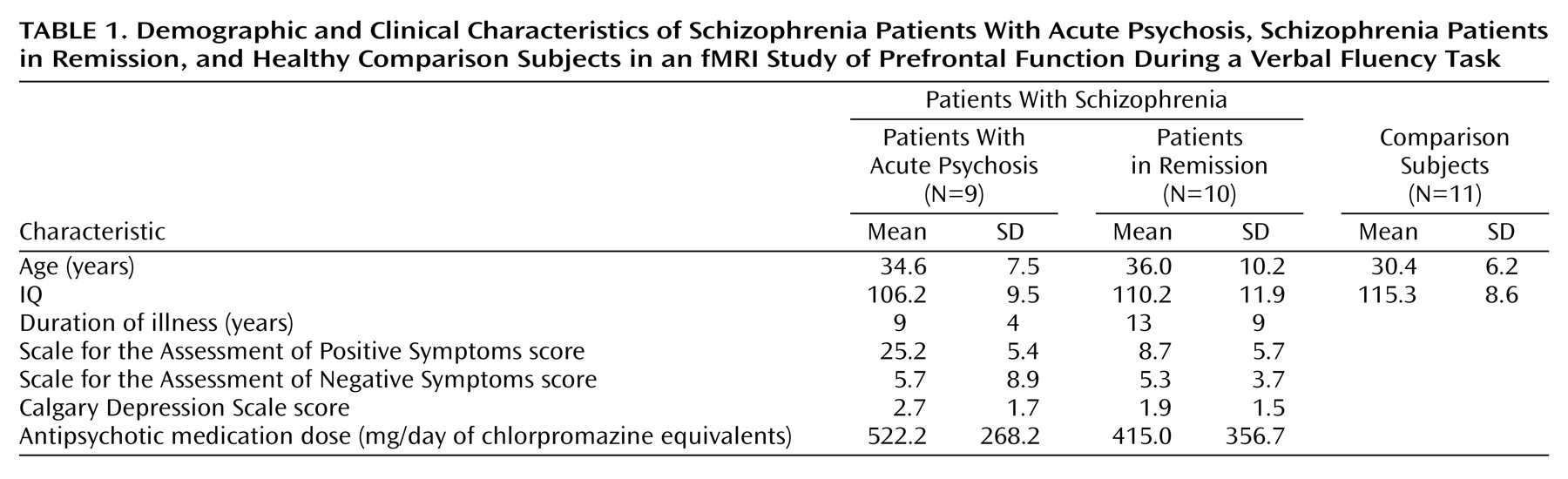
Ingvar and Franzen
(1) coined the term “hypofrontality” to describe a lower ratio of anterior to posterior cerebral activity in patients with schizophrenia, relative to healthy comparison subjects. Hypofrontality has since become the functional neuroimaging finding most associated with schizophrenia
(2), notwithstanding dissenting views and contentious evidence (reviewed in references
3 and
4). Early studies examined subjects in the “resting state”
(1,
5–10), but resting state hypofrontality has not been consistently replicated
(11–
17), nor is it specific to schizophrenia (reviewed in reference
4).
Attenuated prefrontal activation may also be evident when patients with schizophrenia perform cognitive tasks that engage the prefrontal cortex
(8,
18). However, as patients with schizophrenia often perform such tasks more poorly than comparison subjects, this difference could simply reflect impaired performance
(18–
20). Researchers have thus sought to match the performance of schizophrenia patients and comparison subjects, usually by reducing the demands of the task so that patients can perform it as well as can the comparison subjects. The results of these studies have also been inconsistent, with reports of “
hypofrontality”
(21–
23), no difference in frontal activation
(24,
25), and “
hyperfrontality”
(26,
27) in the patients with schizophrenia. The potentially confounding effects of impaired performance can also be examined by experimentally manipulating the demands of the task. Recent studies suggest that prefrontal activity in schizophrenia depends not only on the nature of the task, but also on the level of task difficulty
(23,
27–32).
The aim of the present study was to examine the effects of both task demand and current mental state on prefrontal function in schizophrenia. We utilized a letter verbal fluency task, which is associated with activation in the prefrontal cortex in healthy volunteers
(37–
39). Verbal fluency in schizophrenia has been reported to be associated with impaired activation in the middle and inferior frontal gyri and in the anterior cingulate gyrus
(21,
25,
40,
41). However, some paced studies in which performance in patients was comparable to that in healthy subjects have failed to find differences in prefrontal activation
(33,
36,
42–44). The effects of symptoms and task demand on activation during verbal fluency in schizophrenia have not been explicitly examined previously, although Sommer et al.
