Results of a cerebral microdialysis study in rats
(1) and a positron emission tomography (PET) study of healthy volunteers
(2) indicated that acute and chronic alcohol intake stimulates dopamine release in the ventral and dorsal striatum. Chronic alcohol intake, however, reduced the availability of striatal dopamine D
2/3 receptors
(3,
4), which may represent a compensatory down-regulation that ensures homeostasis of central dopaminergic neurotransmission
(5). Precipitous withdrawal from alcohol during detoxification results in a rapid decrease in dopamine release
(6), whereas the availability and sensitivity of central dopamine D
2/3 receptors increase during the first week of abstinence
(4,
7,
8). In alcoholic subjects, delayed recovery of central dopamine D
2 receptors was associated with an increased risk of relapse
(7,
8) and may be associated with persistent presynaptic dopaminergic dysfunction during early abstinence
(5,
9). However, striatal dopamine transporter availability was reduced during acute detoxification but did not differ from control levels after several weeks of abstinence
(3,
10). To our knowledge, only one study has examined presynaptic dopamine production in detoxified alcoholic subjects, in whom changes in the net uptake of the dopa decarboxylase substrate [
18F]fluoro-
l-dopa ([
18F]DOPA) were reported in restricted subregions of the striatum
(11).
We recently reported reduced availability of dopamine D
2/3 receptors in the ventral striatum and adjacent putamen of abstinent alcoholic subjects, which was associated with a high level of craving for alcohol and an increase in brain activation elicited by alcohol-associated (as opposed to control) cues
(12). To explore further the interaction between presynaptic striatal dopamine production and dopamine D
2/3 receptor availability in recently detoxified alcoholic patients, we measured [
18F]DOPA uptake in the same alcoholic patients and comparison subjects, who also underwent assessment of dopamine D
2/3 receptors with [
18F]desmethoxyfallypride ([
18F]DMFP). We tested the following hypotheses: 1) [
18F]DOPA influx to the striatum is lower in alcoholic subjects, relative to healthy comparison subjects; 2) this lower level of influx correlates with higher levels of craving for alcohol; and 3) alcohol craving is associated with a risk of relapse during a 6-month follow-up period. Lower levels of [
18F]DOPA influx, a marker of impaired presynaptic dopamine production, may predict alcohol craving if it coincides spatially with a low availability of dopamine D
2/3 receptors. Because low striatal dopamine D
2/3 receptor availability was inversely correlated with alcohol craving in abstinent alcoholic subjects
(12), we also explored whether low [
18F]DOPA influx, when normalized to the local availability of dopamine D
2/3 receptors, is associated with alcohol craving.
Results
Contrary to our hypothesis [
