The prototype atypical drug clozapine is effective in some patients who do not respond to classic antipsychotics
(9). In addition, the incidence of extrapyramidal side effects with clozapine is very low
(10), and treatment with clozapine may have an effect on negative symptoms
(11–
13). Interestingly, the striatal D
2 receptor occupancy during clozapine treatment is significantly lower (20%–67%) than that during treatment with classic antipsychotics (70%–90%)
(14–
16). This low D
2 occupancy supports the view that clozapine acts by a different mechanism from that of classic antipsychotic drugs.
Despite the low striatal D
2 receptor occupancy, it has been suggested that the dopamine system is essential for the mechanism of action of clozapine. In addition to occupancy of the D
2 receptors, clozapine treatment induces high occupancy of the serotonin 2A (5-HT
2A) receptors. It has been suggested that the high 5-HT
2A/D
2 ratio underlies the atypical properties of clozapine
(11). An early hypothesis was that clozapine has preferential effects in the limbic and cortical dopaminergic systems
(17–
21). However, the low resolution of older PET systems and the lack of suitable high-affinity radioligands have not allowed detection of the low D
2 densities in extrastriatal regions.
We have developed [
11C]FLB 457, a substituted benzamide with the very high affinity of 20 pM for D
2 and D
3 dopamine receptors in vitro
(22,
23). In a preliminary study using an old PET system
(24,
25), we found that the extrastriatal binding of [
11C]FLB 457 is reduced by treatment with haloperidol and clozapine. The D
2 occupancy was at the same level in the thalamus and the temporal cortex as that determined with [
11C]raclopride in the striatum. Opposite results were demonstrated by Pilowski and co-workers
(26) using single photon emission tomography (SPET) and [
123I]epidepride in seven clozapine-treated patients. Pilowski et al. reported that the D
2 receptor occupancy was low in the striatum but high in the temporal cortex. The results were taken as support for the hypothesis of limbic selectivity of clozapine.
Results
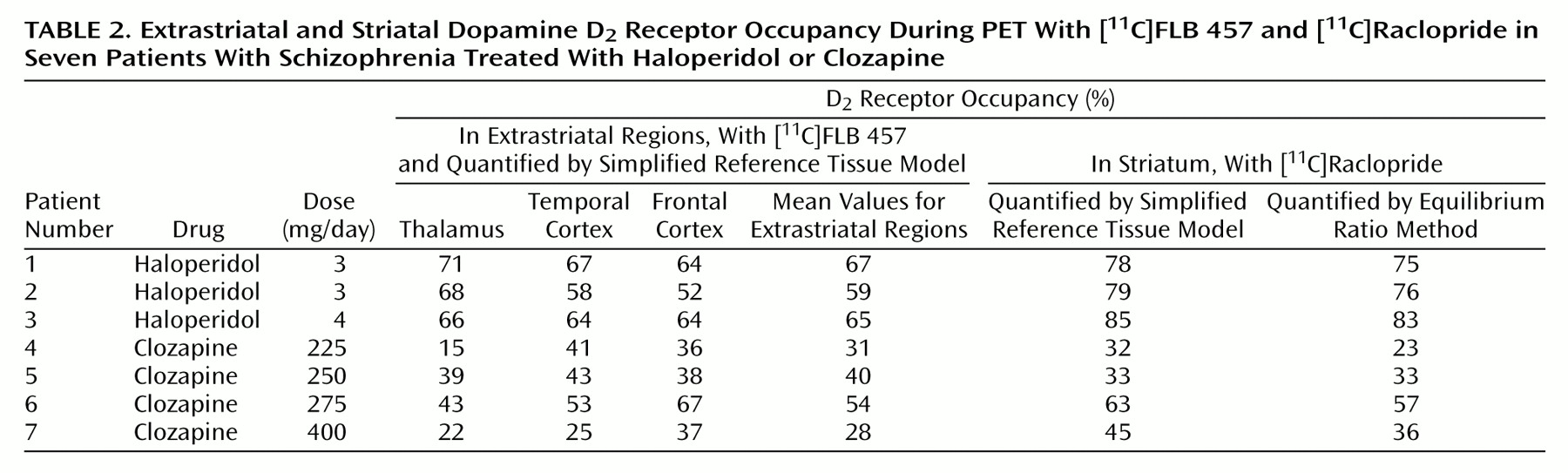
Using the simplified reference tissue model, we found that the D
2 receptor occupancy in the striatum of the haloperidol-treated patients was between 78% and 85% (
Table 2). In the striatum of the four clozapine-treated patients the D
2 receptor occupancy was lower, ranging from 32% to 63% (t=4.29, df=5, p=0.008). The D
2 receptor occupancy values determined with the simplified reference tissue model and the ratio method were very similar (
Table 2). The coefficient of correspondence between methods was 0.98 (a very high agreement).
