Manic-depressive illness (bipolar affective disorder) is a common, severe, chronic, and often life-threatening illness
(1–
3). The discovery of lithium’s efficacy as a mood-stabilizing agent revolutionized the treatment of patients with manic-depressive illness
(1,
4,
5), but despite its role as one of psychiatry’s most important treatments, the cellular and molecular basis for lithium’s therapeutic effects remains to be fully elucidated
(6,
7). It has also been increasingly recognized in recent years that, although lithium has had a remarkable beneficial effect on the lives of millions
(5,
8), a significant percentage of patients respond inadequately
(9–
13). With the growing recognition that manic-depressive illness likely is a heterogeneous group of disorders, the importance of identifying predictors of differential treatment responsiveness or resistance has become increasingly appreciated. Although some important clinical leads have been identified
(10,
11,
13), there is at present a dearth of knowledge about the biochemical factors associated with lithium responsiveness or resistance. Thus, there is a clear need to elucidate the genetic and/or biochemical mechanisms associated with response or resistance to lithium’s actions, not only to better identify patients likely to respond to lithium treatment but also to facilitate the development of new therapeutic agents.
There has been considerable interest in the role of signal transduction pathways, in particular the phosphoinositide system, as potential biochemical targets of lithium’s actions
(5–
7,
14). Activation of a variety of receptors in the CNS induces the activation of phospholipase C isozymes
(15), which catalyze the conversion of phosphatidylinositol 4,5-bisphosphate (PIP
2) to two second messengers, inositol 1,4,5-trisphosphate and diacylglycerol. Inositol 1,4,5-trisphosphate stimulates the mobilization of intracellular Ca
+2, while diacylglycerol activates protein kinase C. Inositol 1,4,5-trisphosphate can be phosphorylated and sequentially dephosphorylated to
myo-inositol. The ability of a cell to maintain sufficient supplies of
myo-inositol is crucial to the resynthesis of the phosphoinositides and the maintenance and efficiency of signaling. Lithium, at therapeutically relevant concentrations, is an inhibitor of inositol monophosphatase and polyphosphate-1-phosphatase, which are involved in recycling inositol mono- and polyphosphates to
myo-inositol
(16,
17). Furthermore, since the mode of enzyme inhibition is uncompetitive, lithium’s effects have been postulated to be most pronounced in systems undergoing the highest rate of PIP
2 hydrolysis (reviewed in references
18 and
19). Thus, Berridge and associates
(20,
21) proposed the “inositol depletion hypothesis,” which posits that the therapeutic effects of lithium are mediated by a depletion of
myo-inositol. Since several subtypes of adrenergic, cholinergic, serotonergic, and metabotropic glutamatergic receptors are coupled to PIP
2 hydrolysis in the brain, the inositol depletion hypothesis, as initially proposed, offered an attractive explanation for lithium’s therapeutic efficacy in treating multiple aspects of manic-depressive illness
(18–
21).
However, although the preclinical studies in toto have tended to demonstrate lithium-induced alterations in receptor-mediated phosphoinositide turnover, numerous methodological questions have been raised (see excellent review by Jope and Williams
[6]). In addition, the temporal dissociation between lithium’s direct inhibition of inositol-1-phosphatase and its delayed clinical effects has led to the proposal that inositol depletion per se may not be responsible for lithium’s therapeutic effects
(7,
22). However, a large body of preclinical data suggests that some of the initial actions of lithium may occur with a reduction of
myo-inositol
(23–
26); this lowering of
myo-inositol may initiate a cascade of secondary changes at different levels of the signal transduction process and gene expression in the CNS, effects that are ultimately responsible for lithium’s therapeutic efficacy
(6,
7,
22). Despite the attractiveness of this hypothesis, to our knowledge it has never been investigated in patients with manic-depressive illness. Thus, there is a clear need to determine whether lithium reduces the levels of
myo-inositol in critical brain regions of individuals with manic-depressive illness and whether individual differences in susceptibility to lithium-induced CNS
myo-inositol reductions are major factors in predicting therapeutic efficacy.
A variety of neuroimaging studies have begun to provide important clues to the neuroanatomical basis of manic-depressive illness, and several converging lines of evidence indicate that abnormalities in the frontal and temporal cortices may play a role in the pathophysiology of manic-depressive illness
(27–
35). Recent developments in magnetic resonance spectroscopy (MRS) allow for the direct and noninvasive monitoring of brain neurochemistry. In this study we used MRS to quantitatively measure regional brain
myo-inositol concentrations at baseline and throughout the course of lithium treatment of bipolar depressed patients. It should be pointed out that the “
myo-inositol resonance” measured by proton MRS, while predominantly
myo-inositol, also contains minor contributions from other neurochemicals, including glycine and inositol-1-phosphate. We acknowledge the potential confounds presented by the compounds glycine and inositol-1-phosphate. However, on the basis of literature findings
(36,
37) and our own data (not shown), we believe these contribute only a minor component (<5%) to the total
myo-inositol resonance. In the present study we investigated the hypothesis that lithium reduces the levels of
myo-inositol in the critical brain regions implicated in this illness in individuals with manic-depressive illness. In addition, we sought to determine whether any potential reductions in
myo-inositol per se are associated with lithium’s therapeutic effects.
METHOD
Subjects
Adult patients were eligible for this study if they gave written informed consent according to procedures approved by the institutional review board, met the diagnostic criteria for bipolar mood disorder, and the most recent episode had been depression. The diagnosis was determined by using the Structured Clinical Interview for DSM-IV
(38). Patients were excluded if they met the diagnostic criteria for any other DSM-IV axis I disorder during the 2 years preceding the index episode. In addition, patients with psychoactive substance abuse or dependence within 1 year of the index episode were excluded (patients with episodic abuse related to manic-depressive illness were not excluded). Patients were also excluded from the study if they had renal disease, hepatic disease, or hematological disease, which put them at greater risk for side effects from lithium, or if they had any of the following, which put them at greater risk for side effects from the MRS procedure: cardiac pacemaker, brain surgery for an aneurysm, recent major surgery, a neurostimulator, or metal fragments in or near the eye or brain.
The effects of lithium on regional brain myo-inositol levels were investigated in 12 patients who met the preceding criteria. Their mean age was 36.3 years (range=22–56), and the group contained seven women and five men. Eleven of these patients had bipolar I disorder (history of major depression plus mania), and one had bipolar II disorder (history of major depression and hypomania). Upon admission the subjects were administered blinded research capsules four times per day, any previous medications were tapered off (through these capsules), and the patients underwent a drug washout period that was at least 14 days long (depending on the half-lives of the previous medications). On completion of the washout period, the patients’ symptoms were reassessed with the Hamilton Depression Rating Scale by trained blinded raters. All patients remained depressed after the washout period (Hamilton depression scale score: mean=18.75, range=11–29). Each patient then underwent a baseline MRS scan (methods to be described) before the initiation of lithium treatment through research capsules. Lithium treatment was initiated, and the dose was adjusted to obtain a therapeutic plasma level (0.8–1.2 meq/liter) over the first week of treatment. Brain myo-inositol levels were measured in these inpatients at three different time points by means of quantitative proton (1H) MRS: at baseline and after acute (5–7 days) and chronic (3–4 weeks) lithium administration by blinded personnel. The Hamilton depression scale was also administered by blinded raters at each of the MRS scan time points. The patients were maintained on a standardized low-monoamine diet throughout the study.
