. First, area 24 receives dense dopaminergic innervations from the ventral tegmental area
(15), and dopamine has been shown to be important in the encoding of information with respect to the prediction, evaluation, and occurrence of reward
(63). Consistent with this view is a recent report by Volkow et al.
(44) of an in vivo demonstration of a strong relationship between dopamine D
2 receptors and anterior cingulate metabolism in humans. Second, lesions of the anterior cingulate cortex (although not consistently restricted to the rostral area) have been associated with inadequate estimation of punishment versus reward cues, abnormal autonomic reactions to emotionally loaded information, low behavioral reactivity, apathy, loss of motivation, blunted emotional responses, and severe deficits in social behavior
(15,
64), all of which closely mirror depressive symptoms. These two lines of evidence suggest that activation of the rostral anterior cingulate cortex may favor response to treatment by fostering an individual’s capacity to monitor present or future behavior with respect to reward or punishment. This interpretation is in agreement with Drevets’s model of depression
(2), which postulates a relationship between activity in the subgenual anterior cingulate cortex and evaluative aspects of behavior in terms of reward and punishment. Of further relevance, Drevets also contended that core symptoms of depression, such as apathy and anhedonia, arise from a failure of the subgenual anterior cingulate to maintain tonic dopaminergic-dependent, reward-related activity.
Consistent with EEG and magnetoencephalographic evidence that the anterior cingulate cortex may be a possible neuronal generator for theta activity in the human brain
(21–
23) is the fact that the differences involving response in our study were observed exclusively in the theta band. Of particular note, alpha activity was not associated with response in the anterior cingulate cortex or any other region. Therefore, the relevance of alpha to depression that has been shown in numerous studies
(1,
3) does not appear to extend to response to treatment.
A limitation of the present study is the relatively low spatial resolution of the method used, which potentially could cast doubt on the specificity of the findings for the rostral anterior cingulate cortex. However, confidence in the localization capability of LORETA for the present study rests on two arguments. First, the results are highly consistent with prior PET and SPECT findings. Second, although EEG source localization techniques have been shown to be less accurate in detecting activity arising from deep brain structures, the depth of the solution for LORETA is slightly underestimated rather than overestimated
(18). A related issue is the evidence that the rostral anterior cingulate effect is lateralized to the right hemisphere, which again replicates the finding of Mayberg et al.
(11). However, caution in interpreting this finding is warranted because of the medial location of the anterior cingulate cortex and the relatively low resolution of LORETA.
In the present study, depressed individuals with higher baseline EEG theta activity within a region of rostral anterior cingulate cortex showed better response to treatment 4–6 months later. The region associated with degree of response to treatment was remarkably close to regions previously indicated in hemodynamic imaging studies of depression
(11–
14). This similarity is noteworthy because the present study used multichannel EEG, an electrophysiological imaging technology that is fundamentally different from PET and SPECT, the primary methods reported in this literature to date. In addition to lending further support for the prior observations that anterior cingulate activation predicts response to treatment in major depression, the present study showed that even among clinical responders, differential anterior cingulate activity was associated with how well they responded to treatment.