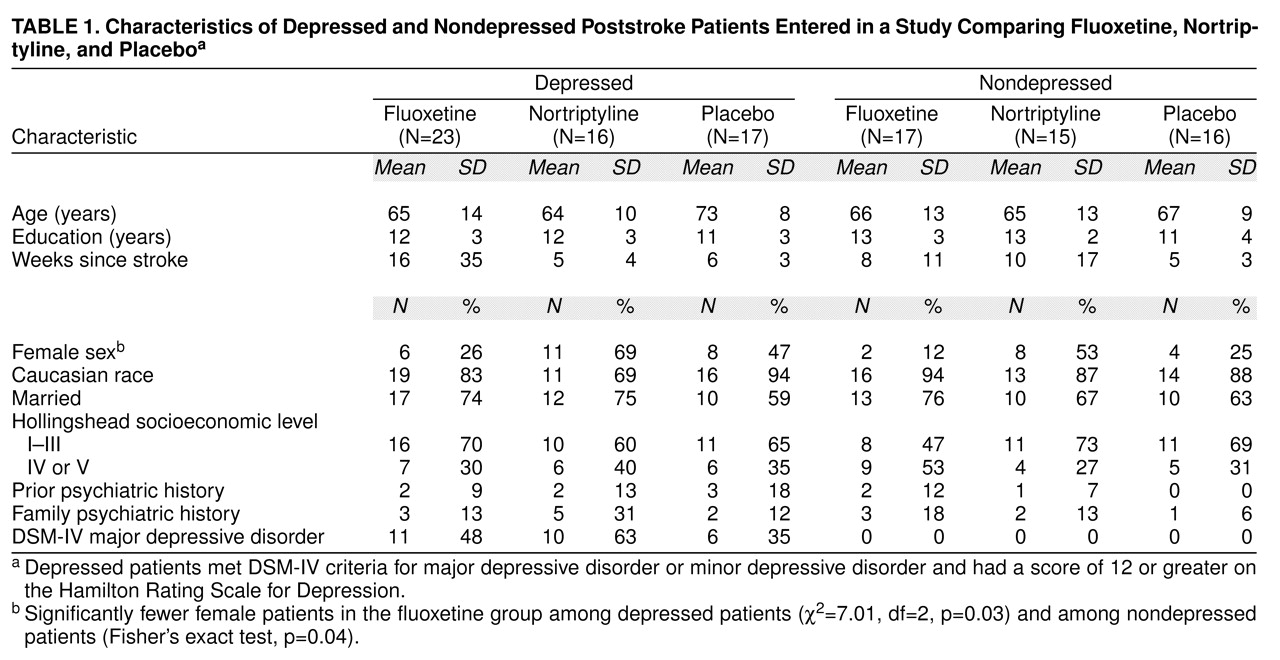
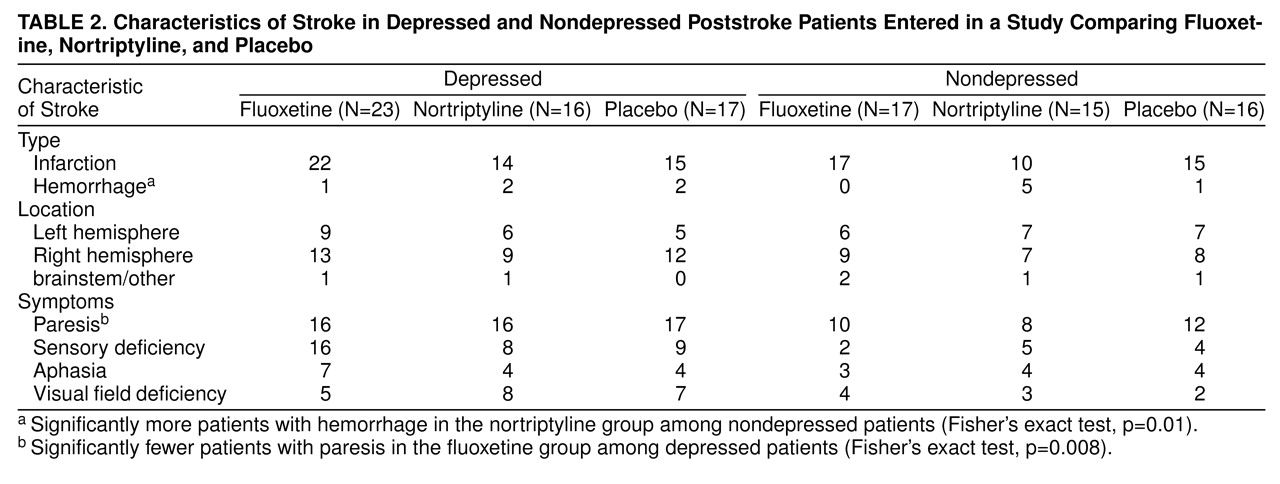
Intention-to-Treat Analysis
Of the nine withdrawals in the depressed fluoxetine group, six dropped out within the first 3 weeks, two dropped out between weeks 4 and 6, and one dropped out between weeks 7 and 9 (three complained of gastrointestinal symptoms, and six refused treatment). Of the depressed nortriptyline patients, one dropped out during the first 3 weeks, one dropped out between weeks 7 and 9, and one dropped out between weeks 10 and 12 (two had medical deterioration, and one refused treatment). In the depressed placebo group, four patients dropped out during the first 3 weeks (one patient died due to pulmonary embolus, one deteriorated medically, and two refused treatment).
In the nondepressed fluoxetine group, four patients dropped out—two during the first 3 weeks, and two between weeks 4 and 6. (One complained of gastrointestinal symptoms, two deteriorated medically, and one refused treatment.) Of the nondepressed nortriptyline group, two patients dropped out—one during the first 3 weeks and one during weeks 4–6. (One