(36) found that the current severity of hallucinations was correlated with greater engagement of the right prefrontal cortex.
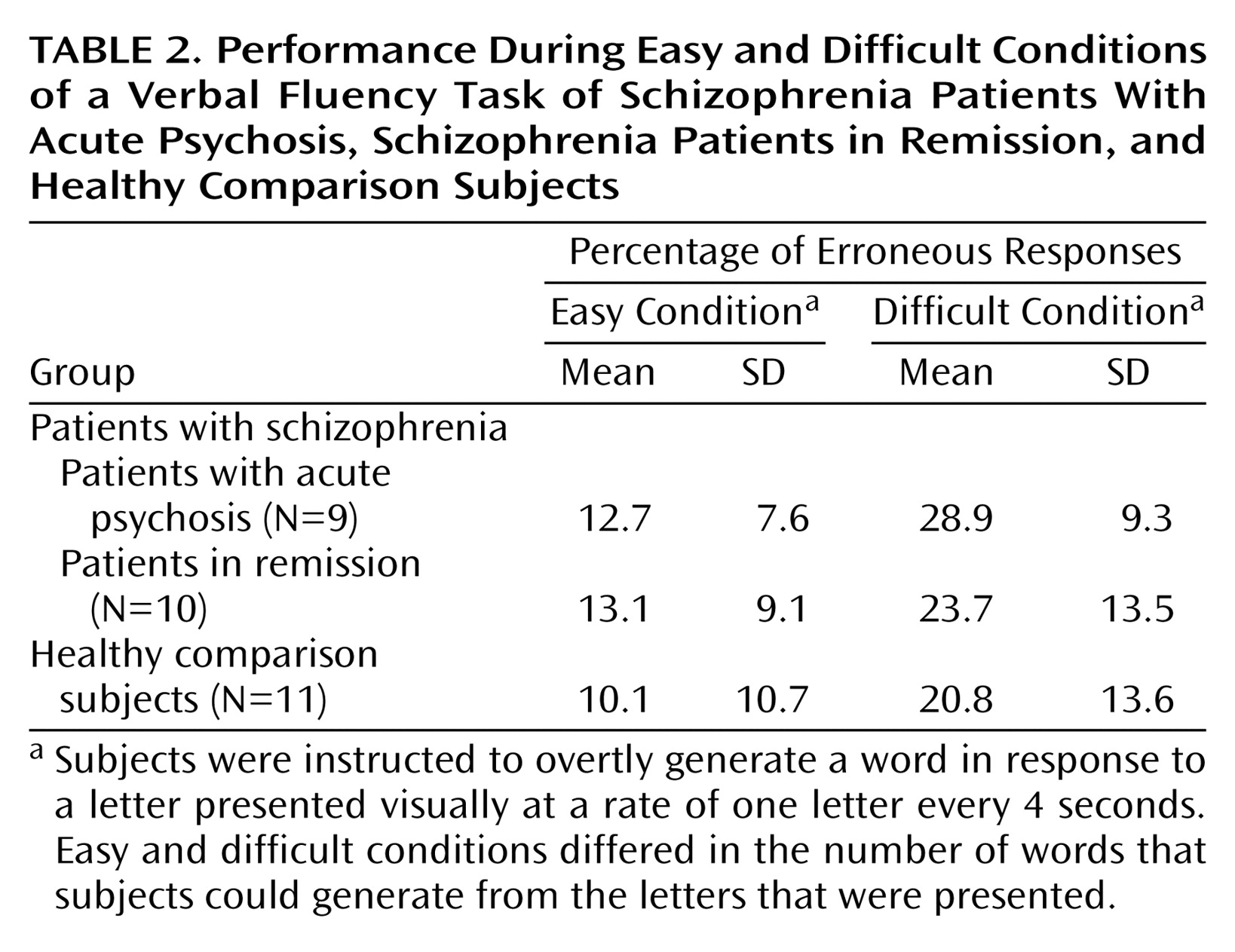
We examined the influence of psychotic state on activation by comparing patients with schizophrenia who had active psychosis and patients who were in remission. To investigate the effects of task demand, we used both “easy” and “difficult” letters as cues; the letters differed in the number of words that subjects could generate from them
(45). However, to minimize potentially confounding effects of between-group differences in task performance, we used a paced version of the task that allowed the patients to perform at a behavioral level similar to that of the comparison subjects, and we acquired overt verbal responses with a “clustered” functional magnetic resonance image (fMRI) acquisition sequence so that subjects could respond in the absence of scanner acoustic noise and task performance could be measured online.