In the haloperidol-treated patients, the D
2 receptor occupancy in the thalamus was 66%–71% (
Table 2). The thalamic D
2 receptor occupancy was lower in clozapine-treated patients, ranging from 15% to 43% (t=4.81, df=5, p=0.005). The D
2 receptor occupancy in the temporal cortex was 58%–67% in the haloperidol group and 25%–53% in the clozapine group (t=3.12, df=5, p=0.003). In the frontal cortex the range of the D
2 receptor occupancy values was 52%–64% for the haloperidol-treated patients and between 36% and 67% for the clozapine treated patients (t=1.53, df=5, p=0.17).
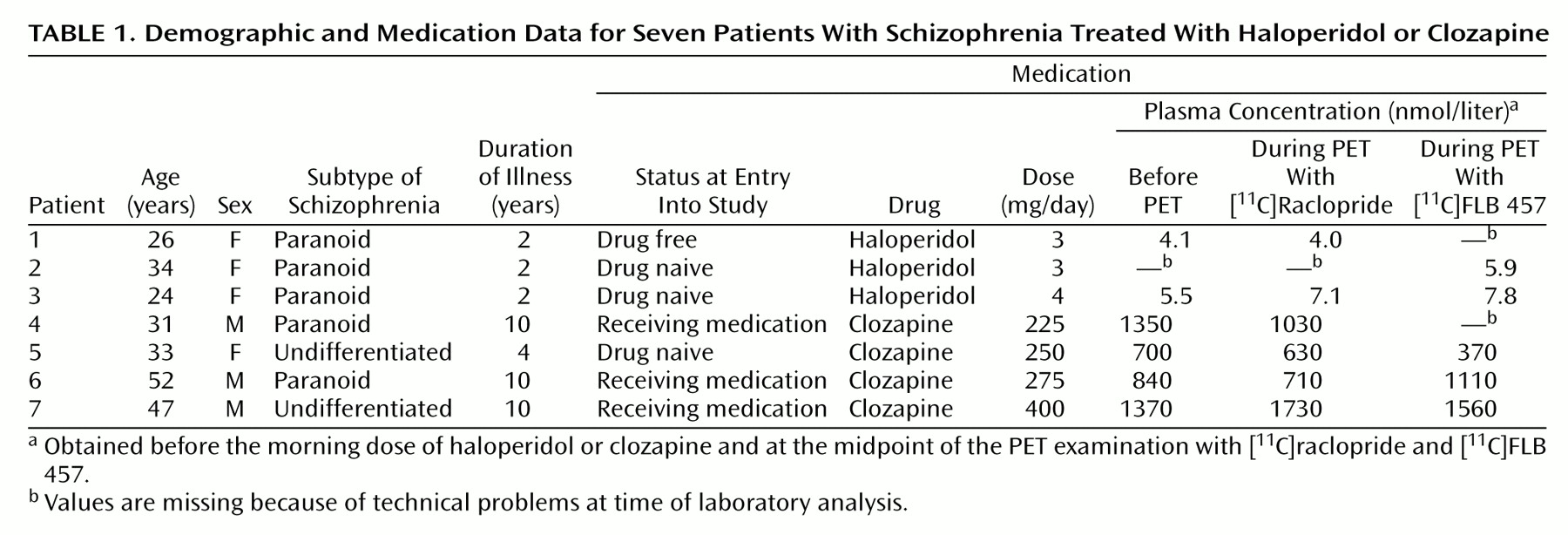
The plasma concentrations of haloperidol and clozapine for all patients were in accordance with reference values from previous studies
(32,
35,
36) (
Table 1).