refused treatment, and one had sedation.) One of the nondepressed placebo patients dropped out between weeks 7 and 9. (The patient developed a rash.) Combining the dropout rates for both depressed and nondepressed patients, the dropout rate was significantly greater in the fluoxetine group than in the nortriptyline and placebo groups (χ2=4.10, df=1, p=0.04).
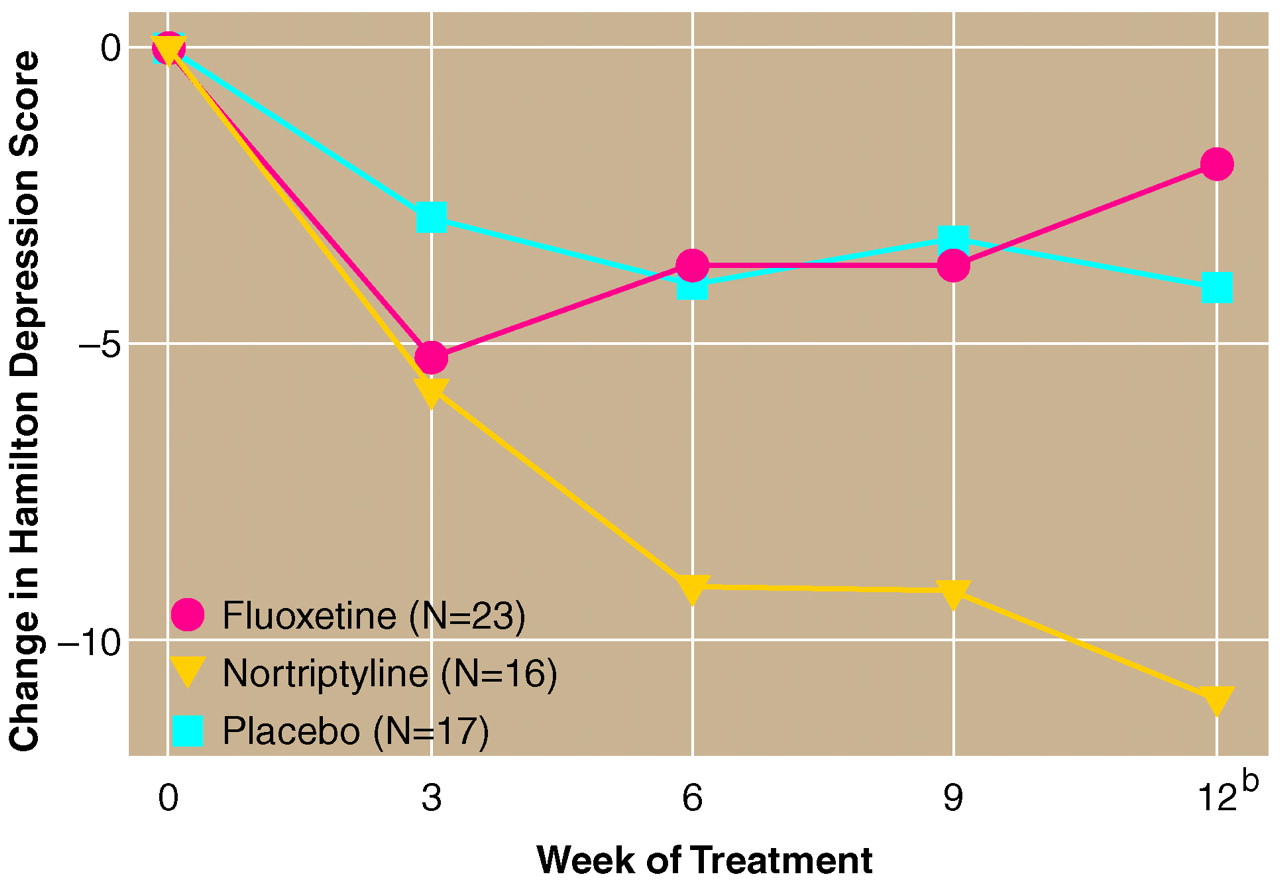
A repeated measures analysis of variance comparing Hamilton depression scale scores for all depressed patients entered in the fluoxetine, nortriptyline, and placebo groups demonstrated a significant time-by-treatment interaction (F=3.45, df=8, 212, p=0.004). A post-hoc analysis of group mean depression scores using the Duncan statistic indicated no intergroup difference at the initial evaluation between the nortriptyline and fluoxetine groups, although the placebo group had a significantly lower mean score than the nortriptyline group (p<0.05). At 12 weeks, the nortriptyline group had a significantly lower mean Hamilton depression scale score than the fluoxetine group, and the placebo group also had a significantly lower mean score than the fluoxetine group (Duncan statistic, p<0.05). The nonparametric analysis of trajectories also revealed a significant difference between the groups (Kruskal-Wallis χ2=8.00, df=2, p<0.02). Follow-up tests with Tukey’s honestly significant difference criterion indicated that the nortriptyline group had more consistent trajectory for improvement on Hamilton depression scale scores than the fluoxetine group.
Because placebo-treated patients had lower Hamilton depression scale scores than the nortriptyline-treated patients at the initial evaluation, we repeated the analyses covarying for baseline scores. This analysis demonstrated the same time-by-treatment interaction (F=3.24, df=6, 156, p=0.01). Post-hoc analysis showed that patients treated with nortriptyline had a significantly greater decline in Hamilton depression scale scores than either placebo- or fluoxetine-treated patients at 12 weeks (
figure 1). There were no differences between fluoxetine and placebo. The successful treatment rate was 10 of 16 (63%) for nortriptyline, two of 23 (9%) for fluoxetine, and four of 17 (24%) for placebo.