18F]DOPA net influx did not differ between the alcoholic patients and the healthy comparison subjects (p>0.05, corrected for the striatal voxel of interest). However, as hypothesized, the severity of alcohol craving measured with the Alcohol Craving Questionnaire was significantly and negatively correlated with [
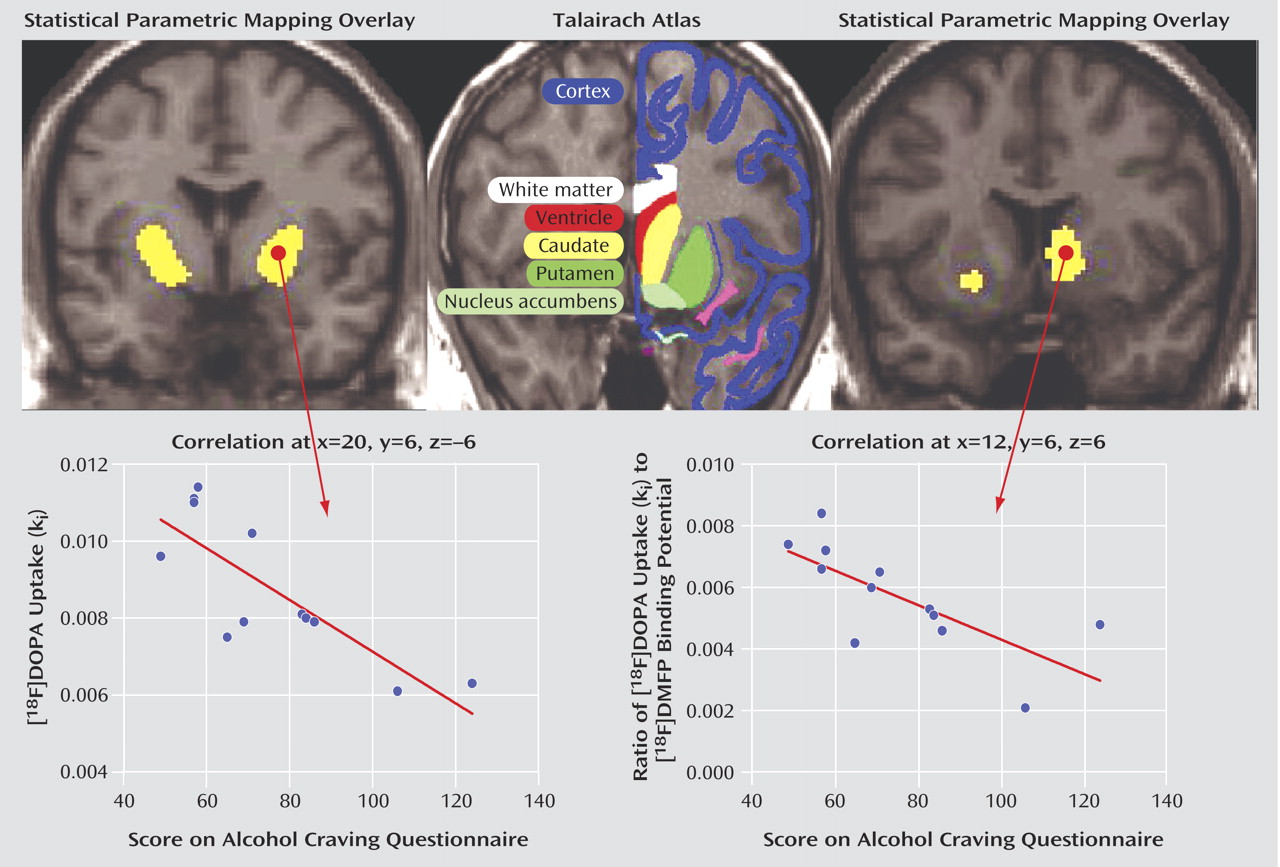
18F]DOPA net influx in the bilateral putamen (right: x=20, y=6, z=–6 [Talairach coordinates]; r=–0.80, N=12) (left: x=–22, y=8, z=–10; r=–0.80, N=12) (p<0.05, corrected for striatal voxel of interest and after Bonferroni’s correction for multiple testing). For illustrative purposes, only the scatter plot of the right side is shown in
Figure 1 (lower left).
Although [18F]DOPA can also label cortical catecholamine fibers, there was no correlation between alcohol craving and the magnitude of net blood-brain clearance in the cerebral cortex. In the healthy comparison subjects, no significant correlation was observed between alcohol craving and the magnitude of net blood-brain clearance. Five alcoholic patients remained abstinent and seven relapsed during the 6-month follow-up period (mean alcohol intake=14.5 kg, SD=22.8). As hypothesized, the severity of alcohol craving measured with the Alcohol Craving Questionnaire was positively and significantly correlated with subsequent alcohol intake during the 6-month observation period (Pearson’s r=0.76, N=7, p<0.05, after Bonferroni’s correction for multiple testing).
As previously reported
(12), dopamine D
2/3 receptor availability was significantly lower in the alcoholic patients than in the healthy comparison subjects in the bilateral putamen and adjacent ventral striatum (right: x=20, y=12, z=–6 [Talairach coordinates]; t=2.39, df=9) (left: x=–28, y=10, z=–8; t=2.21, df=9) (p<0.05, corrected for the striatal voxel of interest). No significant correlation was found between [
18F]DOPA net blood-brain clearance and [
18F]DMFP binding potential. [
18F]DOPA net influx per voxel was normalized to the availability of dopamine D
2/3 receptors in the same voxel (net blood-brain clearance/binding potential ratio). In the alcoholic patients, we observed a negative correlation between the severity of alcohol craving and the magnitude of this ratio throughout the bilateral putamen and the right ventral striatum and caudate (right: x=12, y=6, z=6 [Talairach coordinates of the voxel with maximum significance]; r=–0.73, N=12) (left: x=–22, y=10, z=–8; r=–0.68, N=12) (p<0.05 corrected for striatal voxel of interest). For illustrative purposes, only the scatter plot for the right side is shown in
Figure 1 (lower right). No significant correlation between this ratio and alcohol craving was found in the healthy men.
We did not observe significant correlations between the main outcome variables (severity of alcohol craving, mean [18F]DOPA net influx in the striatum) and potentially confounding variables, such as smoking, age at onset of alcohol dependence, severity of alcoholism, lifetime alcohol intake, and length of time between last alcohol intake and scanning (Pearson’s r range=–0.44 to 0.01, N=12, all p>0.15).