We tested the hypothesis that, independent of psychopathology or task demand, patients with schizophrenia would show attenuated prefrontal activation, relative to healthy comparison subjects. As psychotic phenomena appear to be at least partly mediated by prefrontal areas, we predicted that this difference would be more marked in acutely psychotic patients than in those in remission. We also predicted that higher levels of task demand would exacerbate abnormalities of prefrontal activation in patients with active psychosis but would have little effect on activation in remitted patients.
Discussion
Verbal fluency is a classic neuropsychological test of language production in which subjects generate and articulate a word in response to a cue
(37,
38). Studies of verbal fluency with fMRI have revealed a consistent set of activated regions
(21,
25,
54–58), with the most frequently reported sites of frontal activation being the left inferior frontal gyrus
(25,
54–58), left middle frontal gyrus (dorsolateral prefrontal cortex)
(21,
25,
54,
56,
57), and anterior cingulate gyrus
(54,
58). The healthy comparison group in our study recruited all of these regions.
To assess task performance online we used a paradigm that involved overt speech. Image acquisition during fMRI is associated with an acoustic noise that can make it difficult for subjects and investigators to hear overt verbal responses
(59), and overt articulation can be associated with head movement
(60). We therefore employed a “clustered” acquisition sequence that incorporated brief periods of silence
(61,
62), which allowed subjects to speak and hear their responses in the absence of noise and reduced the risk of articulation-related movement artefacts
(45). Online measurement of behavioral responses indicated that all three groups were able to adequately perform the letter verbal fluency paradigm, with no significant between-group differences in the proportion of errors produced. We also covaried for task performance when comparing activation between groups.
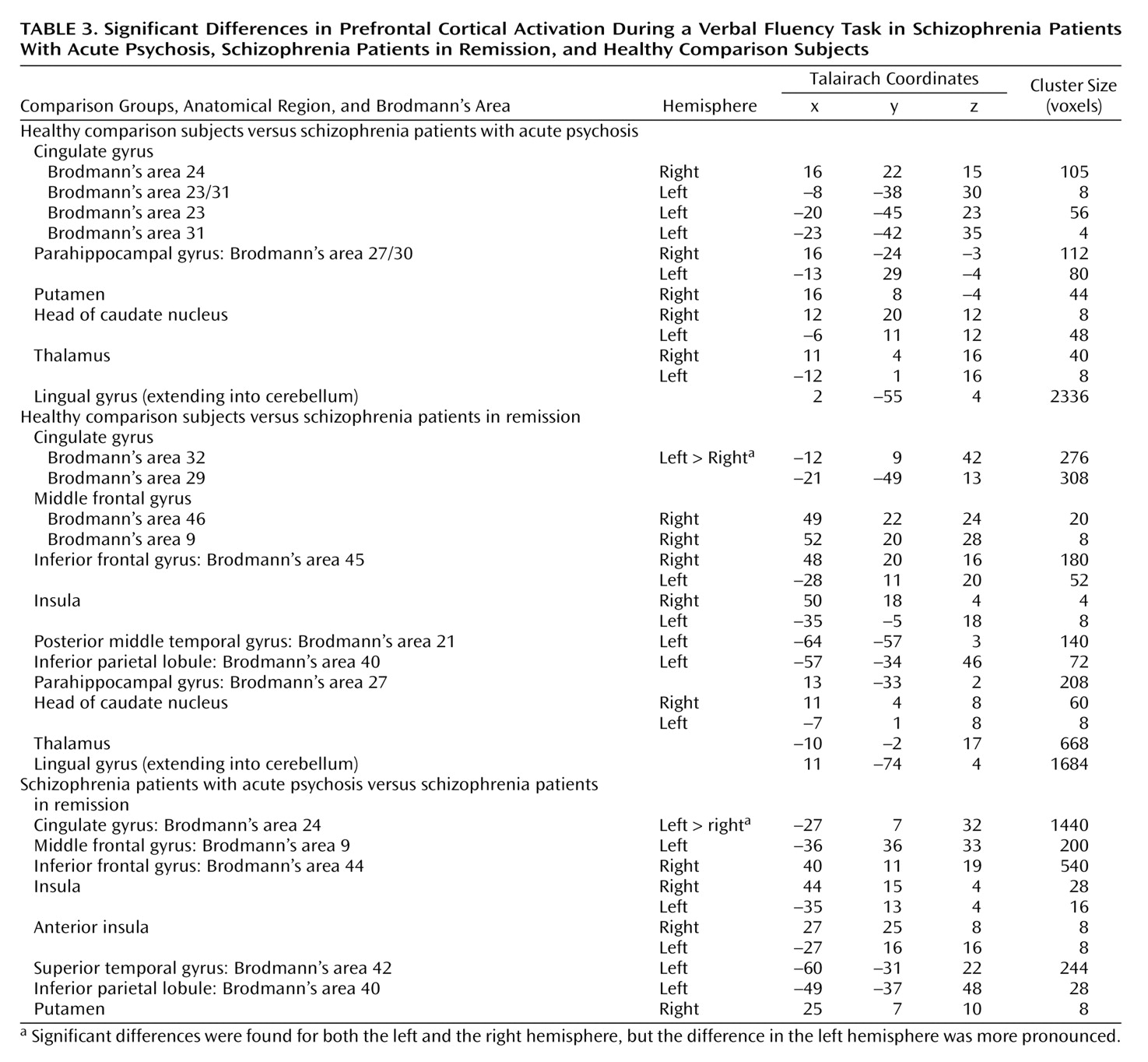
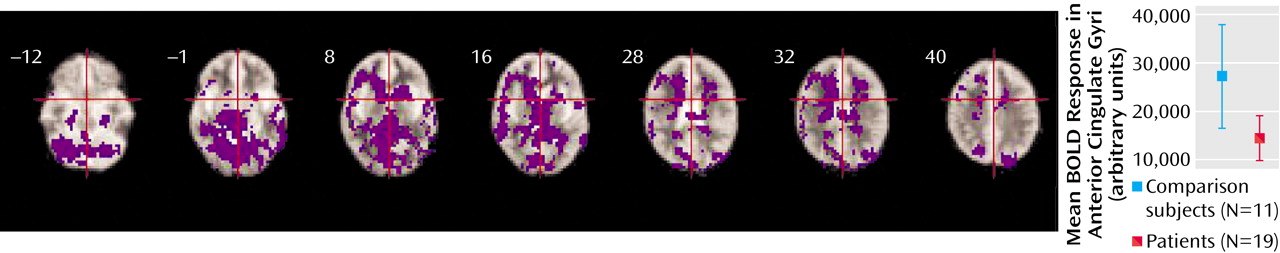
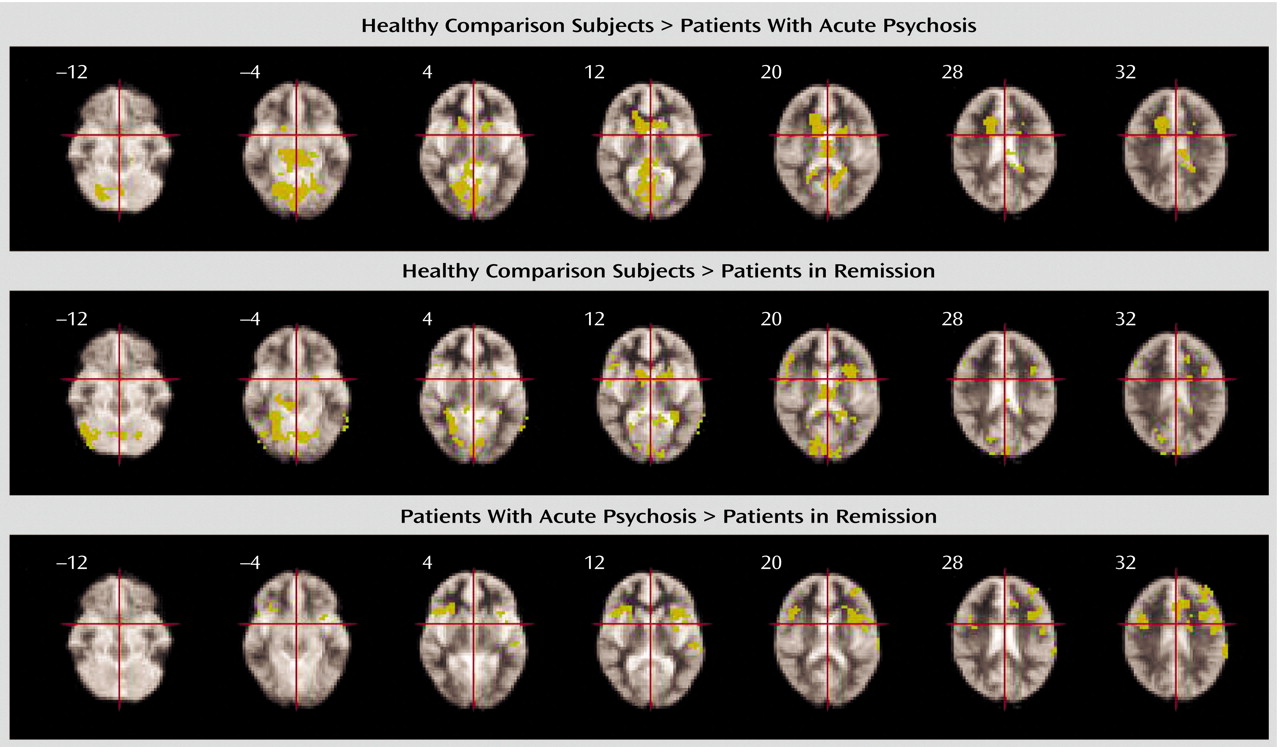
The schizophrenia group as a whole showed impaired activation, relative to the comparison group, in the inferior frontal and anterior cingulate gyri bilaterally and in the right but not the left middle frontal gyrus. These findings are broadly consistent with those reported in the literature, and the absence of differences in the left middle frontal gyrus is compatible with data from previous studies of verbal fluency that used a paced presentation of stimuli in which task performance was equivalent in the patient and comparison groups
(24,
33,
36,
42,
43). We predicted that abnormalities of prefrontal activation would be more evident in patients who were actively psychotic than in patients who were in remission. However, the group in remission showed a more marked attenuation of prefrontal activation, relative to the comparison subjects, than did the acute psychosis group, and direct comparison of the patient groups revealed that there was significantly
greater activation in the inferior frontal and dorsolateral prefrontal, insular, and anterior cingulate cortices bilaterally in the actively psychotic patients. Thus, although the patients in remission engaged these regions less than did the healthy volunteers, in the actively psychotic patients, less activation, relative to that in the comparison subjects, was evident in the anterior cingulate cortex, but not in lateral prefrontal areas. These observations may seem counterintuitive in the light of evidence that prefrontal function in schizophrenia is more impaired in patients with positive psychotic symptoms than those in remission
(44,
63,
64). However, compromised prefrontal function in schizophrenia may also be manifested as increased activation during cognitive processing. For example, Sommer et al.
(36) found a correlation between the severity of auditory hallucinations in schizophrenia and right prefrontal activation during verbal fluency. Moreover, Callicott et al.
(31) reported greater prefrontal activation in patients with schizophrenia than in comparison subjects during a verbal working memory task, and we have found that healthy volunteers with positive psychotic symptoms induced by ketamine show greater engagement of prefrontal areas during verbal fluency than when performing the task while taking placebo
(65). These observations are consistent with the notion that in patients with impaired prefrontal capacity, additional activation may be needed to maintain normal task performance
(31).
