Quetiapine (Seroquel) in the Treatment of Psychosis in Patients With Parkinson's Disease
Abstract
Case Report
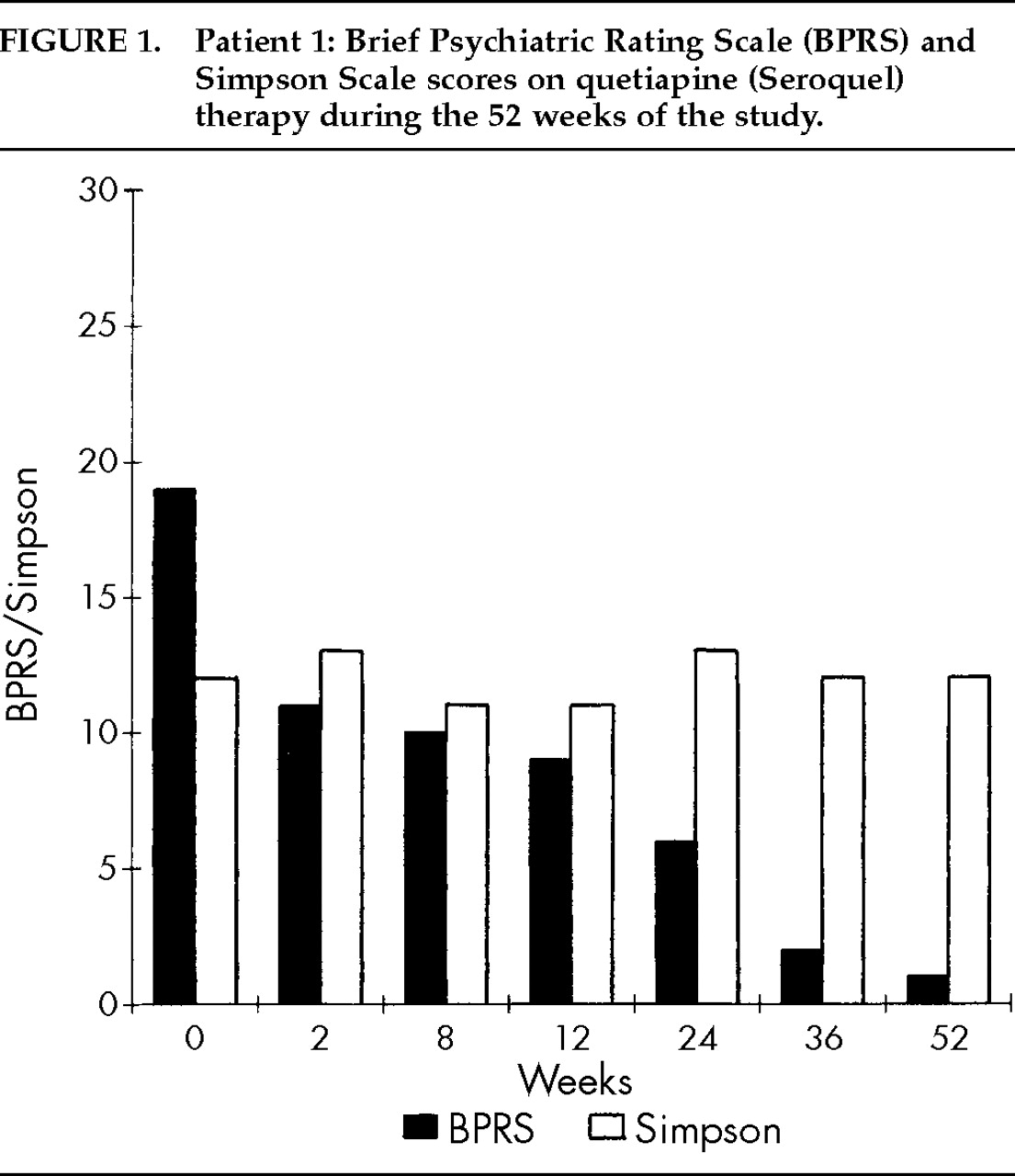
Patient 1. A 74-year-old man with a 2-year history of idiopathic PD and a long-standing history of affective bipolar disorder (well controlled on divalproex sodium) was referred for psychiatric evaluation because of marked visual hallucinations and persecutory delusions. His past medical history was also significant for hypertension and congestive heart failure. His medication regimen included carbidopa/levodopa 100/400 mg/day, furosemide 20 mg/day, doxazocin mesylate 2 mg/day, and divalproex sodium 1,250 mg/day (with valproate blood level ranging between 78 and 98 mg/l). The patient reported seeing people breaking into his backyard to steal his canoe and other belongings, and he was puzzled as to why his dog was not barking or attacking the intruders. He had called the police department on multiple occasions to arrest the intruders. His clinical assessment results included BPRS total score of 19 (where 0 on severity scale indicates absence of symptomatology), CGI-S score of 5 (“markedly ill”), Simpson Scale total score of 12 (10 items, with 1–5 severity scale for each item), AIMS total score of 0 (where 0 on severity scale indicates absence of tardive dyskinesia), UPDRS (motor examination section) total score of 14 (14 items, with 0–4 severity scale for each item), and MMSE total score of 28/30. Urine analysis and routine blood tests (chemistry, cell count, thyroid, VDRL, B12, and folate) were all within normal limits. Magnetic resonance imaging of the brain revealed mild cortical volume loss, with ischemic/degenerative changes in periventricular white matter and basal ganglia. ECG showed normal sinus rhythm with evidence of old inferior infarct.Quetiapine was introduced at 25 mg/day and gradually increased to a maximum dose of 400 mg/day over the subsequent 12 weeks. Delusions and hallucinations resolved almost completely, and quetiapine was well tolerated by the patient, without any worsening in PD. He maintained his improved mental condition and motor function throughout week 52. At completion of study, BPRS total score was 1, CGI-S score was 1 (“not at all ill”), Simpson Scale total score was 12, AIMS total score was 0, UPDRS (motor examination section) total score was 6, and MMSE total score was 29/30. Complete scores on BPRS and the Simpson Scale during the 52 weeks of study are summarized in Figure 1.
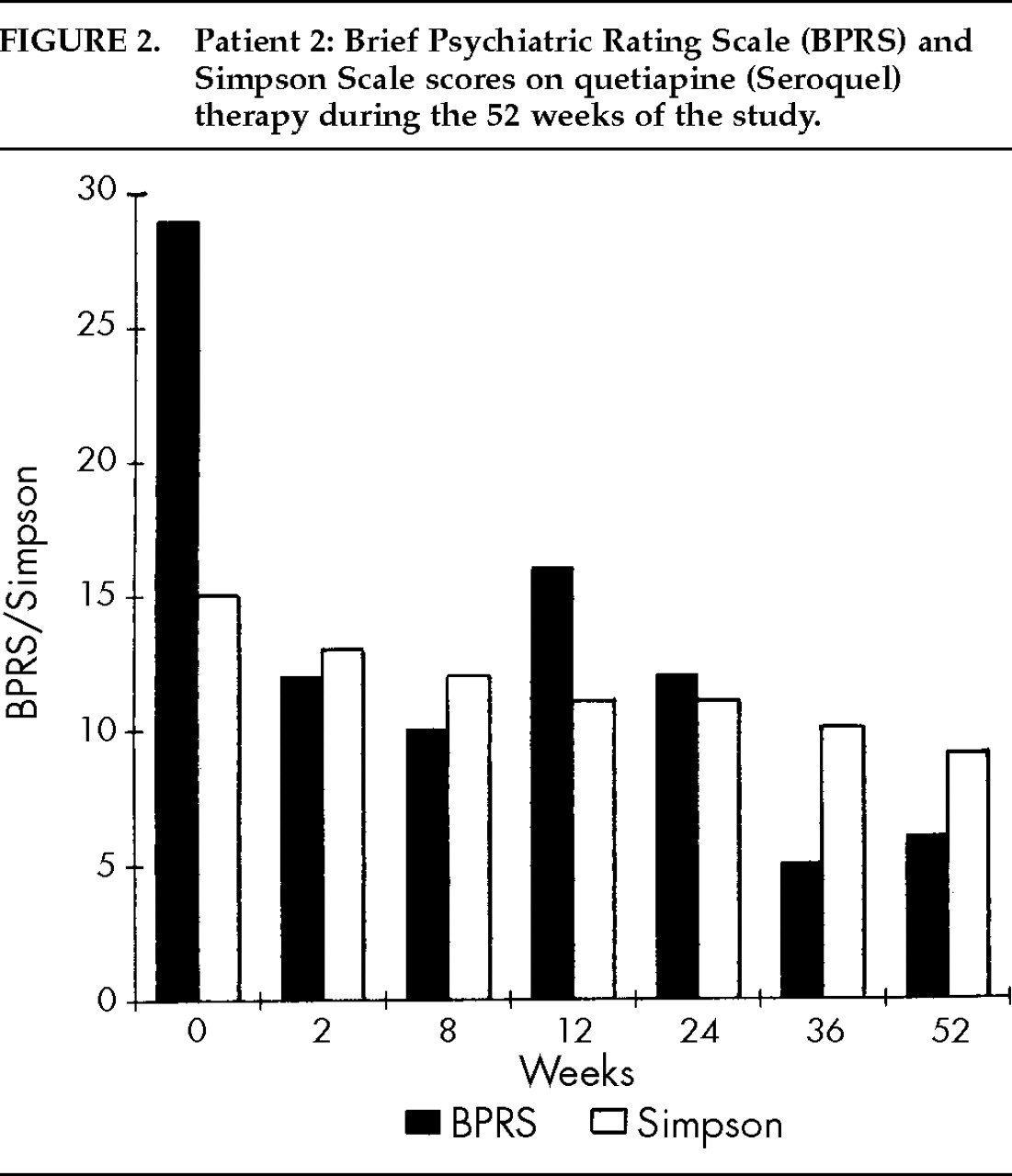
Patient 2. A 74-year-old woman who was diagnosed at age 68 as having idiopathic PD, was referred for psychiatric evaluation because of visual and auditory hallucinations and paranoid delusions. Her past medical history included hypothyroidism, hypertension, peptic ulcer disease, and idiopathic thrombocytopenia purpura, and she had suffered several falls as a result of PD-related postural instability. Her medication regimen was carbidopa/levodopa 75/300 mg/day, selegiline hydrochloride 10 mg/day, levothyroxine 0.05 mg/day, lisinopril 5 mg/day, and omeprazole 20 mg/day. The patient reported seeing animals running in her apartment and hearing her deceased husband whistling at night. In addition, she exhibited mild conceptual disorganization, mild cognitive and memory deficits, paranoid ideations, and anxiety symptoms. Her clinical assessment results included BPRS total score of 29, CGI-S score of 5, Simpson Scale total score of 15, AIMS total score of 0, UPDRS (motor examination section) total score of 19, and MMSE total score of 25/30. Urine analysis and routine blood tests (chemistry, cell count, thyroid, VDRL, B12, and folate) were all within normal limits, except for a platelet count of 78×1,000/mm3. ECG was normal. Brain imaging studies with SPECT using technetium-99m (Ceretec™) disclosed a symmetrical decrease in radiopharmaceutical uptake in bilateral temporal and posterior parietal regions, suggestive of dementia. Brain computed tomography scan showed moderate, diffuse cortical volume loss.Quetiapine was introduced at 25 mg/day and gradually increased to a