MRS Protocol
Quantitative single-voxel
1H MRS examinations were performed by using a 1.5-T clinical scanner (Signa/Horizon 5.6, General Electric, Milwaukee). A stimulated echo acquisition mode pulse sequence
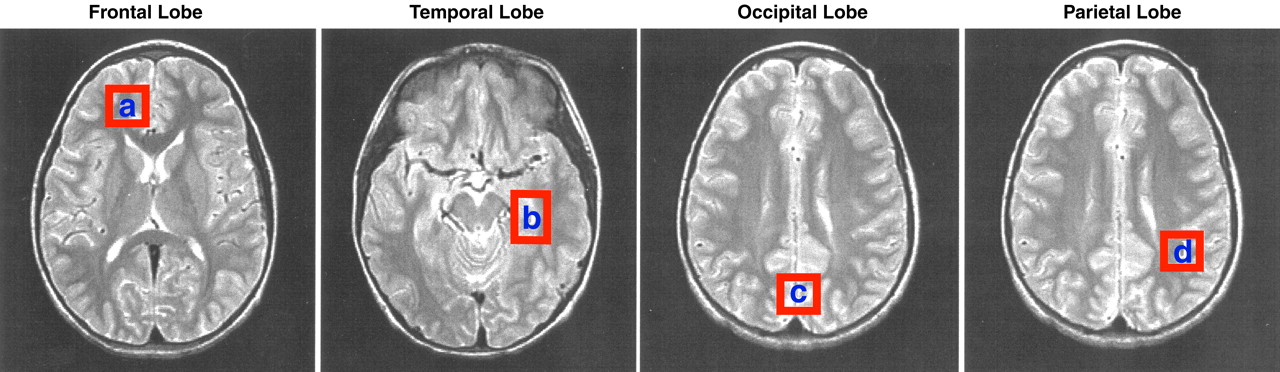
(39) was used to acquire spectra by means of the following acquisition variables and also included unsuppressed water reference scans for neurochemical quantitation: an echo time of 30 msec, a modulation time of 13.7 msec, a repetition time of 2 sec, an eight-step phase cycle, 2048 points, a spectral width of 2500 Hz, and 128 averages for a total acquisition time of approximately 5 minutes. Spectra were acquired from approximately 8-cc regions of interest in the right frontal, left temporal, central occipital, and left parietal lobes (
Figure 1). Special care was taken to place the regions of interest in identical locations at the three time points (baseline, acute treatment, chronic treatment) by using a systematic approach that referenced voxel position to readily identifiable anatomical gyral landmarks within the brain.
Additional procedures were undertaken to evaluate the precision of the voxel placement from scan to scan in this longitudinal study and to control for potential partial-volume effects, which may potentially confound any MRS findings. We used a simple robust semiautomated image segmentation approach to determine the relative percentage of the various components, namely gray matter, white matter, and CSF, making up the voxel. Voxel content within each region of interest remained stable across the three time points; repeated measures analysis of variance (ANOVA) revealed there was no significant difference over time for gray matter, white matter, or CSF. Individual analysis of the combined data for voxel tissue content over time demonstrated that the voxel content was highly correlated and highly significant (df=59, p<0.0001) in all comparisons (baseline versus acute, r=0.96; baseline versus chronic, r=0.93; acute versus chronic, r=0.93).
Quantitative MRS Analysis
The compounds that were identified in the short-echo
1H MRS brain studies were
N-acetylaspartate, glutamine/glutamate/γ-aminobutyric acid (GABA), creatine/phosphocreatine, choline compounds, and
myo-inositol. The area under each of the resonances is proportional to the concentration of the specific neurochemical compound. Individual peak areas were fit by using time-domain analysis software
(40,
41), and the concentration of each compound was multiplied by 10,000. The result is reported as the ratio to brain water concentration (×10
4/water). This water-referencing method has been used in the field for over a decade and has been validated by a number of research groups
(42–
49). The analysis software is publicly available (http://carbon.uab.es/mruiwww) and eliminates much of the subjectivity previously involved in determining spectral peak areas.
Briefly, the software performs an automated fit of the unsuppressed water peak to determine its peak area and also uses the phase of the water peak to apply an automated zero-order phase correction to the metabolite data. After this, the user enters a priori information regarding the metabolite data in order to give the software starting values for its fitting process. The a priori information given includes the expected chemical shifts for each of the major chemical compounds appearing in the typical proton brain spectrum, as well as a starting line width determined by the corresponding water line width. The chemical shift values given to the program are based on literature values, which are 2.02 ppm for
N-acetylaspartate, 2.30 ppm for the glutamine/glutamate/GABA complex, 3.03 ppm for creatine/phosphocreatine, 3.22 for choline compounds, and 3.56 for
myo-inositol
(50–
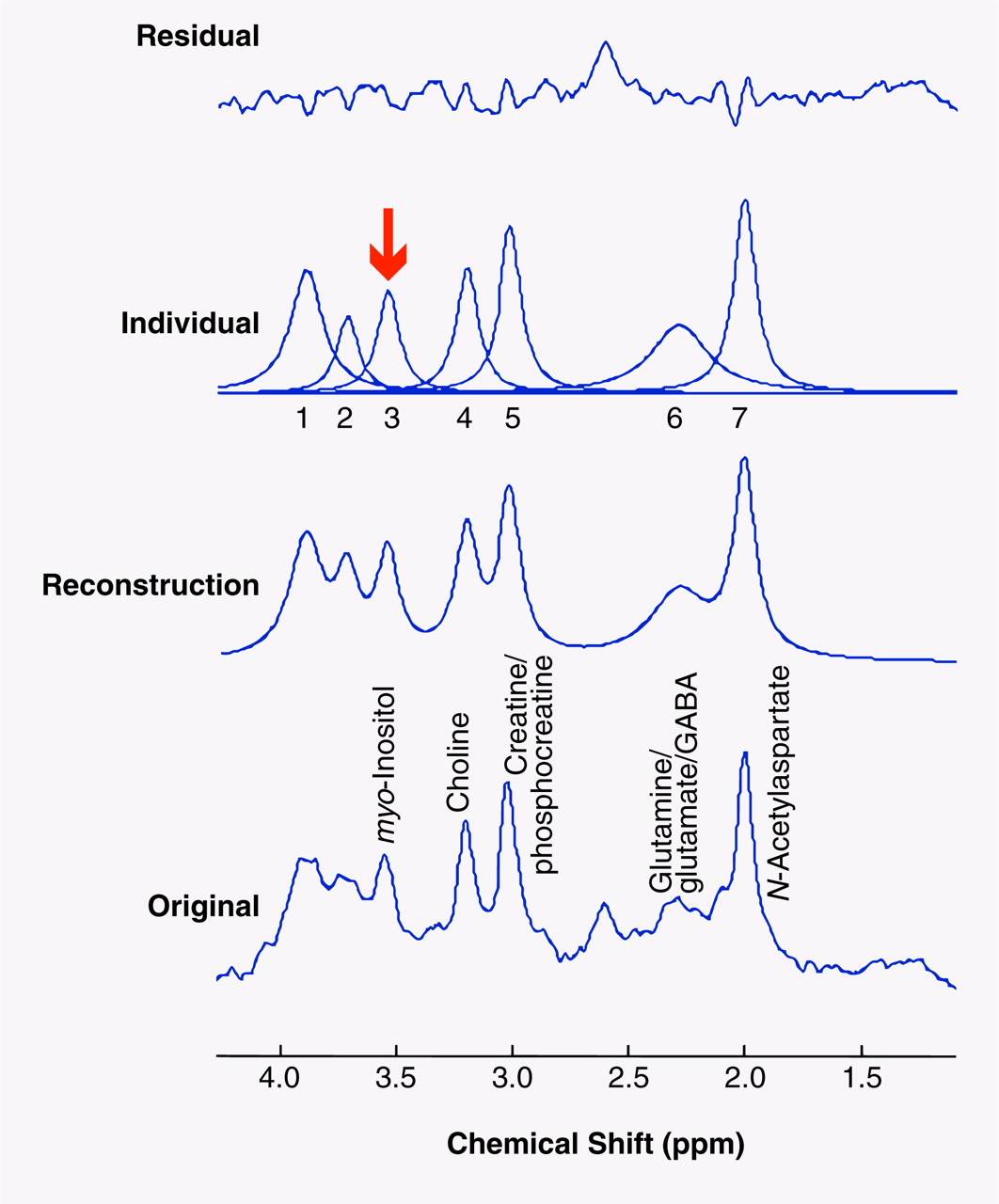
54). With this input, the software then attempts to fit the metabolite spectrum and display its results both visually and in a file that can be pasted into a spreadsheet analysis program. In order to achieve reliable fits for
myo-inositol, one must also fit the additional and partially overlapping peaks containing creatine/phosphocreatine and glutamine/glutamate/GABA in the immediate area of
myo-inositol. (These are the peaks labeled 1 and 2 in figure 2, respectively.) The visual and quantitative results are inspected for goodness of fit and either accepted or reiterated again for improvement. Most of the spectra (approximately 80%) in this study required only one iteration apiece to achieve a satisfactory fit, with the majority of the others being successful after two iterations. The visual output of the program from a typical spectrum fit is shown in
Figure 2. The analyzers are trained to accept a fit if the residual shows predominantly unstructured noise. The areas of the water peaks and neurochemical peaks are then entered into a spreadsheet, and the data for the individual neurochemical peaks are then multiplied by 10,000 (factor chosen for convenience in reporting data) and then divided by the unsuppressed brain water peak area (×10
4/water). We specifically did not attempt to correct for water and neurochemical relaxation effects with this technique, as obtaining these values on each of our patients would be prohibitively time consuming (measurement would take an additional 2 hours for each subject). We did, however, use acquisition variables that minimized the uncertainty in our neurochemical concentration estimates due to relaxation effects. Specifically, we used a short echo time, 30 msec, to minimize T
2 signal decay and a standard repetition time of 2 seconds in order to minimize T
1 error resulting from collection of spectra under less than fully relaxed conditions. This is a common tradeoff in clinical research studies.
Two research assistants (J.K.P. and M.W.F.) trained in nuclear magnetic resonance (MR) spectral analysis using this protocol evaluated the data in this study. The individuals were blind to the study information and to each other’s results. Intraclass correlation coefficient analysis revealed an interrater reliability of greater than 98% for in vivo quantitative measurement of brain
myo-inositol concentration. We did not attempt to separate out the various minor components of the
myo-inositol resonance, including inositol-1-phosphate and glycine. However, as discussed in the introduction, it is unlikely that these compounds contribute more than 5% of the
myo-inositol resonance. The variability of our MRS measurement of brain
myo-inositol (in normal volunteers) assessed at different time points has been described elsewhere
(55) and is less than plus or minus 5%.
Data Analysis
Temporal changes in the myo-inositol concentrations were assessed by using within-subjects repeated measures ANOVA for each region of interest and time point separately. When there was violation of the sphericity assumption, the degrees of freedom were adjusted with the Greenhouse-Geisser epsilon. Post hoc tests with Bonferroni correction were used to identify significant differences between the scans at baseline, acute treatment, and chronic treatment. All reported p values are two-sided.
RESULTS
Of the 144 potential in vivo proton brain spectra (four brain regions at three time periods for each of the 12 subjects), 128 (89%) were available and judged to be of adequate quality to undergo quantitative analysis. Two subjects did not complete the scans during chronic treatment (loss of eight spectra), and eight other spectra were discarded because of poor quality due to inadequate water suppression, subject motion during the scan, or magnetic susceptibility artifacts.
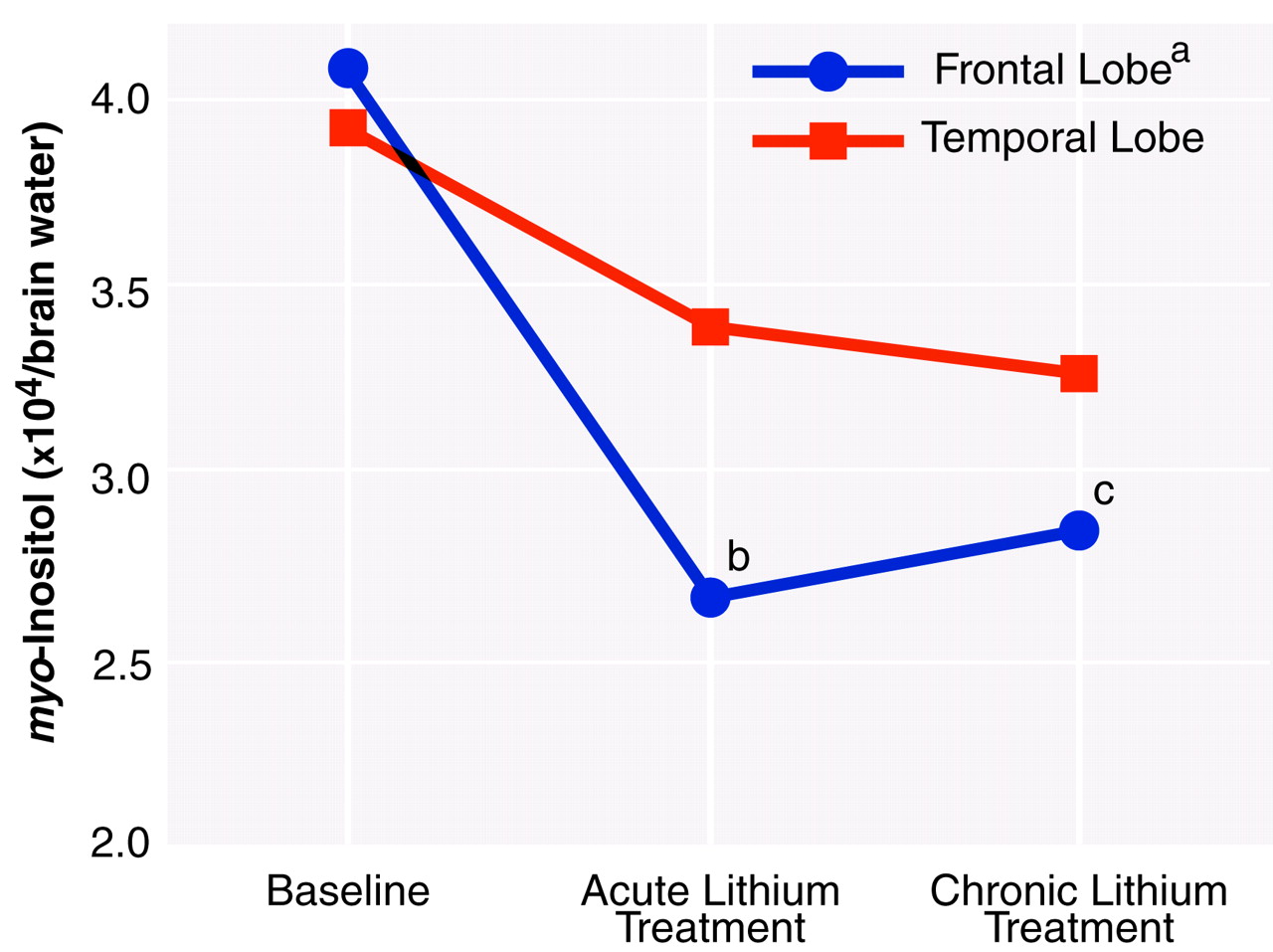
Figure 3 is a graph of the proton MRS measures of
myo-inositol concentration at each time period for the frontal and temporal lobes. Overall, the
myo-inositol concentration in the frontal lobe showed significant changes over time. Specifically, in the right frontal lobe the
myo-inositol concentrations during both acute and chronic treatment were significantly lower than at baseline before we corrected for multiple comparisons. After correction, the two contrasts failed to reach the nominal level of significance. Although the temporal lobe data appeared to show a pattern somewhat similar to that for the frontal lobe, they failed to reach statistical significance. The temporal lobe data, however, had larger standard deviations, reflecting the difficulty in obtaining accurate measurements because of magnetic susceptibility artifacts from nearby sinuses and bone. No significant differences or trends were observed in the occipital and parietal lobes.
Because
myo-inositol plays an important role in osmotic equilibrium, in addition to its role in the phosphoinositide signaling system, it may be possible that other neurochemicals are increasing as
myo-inositol decreases. We did observe a change in another neurochemical measure within the frontal lobe region. Choline compounds showed a significant change in the frontal lobe over the course of lithium administration; however, the observed change was also a decrease. This observation has recently been presented in preliminary form
(56); however, a complete discussion of this interesting finding is beyond the scope of this paper. There was no significant change or trend in the creatine/phosphocreatine concentration (commonly used as an internal MRS standard) in any of the brain regions investigated over the course of lithium administration.
Analysis of the scores on the Hamilton Depression Rating Scale at each of the time points corresponding to the MRS scans revealed a significant mean decrease (–36%) in the severity of depression over the course of the study (F=4.64, df=2, 18, p=0.02). The score during chronic lithium treatment was significantly lower than the score at baseline (t=3.58, df=9, p=0.006).
DISCUSSION
In this longitudinal study we have shown for the first time, to our knowledge, that lithium treatment reduces
myo-inositol levels in the right frontal lobe. The major effects were observed early in treatment and are consistent with lithium’s acute inhibition of inositol-1-phosphatase, which has been demonstrated in preclinical studies. However, the patients’ clinical state was clearly unchanged at this time, indicating that lowering of
myo-inositol per se does not underlie lithium’s therapeutic effects. It is a heuristic working hypothesis that some of the initial actions of lithium may occur with a reduction of
myo-inositol and that this reduction of
myo-inositol initiates a cascade of secondary changes in the protein kinase C signaling pathway and gene expression in the CNS, effects that are ultimately responsible for lithium’s therapeutic efficacy
(5,
6).
As discussed previously, the frontal lobe is one of the areas that has been implicated in the pathophysiology of this illness. Recent imaging and postmortem studies of this region
(29–
32) have documented both neuronal and glial cell abnormalities in subjects with familial manic-depressive illness. The frontal lobe region of interest used in this MRS study is larger than the areas examined in the imaging and postmortem studies and contains large proportions of both white and gray matter. However, it is interesting that the largest neurochemical changes we found are in this same general region. Although not as striking as the frontal lobe findings, the region of interest in the left temporal lobe (a brain region also implicated in manic-depressive illness) also showed similar overall effects; however, these failed to reach statistical significance. Because of the greater inherent variability of the data acquired from the temporal lobe region, a larger number of patients is needed to definitively determine whether there is indeed a reduction of
myo-inositol in this region with lithium administration. In the two control brain regions (e.g., regions not strongly implicated in the pathophysiology of manic-depressive illness)—occipital and parietal lobes consisting predominantly of gray and white matter, respectively—we observed no significant changes or trends in
myo-inositol levels during the course of lithium treatment, suggesting a local rather than “whole brain” phenomenon. Intriguingly, this finding is consistent with the uncompetitive nature of lithium’s inhibition of inositol-1-phosphatase
(18–
21), which suggests that the regions of the brain most affected by the pathophysiology of the illness are the most susceptible to lithium-induced
myo-inositol changes. It has to be acknowledged, however, that the parietal and occipital lobes showed lower basal
myo-inositol levels; thus, a role of “floor effects” in lithium’s regionally selective effects cannot be entirely ruled out. Although we observed changes in the 3.56-ppm region of the proton MR spectrum, which is entirely consistent with a lithium-induced lowering of the
myo-inositol concentration, we again acknowledge that there may be minor contributions in this region of the spectrum from glycine and inositol-1-phosphate. However, any modest lithium-induced increases in inositol-1-phosphate in this peak that may be occurring suggest that we may, in fact, be underestimating the changes in
myo-inositol levels.
We are aware of few previous studies in which lithium’s effects on CNS
myo-inositol levels have been examined. Indirect CSF measures in one previous study
(57) did not show any differences in CSF
myo-inositol levels between lithium-treated patients and healthy comparison subjects; by contrast, another study
(58) showed a lithium-induced reduction in CSF inositol monophosphatase activity. Given the regional brain differences in lithium’s effects that we found in our study, it is perhaps not altogether surprising that the indirect CSF studies have not revealed consistent findings. There is one other study in which lithium’s effects on CNS
myo-inositol were examined by using MRS. Silverstone and associates
(59) did not observe any significant effects of 7 days of lithium administration on
myo-inositol levels in the temporal lobe of healthy volunteers. Silverstone et al. did not investigate lithium’s effects in the frontal lobe, and the study has a number of additional major methodological differences from our present study, including the use of
myo-inositol/creatine ratios and healthy volunteers rather than patients with manic-depressive illness. Nevertheless, their results are consistent with the results of our present study.
Like investigators in previous studies
(60–
63), we found that even with a small study group, 4 weeks of lithium administration was associated with a significant reduction in Hamilton depression scale scores in our bipolar depressed patients. The authors of a comprehensive review of nine published studies
(64) concluded that lithium is markedly more effective than placebo in the treatment of bipolar depression; however, those authors noted that the antidepressant effects demonstrated in the placebo-controlled studies often did not become evident until the third or fourth week of treatment. Given the now well-documented ability of antidepressants to precipitate manic episodes and to induce rapid cycling
(65–
69), lithium monotherapy, when possible, may indeed be the treatment of choice for bipolar depression for many (but not all) patients. Thus, delineating the biochemical processes underlying the ultimate responsiveness or resistance to lithium’s antidepressant effects is clearly a worthwhile endeavor. The observation that the administration of large doses of
myo-inositol may have antidepressant efficacy in some patients
(25,
70,
71) highlights the overall complexity of the system. The limited number of patients studied to date does not allow for comparison of differences between responders and nonresponders at this point. This, however, is clearly an interesting question worthy of further investigation in a larger study group.
In conclusion, a decrease in myo-inositol concentration with acute lithium administration in the right frontal lobe of patients with manic-depressive illness was clearly documented. Furthermore, lowering of myo-inositol levels per se did not appear to be associated with therapeutic efficacy. It remains a working hypothesis that the initial reduction of myo-inositol initiates a cascade of secondary changes in the protein kinase C signaling pathway and gene expression in the CNS, effects that are ultimately responsible for lithium’s therapeutic efficacy. Clearly, further study is warranted to delineate the role of lithium-induced reductions in regional brain myo-inositol levels and to determine whether, indeed, acute lithium-induced changes in brain myo-inositol concentration can predict an individual’s ultimate response or nonresponse to this agent.