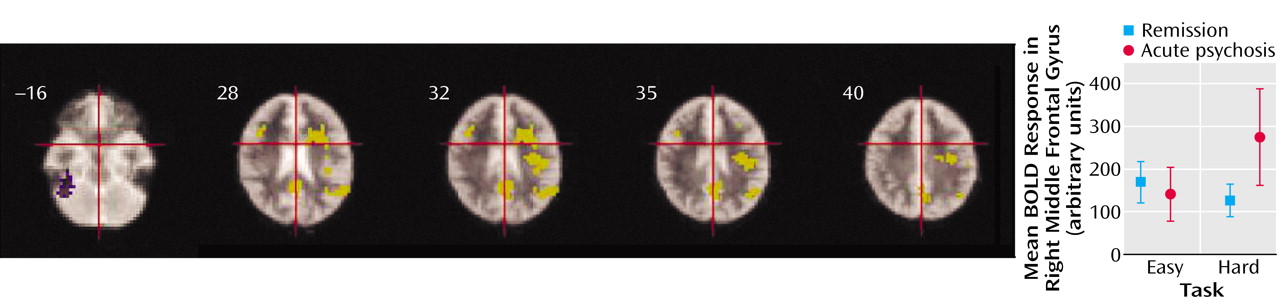
As predicted, abnormalities of prefrontal activation became more marked with increased task demand in patients with positive symptoms. However, comparison of the acute psychosis group and the group in remission revealed that rather than leading to a further reduction of activation, increasing demand in the symptomatic patients was associated with
greater engagement of the right prefrontal and anterior cingulate cortex. The findings cannot be easily attributed to thought disorder or other disorganization symptoms, as neither group had significant measures on these symptoms. Although we cannot exclude the possibility that we may have detected subtle degrees of formal thought disorder if we had used detailed instruments for this purpose, we think this outcome is unlikely. None of the patients had any previous history of formal thought disorder, and patients with formal thought disorder usually have a record of this symptom across the course of illness. Instead, these observations suggest that the effect of increasing the demands of verbal fluency in schizophrenia depends on the level of positive psychotic symptoms at the time of scanning. If actively psychotic patients need to engage the prefrontal cortex to a greater extent than those in remission to attain a given level of performance, this greater extent of activation may be particularly evident when the task becomes more demanding. In our study, patients were able to maintain adequate task performance, even when the demands were increased. However, had performance substantially deteriorated, the effects of task demand on activation may have been quite different. Thus, data from other studies indicate that attenuated engagement of the prefrontal cortex in schizophrenia is evident only when the task load has been increased to the extent that patients can no longer perform the task
(28,
30). Alternatively, we cannot exclude the possibility that the remitted patients differed from the psychotic group on neuropsychological performance, as formal measurements were not performed, and that this difference may have been related to their hypofrontality.
A further speculative explanation involves an influence of dopaminergic neurotransmission on cerebral blood flow and the blood-oxygen-level-dependent response that is measured in fMRI. Acute psychosis in schizophrenia, but not schizophrenia in remission, is associated with increased phasic dopamine release, a “hyperdopaminergic” state
(66–
69). The severity of central dopaminergic dysfunction appears to be correlated with abnormal prefrontal activation during the Wisconsin Card Sorting Test in schizophrenia
(70–
74). Experimental administration of dopaminergic agents modulates prefrontal activation during cognitive tasks
(75,
76), and their effects on activation during verbal fluency differ between patients with schizophrenia and comparison subjects
(42). Differential activation in prefrontal regions in patients with active psychotic symptoms, relative to patients in remission, may thus be related to a state-related perturbation of central dopaminergic activity, which might also underlie their positive symptoms. This possibility could be further investigated by directly comparing the effects of dopaminergic agents on prefrontal activation in actively psychotic patients and patients in remission.
One limitation of the present study was that all the subjects with schizophrenia were taking antipsychotic medication. Treatment with antipsychotic medications has been associated with increased metabolism in the basal ganglia
(77–
82). Their effect on activity in the prefrontal cortex is less clear, although Honey et al.
(83) reported that patients treated with risperidone showed greater activation during a verbal working memory task than when they were treated with typical antipsychotics. However, a differential effect of atypical versus typical antipsychotic drugs is unlikely to account for the differences between the patient groups in our study, as the proportions of patients who received atypical and typical antipsychotics in each group were comparable.
Overall our data suggest that while schizophrenia is associated with impaired prefrontal function, how this impairment is manifest in functional imaging studies of cognitive tasks can vary with the severity of psychotic symptoms at the time of scanning and the level of task difficulty. When patients can carry out a task as well as comparison subjects, the presence of active psychosis and increasing task demand may necessitate engagement of prefrontal areas in order to maintain behavioral performance. On the other hand, if the task is such that patients can no longer carry it out, the engagement of the prefrontal cortex may be attenuated.