maximum dose of 200 mg/day over the following 16 weeks. The patient's mental condition improved dramatically without significant change in parkinsonism or emergence of any significant side effects. Despite a gradual cognitive and memory deterioration due to a dementing process, she remained free of delusions and hallucinations over the subsequent 36 weeks. At completion of study, BPRS total score was 6, CGI-S score was 2 (considered as “borderline ill”), Simpson Scale total score was 9 (item number 1 for “gait” assessment was considered “not ratable” because of a gait disability related to previous hip fracture), AIMS total score was 0, UPDRS (motor examination section) total score was 23, and MMSE total score was 14/30. Complete scores on the BPRS and Simpson Scale during the 52 weeks of study are summarized in Figure 2.
DISCUSSION
ACKNOWLEDGMENTS
References
Information & Authors
Information
Published In
History
Authors
Metrics & Citations
Metrics
Citations
Export Citations
If you have the appropriate software installed, you can download article citation data to the citation manager of your choice. Simply select your manager software from the list below and click Download.
For more information or tips please see 'Downloading to a citation manager' in the Help menu.
There are no citations for this item
View Options
View options
PDF/ePub
View PDF/ePubGet Access
Login options
Already a subscriber? Access your subscription through your login credentials or your institution for full access to this article.
Personal login Institutional Login Open Athens loginNot a subscriber?
PsychiatryOnline subscription options offer access to the DSM-5-TR® library, books, journals, CME, and patient resources. This all-in-one virtual library provides psychiatrists and mental health professionals with key resources for diagnosis, treatment, research, and professional development.
Need more help? PsychiatryOnline Customer Service may be reached by emailing [email protected] or by calling 800-368-5777 (in the U.S.) or 703-907-7322 (outside the U.S.).

