N;t4>europathological and neuroimaging data indicate that disruption of frontal-subcortical circuits is critical for the development of affective and behavioral symptoms in individuals with and without dementia.
1 Positron emission tomography (PET) studies have shown that individuals with idiopathic depression have diminished glucose metabolism in the prefrontal cortex and caudate nuclei,
2 and that depressed Huntington's disease and Parkinson's disease patients have diminished metabolism in the orbitofrontal cortex compared with control subjects.
3,4 More recent studies conducted in elderly individuals with major depression have shown decreased (absolute) regional cerebral blood flow in the left anterior cingulate and left dorsolateral prefrontal cortex.
5 Furthermore, magnetic resonance imaging volumetric studies in elderly individuals have shown that minor depression is associated with smaller prefrontal volume
6 and major depression is associated with reduced frontal and temporal lobe volumes.
7 Damage to frontal-subcortical circuits may be a common substrate for affective disorders in late-life mood disorders,
7 and perhaps in other neurological diseases as well (e.g., Huntington's disease, Parkinson's disease).
Neuropathological studies in AD patients with depression have shown a greater neuronal loss in the central superior raphe nucleus, locus ceruleus, and substantia nigra than found in AD patients without depression.
8,9 However, single-photon emission computed tomography (SPECT) studies have shown that AD patients with depression have lower left temporoparietal blood flow than those without depression,
10 and PET studies have found lower metabolism in parietal lobes
11 or in the anterior cingulate gyrus.
12Emotional lability, described as episodes of crying that cannot be suppressed, has been reported in 10%
13 to 25%
14 of AD patients. AD patients with emotional lability have more depression, anxiety, and dysthymia than those without.
14 In addition, there is a greater subcortical atrophy in patients with affective pathological crying, which may indicate a greater frontal-subcortical dysfunction.
14Apathy, described as a lack of energy and anhedonia with or without depressed moods, has been reported in AD patients,
15 and SPECT studies have shown that AD patients with apathy have lower blood flow in the right temporal-parietal area
16 or frontal areas.
17,18 However, apathy appears to be more common in subcortical syndromes (e.g., Parkinson's disease, progressive supranuclear palsy).
19,20In this study we examined the hypothesis that depression, emotional lability, and apathy in AD patients are associated with cortical-subcortical dysfunction.
METHODS
We reviewed the psychiatric characteristics of 8 patients with the diagnosis of probable AD
21 who were participating in a longitudinal study of dementia at the University of Pittsburgh Alzheimer's Disease Research Center and who had completed a PET scan. Each patient underwent an extensive evaluation, including medical, neurological, psychiatric, social work/nursing, and neuropsychological evaluations, as previously described.
22 None of the patients had significant signs of cerebrovascular disease, and those with a score greater than 4 on the Hachinski Rating Scale
23 or evidence of possible infarction on neuroimaging studies (either CT or MR scan) were not included in this study.
We first examined the relative regional cerebral blood flow (rel-CBF) of the 8 AD patients and 9 nondemented elderly control subjects to determine specific differences between groups (
Table 1). None of the nondemented control subjects had experienced any psychiatric illness or were taking psychiatric medication. Second, the rel-CBF values of the 3 AD patients with mood disorders (1 with major depression, 1 with emotional lability, and 1 with apathy) were compared with those of 5 AD patients without major depression, emotional lability, apathy, psychosis, or mood-related disorders who were matched by education level and severity of dementia as measured by the Mini-Mental State Examination (MMSE).
24 All 3 patients were experiencing these symptoms at the time of the PET scan session, and none of them were taking psychotropic medication.
Psychiatric Assessment
The psychiatric evaluations were conducted by board-certified psychiatrists using a semistructured interview
25 with both patients and primary caregivers. The Hamilton Rating Scale for Depression
26 interview was completed by the psychiatrists on the basis of data from each patient and primary caregiver. The diagnosis of major depression was made according to the DSM-IV criteria.
27 Lability of mood was considered to be present when patients developed a sudden onset of laughing or crying that they were unable to suppress and that was associated with altered moods. We differentiated emotional lability from pathological laughing or crying when these symptoms occurred without alteration of the mood.
Positron Emission Tomography
Subjects were scanned in a Siemens 951R/31 PET scanner. At the end of the transmission scan, 40–50 mCi of [
15O]-water was injected as an intravenous bolus to measure rel-CBF. Data acquisition began 30 seconds after [
15O]-water injection. The actual rel-CBF values were calculated from the [
15O]-water time activity data by using a standard compartment model with correction for dispersion and timing delays in the blood activity curve.
28 The rel-CBF was examined in 9 regions of interest (ROIs): orbitofrontal, anterior cingulate, frontal dorsolateral, superior temporal, middle temporal, parietal, occipital, basal ganglia, and thalamus. The rel-CBF data for each ROI were normalized to the whole brain activity by computing an average of all of the ROIs and dividing each ROI by this denominator. The patients' MR scans and a neuroanatomy atlas
29 were used to identify the ROIs. The MR images were not digitally coregistered to the PET images, but they were used to visually guide the identification of the ROIs.
RESULTS
Nondemented control subjects showed higher rel-CBF in the left dorsolateral prefrontal cortex, right superior temporal, and right and left parietal cortex than AD patients. The MMSE
24 and Mattis Dementia Rating Scale
30 scores were higher in nondemented control subjects than in AD patients (see
Table 1).
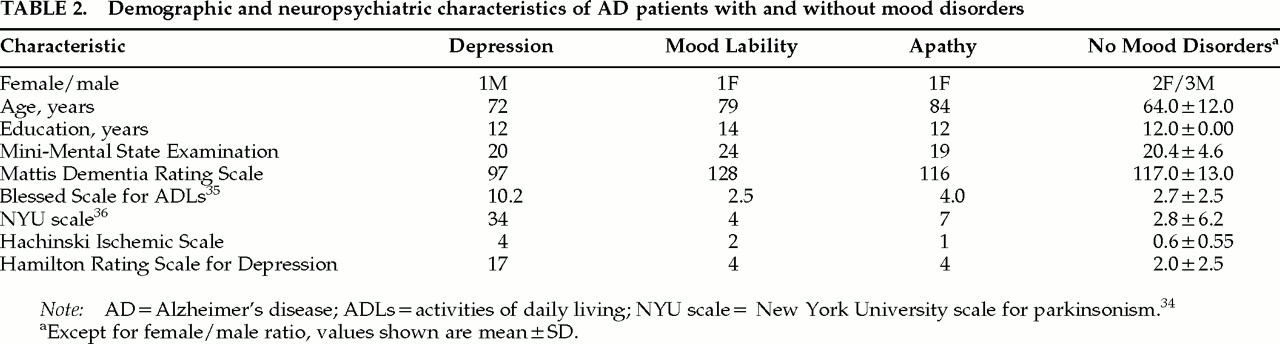
All AD subjects had at least 12 years of education. The patients were matched to their non–mood-disordered control subjects by age and MMSE
24 scores, but the patients with emotional lability and apathy were older than the AD control subjects. The demographic characteristics of the patients are shown in
Table 2.
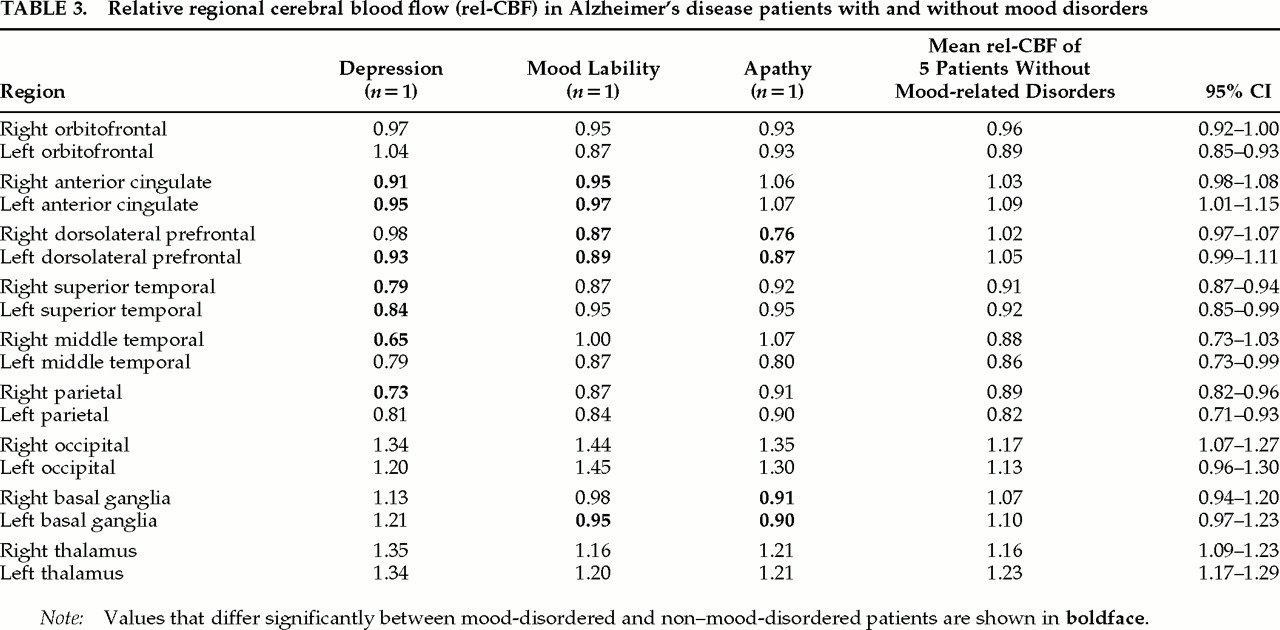
The rel-CBF comparisons between the two AD patient groups are shown in
Table 3. The patient with major depression had decreased rel-CBF in the anterior cingulate and superior temporal, bilaterally, as well as in the left dorsolateral prefrontal, right middle temporal, and right parietal cortices. The patient with emotional lability showed decreased rel-CBF in the anterior cingulate and dorsolateral prefrontal cortices, bilaterally, and in the left basal ganglia. The patient with apathy had decreased rel-CBF in the basal ganglia and the dorsolateral prefrontal cortex, bilaterally.
All three patients showed increased rel-CBF in occipital areas. In addition, the patient with depression showed increased rel-CBF in the thalamus; and the patient with apathy showed increased rel-CBF in the right middle temporal area (see
Table 3). These results are a consequence of normalizing the data relative to the whole brain, which accentuated the relatively higher rel-CBF found in these regions.
DISCUSSION
These findings support the hypothesis that a frontal-subcortical abnormality is associated with depression in dementia. It has been suggested that damage to the dorsolateral and orbitofrontal cortex serves as a common anatomic substrate in primary and secondary mood disorders in psychiatric and neurological disease. In this study, we examined the role of cortical-subcortical circuits in the etiology of depression, emotional lability, and apathy in patients with AD—where the primary site of abnormality is “cortical,” with subsequent subcortical involvement due to deafferentation.
The anterior cingulate abnormality in the depressed patient is consistent with abnormalities found in recent PET studies conducted in elderly nondemented individuals with major depression.
5 The presence of depression was also associated with bilateral temporal lobe hypoperfusion, which suggests the possibility of more widespread compromise of limbic structures. Indeed, nondemented untreated depressed subjects can have altered amygdala activity
31 and diminished temporal lobes and amygdalar cerebral blood flow.
32The patient with emotional lability showed decreased rel-CBF in anterior cingulate cortex and basal ganglia. This finding is related to the fact that AD patients with pathological affect have more atrophy of caudate nucleus than AD patients without these symptoms.
14 Furthermore, the patient with apathy showed a diminished rel-CBF in the right dorsolateral prefrontal cortex and basal ganglia. Studies of AD and Parkinson's disease patients have shown that the presence of apathy is associated with frontal lobe dysfunction.
15,17,18 Interestingly, in stroke patients frontal and temporal lobe hypoperfusion was associated with apathy when the latter occurred with symptoms of major depression.
33Although this is a small case series, these data add to the growing evidence that specific mood-related disorders in AD patients, like those in nondemented subjects, are associated with a mesolimbic system dysfunction. Specifically, there is dysfunction of the prefrontal and temporal cortices, with a subsequent involvement of their afferent and efferent connections to the ventral tegmental area, substantia nigra, and ventral striatum (ventromedial caudate and nucleus accumbens), areas that appear to mediate motivation and affect.
34ACKNOWLEDGMENTS
This study was funded by Grants AG03705 and AG05133 from the National Institute on Aging and Grants MH57078, MH49936, and MH01621 from the National Institute of Mental Health. J.T.B. is recipient of the Research Scientist Development Award, Level II (K02-MH01077).