Discussion
This study shows that in abstinent alcoholic patients, striatal [
18F]DOPA net influx in the striatum correlated inversely with the severity of alcohol craving as assessed with the Alcohol Craving Questionnaire
(17). Alcohol is known to stimulate dopamine release in the striatum in humans and experimental animals
(1,
3,
5,
29), and alcohol consumption may be specifically rewarding in subjects with a striatal dopamine deficit. After detoxification, synaptic dopamine release was found to decrease rapidly in microdialysis studies of experimental animals
(6). Thus, we hypothesized that alcoholic patients with a persistent striatal dopamine deficit may experience stronger cravings for alcohol. In support of this hypothesis, the results of our study suggest that low capacity for dopamine production in nigrostriatal fibers predicts severity of alcohol craving in detoxified alcoholic patients. However, contrary to our hypothesis, we did not observe a significant group difference in [
18F]DOPA net influx between detoxified alcoholic patients and healthy comparison subjects. This observation indicates that dopamine synthesis capacity per se may not be affected in detoxified alcoholic patients. Rather, a low level of dopamine synthesis may contribute to alcohol craving if it coincides with other factors, such as a local reduction in the availability of dopamine D
2/3 receptors.
In a previous study of striatal dopamine D
2/3 receptor availability in the same group of alcoholic patients, we observed a significant negative correlation between alcohol craving and the availability of dopamine D
2/3 receptors, specifically in the ventral striatum
(12). Therefore, in the present study we used SPM analysis to search for regions in which the [
18F]DOPA net influx, relative to the local availability of dopamine D
2/3 receptors, correlated with the severity of alcohol craving in abstinent alcoholic patients. In accordance with the prediction that the relationship between dopamine synthesis capacity and D
2/3 receptor availability is altered in alcoholic patients, we observed a negative correlation between alcohol craving and the magnitude of the net blood-brain clearance/binding potential ratio, indicating that the alcoholic patients who exhibited the most severe craving had a low capacity for dopamine synthesis, relative to the number of available D
2/3 receptors in the putamen and adjacent ventral striatum.
However, in the context of the receptor competition binding model
(30), low availability of binding sites for benzamide radioligands could indicate high basal occupancy by dopamine. Indeed, the availability of D
2/3 binding sites in this group of detoxified alcoholic patients was lower than in age-matched healthy men. Given the present finding of reduced dopamine synthesis capacity in alcoholic patients, we suggest that the lower availability of dopamine receptors cannot readily be attributed to increased occupancy by dopamine. However, the effects of occupancy on the magnitude of binding potential cannot be ascertained from single PET studies. Although the ventral striatum/nucleus accumbens is most strongly linked to the rewarding properties of drugs
(5,
9,
29), we found the highest correlation between the net blood-brain clearance/binding potential ratio and alcohol craving in the dorsal striatum. Although this observation must be qualified by the spatial resolution of the tomography, the results of some previous studies suggest that dopamine release in the dorsal striatum is involved in habit formation, which may play a preeminent role in stereotypical drug and alcohol intake and relapse
(31,
32).
The relevance of alcohol craving for the relapse risk of detoxified alcoholic patients remains a topic of debate. Some studies suggested that relapse is triggered by habit formation and automatic drug intake rather than by conscious drug-craving
(32,
33). However, in the present study, conscious alcohol craving, as measured by the Alcohol Craving Questionnaire, was significantly and positively correlated with the subsequent amount of alcohol intake during the 6-month follow-up period, whereas no significant correlations were found between subsequent alcohol intake and the severity of alcoholism
(15), lifetime alcohol intake
(16), or the number of cigarettes smoked per day. This observation supports the hypothesis that alcohol craving is associated with the risk of relapse in detoxified alcoholic patients
(5,
9,
34). Furthermore, we found that low [
18F]DOPA net influx, which was correlated with craving, served as an independent predictor of relapse in the present group of alcoholic patients. In conjunction with our earlier study of [
18F]DMFP binding in the same subjects, we conclude that an insufficiency of dopamine transmission in the striatum underlies the propensity to relapse.
Some potential limitations of the study should be considered. The patients lie in the scanner with their eyes closed, and although they were instructed to relax, some of them may have experienced various degrees of alcohol craving, which might have interacted with the PET measures. Moreover, the observed correlations between [18F]DOPA net influx and craving do not imply causation. The nature of the interaction between striatal dopamine synthesis capacity, D2/3 receptor availability, and alcohol craving might be elucidated in PET studies in which the sensitivity of dopamine receptor availability to pharmacologically evoked dopamine release is compared in abstinent alcoholic patients and healthy comparison subjects.
In conclusion, the results of this study show that alcohol craving is associated with high risk of relapse and a low level of striatal [18F]DOPA net influx, an index of dopamine synthesis capacity. In this context, we speculate that alcohol-induced dopamine release during relapse may compensate for a relative deficit in dopamine neurotransmission. The [18F]DOPA net influx normalized to the local availability of dopamine D2/3 receptors correlated negatively with alcohol craving in abstinent alcoholic patients. Thus, the full spectrum of perturbed dopamine transmission in alcoholism reflects presynaptic changes in dopamine synthesis and postsynaptic changes in receptor availability, assessed by combined [18F]DOPA/[18F]DMFP PET studies. The results of our study support the hypothesis that a deficit in striatal dopamine neurotransmission contributes to alcohol craving.