Discussion
Previous PET studies have consistently demonstrated high D
2 receptor occupancy in the striatum during treatment with classic antipsychotics
(4,
7,
8). In patients treated with the atypical antipsychotic drug clozapine, however, a significantly lower striatal D
2 receptor occupancy has been reported
(15,
16). In the present study, which used a PET system with high resolution and three-dimensional data acquisition, the striatal D
2 receptor occupancy was higher in patients treated with haloperidol (78%–85%) than in patients treated with clozapine (32%–63% ). The results for [
11C]raclopride binding are thus consistent with earlier studies
(15,
16).
The D
2 receptor occupancy in the extrastriatal regions for the haloperidol-treated patients was approximately at the same level as in the striatum, which is consistent with previous results
(25).
Limbic selectivity has been proposed as a possible mechanism of action of clozapine, the prototype atypical antipsychotic drug. This hypothesis, first formulated in the 1970s, was originally based on animal data and postulated that there is a limbic selectivity in the functional effects of clozapine
(17,
18). A receptor ligand study of both rat and human brain tissue
(19) suggested that clozapine binds preferentially to D
2 receptors in extrastriatal regions. This view was further supported by the finding that clozapine has a different affinity for distinct isoforms of the D
2 receptor
(37). In the present study, PET was used to test the hypothesis of preferential binding of clozapine to extrastriatal D
2 receptors. We found that the mean D
2 receptor occupancy in clozapine-treated patients ranged from 28% to 54% in the three extrastriatal regions (
Table 2). The corresponding values for the striatum in this group ranged from 32% to 63% (t=0.97, df=3, p=0.41). Preferential extrastriatal D
2 receptor occupancy induced by clozapine treatment was thus not supported by our data.
Determination of extrastriatal D
2 receptor occupancy was made with the radioligand [
11C]FLB 457. The high affinity of [
11C]FLB 457 enables quantification of D
2 receptor binding in extrastriatal regions with low density of D
2 receptors
(22). In previous studies, striatal D
2 receptor occupancy has routinely been quantified by using the ratio equilibrium method, which requires a peak equilibrium model in radioligand binding during the time of the PET examination
(24). The time required for equilibrium is dependent on the density of the receptors
(23,
25). In the striatum, a region with very high density of D
2 receptors, equilibrium with [
11C]FLB 457 will not be reached until several hours after the 63-minute data acquisition time used in this study
(38). As previously shown, the short acquisition time for [
11C]FLB 457 binding in the striatum is thus not sufficient for reliable calculations of the binding potential according to the peak equilibrium or the simplified reference tissue model
(23). Therefore, [
11C]FLB 457 binding in the striatum was not included in the present analysis.
For calculation of the D
2 receptor occupancy in the extrastriatal regions, we used the simplified reference tissue model developed by Lammertsma and Hume
(33), who described it as stable for calculation of small receptor quantities. To allow comparison with previous [
11C]raclopride data in the striatum, we used not only the simplified reference tissue model but also the well established equilibrium ratio method
(6). There was a significant correspondence in the results of the two methods, suggesting that the methods are comparable.
For calculation of receptor occupancy during drug treatment, reference values obtained in a drug-free state are required. The optimal reference value is that obtained from a recent examination of the same patient before initiation of drug treatment. However, since clozapine is used mainly for patients whose illness is resistant to classic neuroleptics, it may not be possible to perform PET examinations in a drug-free state for ethical reasons. Accordingly, in the clozapine group, where three of the four patients were receiving medication when they entered the study, a mean value obtained in healthy comparison subjects was used as a reference value. The error introduced by this procedure is within a few percent and has been discussed in detail elsewhere
(15).
In a study by Pilowski et al.
(26), who used SPET and the high-affinity radioligand [
123I]epidepride, the D
2 receptor occupancy was estimated in seven patients treated with clozapine and five patients treated with typical antipsychotic drugs. Pilowski et al. reported that in all clozapine-treated patients the D
2 receptor occupancy was low in the striatum but high in the temporal cortex. In the patients treated with typical antipsychotics, the D
2 receptor occupancy was high both in the temporal cortex and the striatum. The results were taken as a support for the hypothesis of limbic selectivity as the mechanism of action of clozapine.