Efficacy Analysis
A repeated measures analysis of variance of mean Hamilton depression scale scores comparing the depressed patients in the three treatment groups who completed the 12-week study (14 patients in the fluoxetine group, 13 in the nortriptyline group, and 13 in the placebo group) demonstrated a significant time-by-treatment interaction (F=3.65, df=8, 148, p=0.001). Post-hoc analysis demonstrated no significant intergroup differences during the initial evaluation. The nortriptyline group, however, had a significantly lower mean Hamilton depression scale score than the fluoxetine group at 12 weeks (Duncan statistic, p<0.05). The placebo group also had a significantly lower mean score than the fluoxetine group at 12 weeks (Duncan statistic, p<0.05). Nonparametric analysis of the trajectories provided similar results. There were overall differences in trajectories among the three treatment groups (Kruskal-Wallis χ2=8.07, df=2, p<0.02). Follow-up tests with Tukey’s honestly significant difference criterion indicated that nortriptyline was associated with more consistently improving trajectories compared with either the fluoxetine or placebo groups. To account for the lower scores of the placebo group at the initial evaluation, repeated measures analysis of variance covarying for baseline scores was done. The analysis revealed a significant time-by-treatment interaction (F=3.58, df=6, 108, p=0.005). Although there were no differences between fluoxetine and placebo at any time point, nortriptyline was superior to placebo and fluoxetine at 12 weeks of treatment and to placebo at 9 weeks of treatment (Duncan statistic, p<0.05).
The successful treatment rate was 10 of 13 (77%) for nortriptyline, two of 14 (14%) for fluoxetine, and four of 13 (31%) for placebo. The overall rate of response was significantly different than a random distribution (χ2=11.70, df=2, p=0.003). The response rate was significantly higher in the nortriptyline group than in either the fluoxetine (Fisher’s exact test, p=0.002) or placebo groups (Fisher’s exact test, p=0.05), and the response rates in the fluoxetine and placebo groups were not significantly different from each other.
Treatment Effects on Anxiety and Impairment Scores
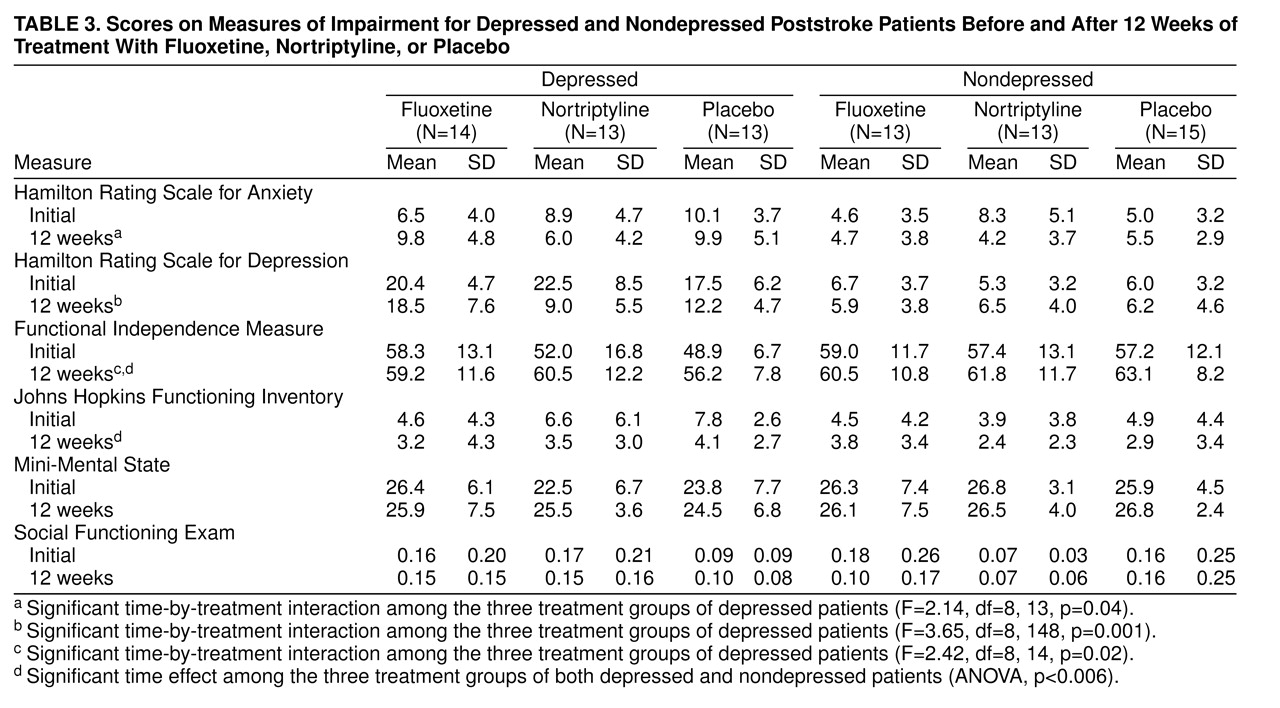
Scores for the Hamilton depression and anxiety scales as well as the impairment scales before and after treatment for the fluoxetine, nortriptyline, and placebo groups are shown in
table 3. Among the depressed patients, repeated measures analysis of variance comparing the nortriptyline, fluoxetine, and placebo groups on the Hamilton anxiety scale revealed a significant time-by-treatment interaction (F=2.14, df=8, 13, p=0.04). Although no significant post-hoc differences between groups were found at any time point, the nortriptyline group had a mean 2.9-point decline in the Hamilton anxiety scale score, the fluoxetine group had a mean 3.2-point increase in the Hamilton anxiety scale score, and the placebo group’s mean score did not change.
Analysis of scores on the impairment scales for depressed patients revealed a significant time-by-treatment interaction for the Functional Independence Measure (F=2.42, df=8, 14, p=0.02). Post-hoc analysis covarying for baseline scores revealed that improvement in Functional Independence Measure scores was significantly greater in the nortriptyline group than in the fluoxetine group at 9 and 12 weeks (Duncan statistic, p<0.05) and in the placebo group compared with the fluoxetine group at 12 weeks (
table 3). Thus, the fluoxetine group did not show the same amount of recovery in activities of daily living as the nortriptyline or placebo groups.
Comparison of the depressed fluoxetine, nortriptyline, and placebo groups on other impairment measures demonstrated a significant time effect for results on the Johns Hopkins Functioning Inventory (F=11.80, df=4, 148, p=0.0001) but no time-by-treatment interaction. There were no significant treatment or time effects for results on the Mini-Mental State or the Social Functioning Exam.
Potentially Confounding Variables
Because five of the nortriptyline-treated patients, three of the fluoxetine-treated patients, and one placebo-treated patient in the depressed group were treated in Argentina, we reanalyzed the results after removing the data for these patients. Using only the Iowa patients, analysis of Hamilton depression scale scores in either the intention-to-treat or the efficacy analysis showed a significant treatment-by-time interaction (F=2.22, df=8, 176, p=0.028, for the intention-to-treat analysis; F=2.72, df=8, 112, p=0.009, for the efficacy analysis). Assessment of differences at each time point demonstrated for both analyses that the nortriptyline-treated group had significantly lower scores than the fluoxetine-treated group at 12 weeks (Duncan statistic, p<0.05). There were also no significant effects of removing these patients on the treatment outcome for Hamilton anxiety scale scores, Functional Independence Measure scores, Johns Hopkins Functioning Inventory scores, Mini-Mental State scores, and Social Functioning Exam scores.
Five of the fluoxetine-treated patients who completed the study and one who dropped out were given active treatment during the second 3-month period, but only one of the nortriptyline-treated patients received active treatment during that period. To control for this potentially confounding variable, we repeated the intention-to-treat and efficacy analyses using only first 3-month or only second 3-month data for the fluoxetine group. Significant treatment-by-time interactions were found for both the first 3-month data and the second 3-month data (F=3.90, df=8, 128, p=0.004, first 3 months; F=2.57, df=8, 112, p=0.01, second 3 months). In both analyses, nortriptyline-treated patients had a significantly lower Hamilton depression scale score than fluoxetine-treated patients at 9 weeks and 12 weeks for the first 3 months and at 12 weeks for the second 3 months (Duncan statistic, p<0.05).
Because the fluoxetine-treated patients showed an increase in Hamilton depression scale scores between week 3 and week 12, we analyzed weight change and dose of fluoxetine in these patients to determine whether gastrointestinal side effects or doses over 20 mg of fluoxetine might explain the increased scores. To eliminate the effect of depression on weight, we analyzed weight change in nondepressed patients. Of the seven nondepressed treated with fluoxetine for whom we had data, six had lost more than 10 lb during the 3 months of treatment (mean=14.0 lb, SD=5.65), whereas none of the eight nortriptyline-treated patients (mean gain=7.6 lb, SD=16.49) or the six placebo-treated patients (mean gain=5.0 lb, SD=4.62) had lost weight (Fisher’s exact test, p=0.002). When depressed and nondepressed patients were included in the analysis of weight change, 10 of 12 fluoxetine-treated patients had lost 10 lb or more (mean=15.1 lb, SD=8.07 lb, or 8% of initial body weight), whereas only two of 13 nortriptyline-treated patients and one of 11 placebo-treated patients had lost 10 lb or more (Fisher’s exact test, p=0.0004). These results indicate that during the initial 12 weeks of treatment, fluoxetine induced a significant weight loss in the majority of these elderly stroke patients. There was, however, no significant correlation between the amount of weight loss and change in Hamilton depression scale scores among the depressed or nondepressed patients.
Furthermore, we examined pulse and blood pressure before treatment and at the 12-week follow-up. The mean pretreatment vital signs for the depressed nortriptyline, fluoxetine, and placebo groups were pulse 78 bpm, blood pressure 126/74 mm Hg; 72 bpm, 110/80 mm Hg; and 79 bpm, 133/74 mm Hg, respectively. After 12 weeks of treatment the vital signs were 87 bpm, 134/80 mm Hg; 73 bpm, 132/78 mm Hg; and 74 bpm, 133/73 mm Hg, respectively. There was a significant change in pulse (F=4.94, df=2, 26, p=0.02), and the increase in the nortriptyline group was significantly greater than that in the placebo group (Duncan statistic, p<0.05) but not significantly different than the pulse change in the fluoxetine group. There were no significant intergroup differences in pre- to posttreatment blood pressure. Among the nondepressed patients, we found a similar mean increase of four beats in pulse in both the nortriptyline and fluoxetine groups and an increase of 11 beats in the placebo group (F=4.21, df=2, 27, p=0.01). There were no changes in blood pressure among the nondepressed patients.
We also reanalyzed our data for the first 6 weeks of treatment when all patients were receiving a maximum dose of 20 mg/day of fluoxetine. Repeated measures analyses covarying for baseline scores in the intention-to-treat analysis revealed a significant time-by-treatment interaction (F=3.47, df=2, 53, p=0.04); the nortriptyline group had a significantly greater decrease in mean Hamilton depression scale score than either the fluoxetine or placebo groups at 6 weeks (Duncan statistic, p<0.05). Thus, the 40-mg dose of fluoxetine was not responsible for the failure of the SSRI to produce the same response to treatment as nortriptyline.
We next examined results by controlling for the increased number of male patients in the fluoxetine group. All groups were balanced for comparable male-female ratios. Intention-to-treat and efficacy analyses demonstrated a time-by-treatment interaction (F=2.59, df=8, 160, p=0.01), with the nortriptyline-treated group having significantly lower Hamilton depression scale scores than the fluoxetine- and placebo-treated groups at 12 weeks of treatment (Duncan statistic, p<0.05).