In contradiction to this SPET study, the present findings do not support preferential high occupancy in the extrastriatal regions. There are several methodological differences between these studies that may contribute to the discrepant results. One important issue is the different imaging systems, PET versus SPET. Another issue concerns the choice of radioligand and the method used for quantification of receptor occupancy. A simple ratio approach using a high-affinity radioligand such as [123I]epidepride without validation of equilibrium conditions may yield an underestimation of the D2 receptor occupancy in the high-density striatum in comparison with the D2 receptor occupancy in the low-density extrastriatal regions.
In conclusion, this study confirms that classic as well as atypical antipsychotics induce D2 receptor occupancy in extrastriatal regions. However, the hypothesis of preferential extrastriatal D2 receptor occupancy as the mechanism of action of clozapine was not supported by our data.
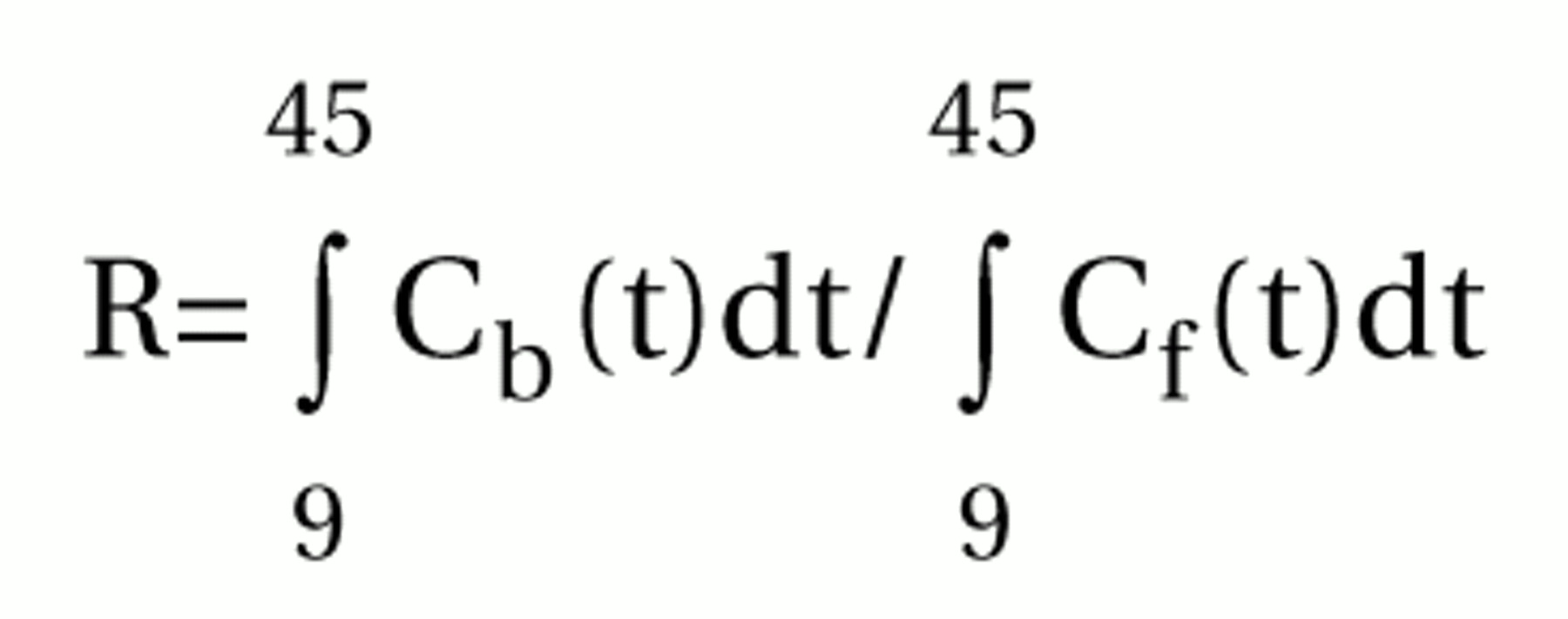 :
: