The Optimization of Treatment and Management of Schizophrenia in Europe (OPTiMiSE) Trial: Rationale for Its Methodology and a Review of the Effectiveness of Switching Antipsychotics
Abstract
Background:
Methods:
Results:
Discussion:
Introduction
Review of Prospective Randomized Studies on Switching the Drug in Initial Nonresponders to Antipsychotics
Method
Results
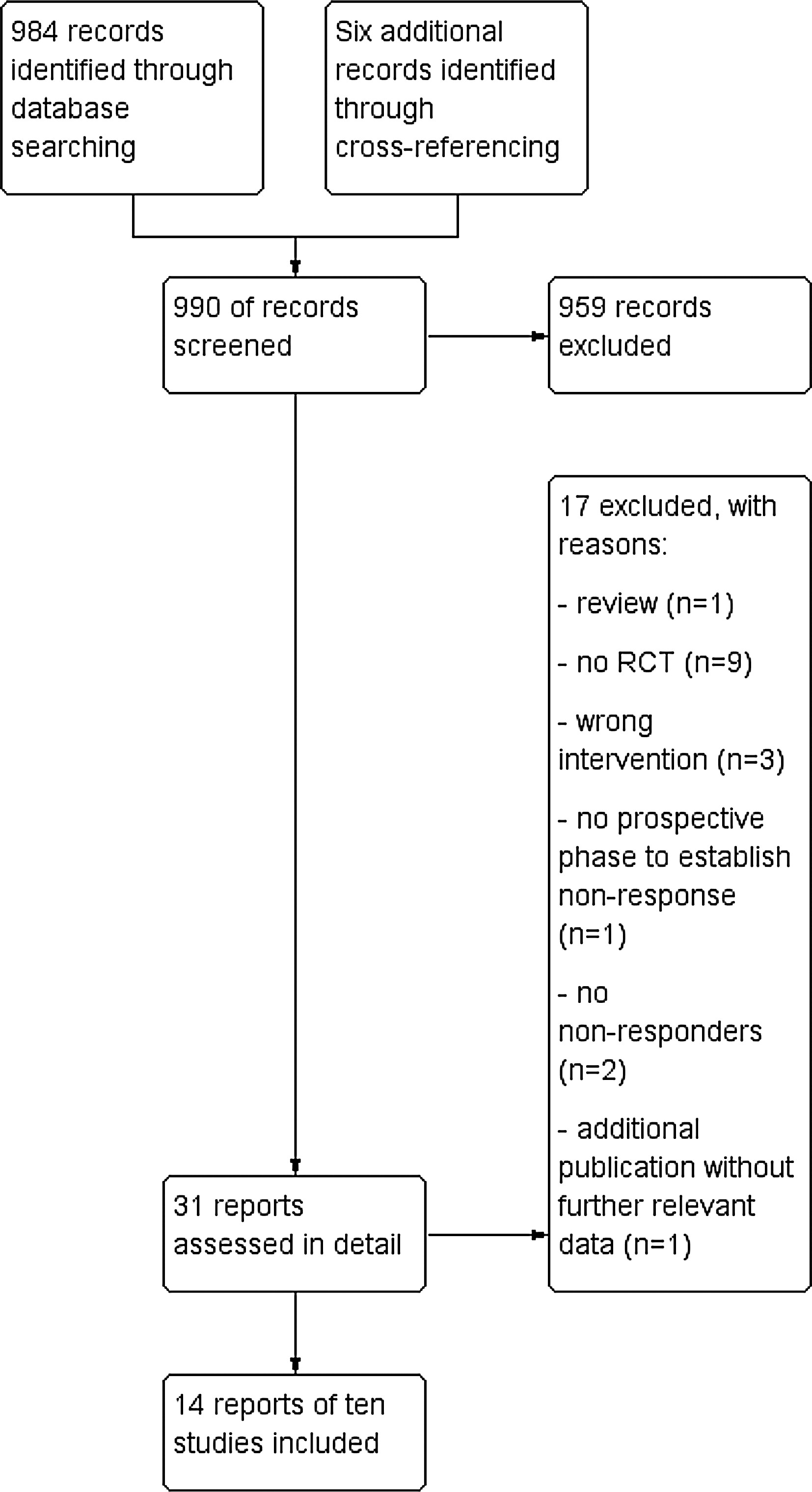
| Author, Country | Diagnosis | “Run-in” phase: drugs (dose), duration, blinding, number of participants | Randomized phasea: drug groups (n), duration, blinding | Main resultb |
|---|---|---|---|---|
| Kinon et al,15 USA | Schizophrenia, schizoaffective, or schizophreniform disorder DSM-III-R | Fluphenazine 20 mg/d, 4 wk, open, n = 156 | Switch to haloperidol 20 mg/d (n = 13), increase fluphenazine dose to 80 mg/d (n = 16) or stay on fluphenazine 20 mg/d (n = 18), 4 wk, db | No significant efficacy difference between groups. Only 9% responded in the randomized phase |
| Klimke et al,19 Germany | Acute schizophrenia, ICD-9 | Haloperidol, 15 mg/d, 3 days, open, n = 50 | Switch to perazine 300 mg/d (n = 12) or stay on haloperidol 15 mg/d (n = 13), 3 wk, db | No significant difference between groups |
| Shalev et al,20 Israel | Schizophrenia, chronic, or subchronic with acute exacerbation DSM-III | Haloperidol (target dose 20 mg/d, perphenazine (target 32 mg/d), levomepromazine (target 300 mg/d), open, n = 75 | Second phase: Switch to one of the remaining antipsychotics (n = 20), 4 wk, open third phase: Switch to the remaining drug (n = 9), 4 wk, open | No significant difference between groups. Overall improvement rate 95% |
| Suzuki et al,21 Japan | Schizophrenia, DSM-IV | Olanzapinec (flexible dose), quetiapine (flexible dose)c, risperidone (flexible dose)c, max. 8 wk, open, n = 78 | Second phase: Switch to one of the remaining antipsychotics (n = 37), max. 8 wk, open. Third phase: Switch to the remaining drug (n = 19), max. 8 wk, open | No significant differences between drugs in the primary outcome. Quetiapine less effective than olanzapine and risperidone in secondary outcomes |
| Hatta et al,22 Japan | Schizophrenia, schizophreniform, or schizoaffective disorder, DSM-IV | Risperidone (max. dose 6 mg/d), 2 wk, open, n = 73 | Olanzapine (max. 20 mg/d) or risperidone (max. 6 mg/d), n = 20, 2 wk, sb | No significant difference between switchers and stayers. Early responders improved more than early nonresponders |
| Hatta et al,22 Japan | Schizophrenia, schizophreniform, or schizoaffective disorder, DSM-IV | Olanzapine (max. dose 20 mg/d), 2 wk, sb, n = 58 | Risperidone (max. 6 mg/d), olanzapine (max. 20 mg/d) or, n = 20, 2 wk, sb | No significant difference between switchers and stayers. Early responders improved more than early nonresponders |
| Hatta et al,23 Japan | Schizophrenia, schizophreniform, or schizoaffective disorder, DSM-IV | Risperidone (starting dose 3 mg/d, n = 74) or olanzapine (starting dose 10 mg/d, n = 86) chosen at the physicians’ discretion, 2 wk, sb | Add the other drug or switch to the respective other drug, same doses, 10 wk, n = 51, sb | No significant difference between combining drugs and switching to the other drug |
| McEvoy et al,25 USA | Schizophrenia DSM-IV | Olanzapine (7.5–30 mg/d), perphenazine (8–32 mg/d), quetiapine (200–800 mg/d), risperidone (1.5–6 mg/d), ziprasidone (80–160 mg/d), max. 18 mo, db, n = 1493 | Participants who discontinued phase 1d were randomized to clozapine, olanzapine, quetiapine or risperidone (patients could not receive the same drug as in phase I), n = 99, max. 18 mo, db except clozapine | Time to discontinuation was significantly longer for clozapine than for quetiapine and risperidone, but not longer than for olanzapine |
| Stroup et al,26 USA | Schizophrenia DSM-IV | Olanzapine (7.5–30 mg/d), perphenazine (8-32 mg/d), quetiapine (200-800 mg/d), risperidone (1.5-6 mg/d), ziprasidone (80–160 mg/d), max. 18 mo, db, n = 1493 | Participants who discontinued phase Id were randomized to ziprasidone, olanzapine, quetiapine, or risperidone (patients could not receive the same drug as in phase I), n = 444, max. 18 mo, db | Time to discontinuation was significantly longer for olanzapine and risperidone than for quetiapine and ziprasidone |
| Kinon et al,30 USA | Schizophrenia, schizophreniform, or schizoaffective disorder, DSM-IV | Risperidone (2–6 mg/d), 2 wk, open, n = 628 | Switch to olanzapine 10–20 mg/d or staying on risperidone, n = 378, 10 wk, db | Switchers to olanzapine had a statistically significantly larger symptom reduction than stayers on risperidone. |
| Leucht et al31 ongoing Germany, Romania | Schizophrenia or schizoaffective disorder DSM-IV | Amisulpride (200–800 mg/d) or olanzapine (5–20 mg/d), 2 wk, randomized, db, n = 350 | Switching to the respective other drug or staying on the same drug, same doses, 6 wk, n = not indicated, db | Ongoing study |
OPTiMiSE Trial Design
Study Flow
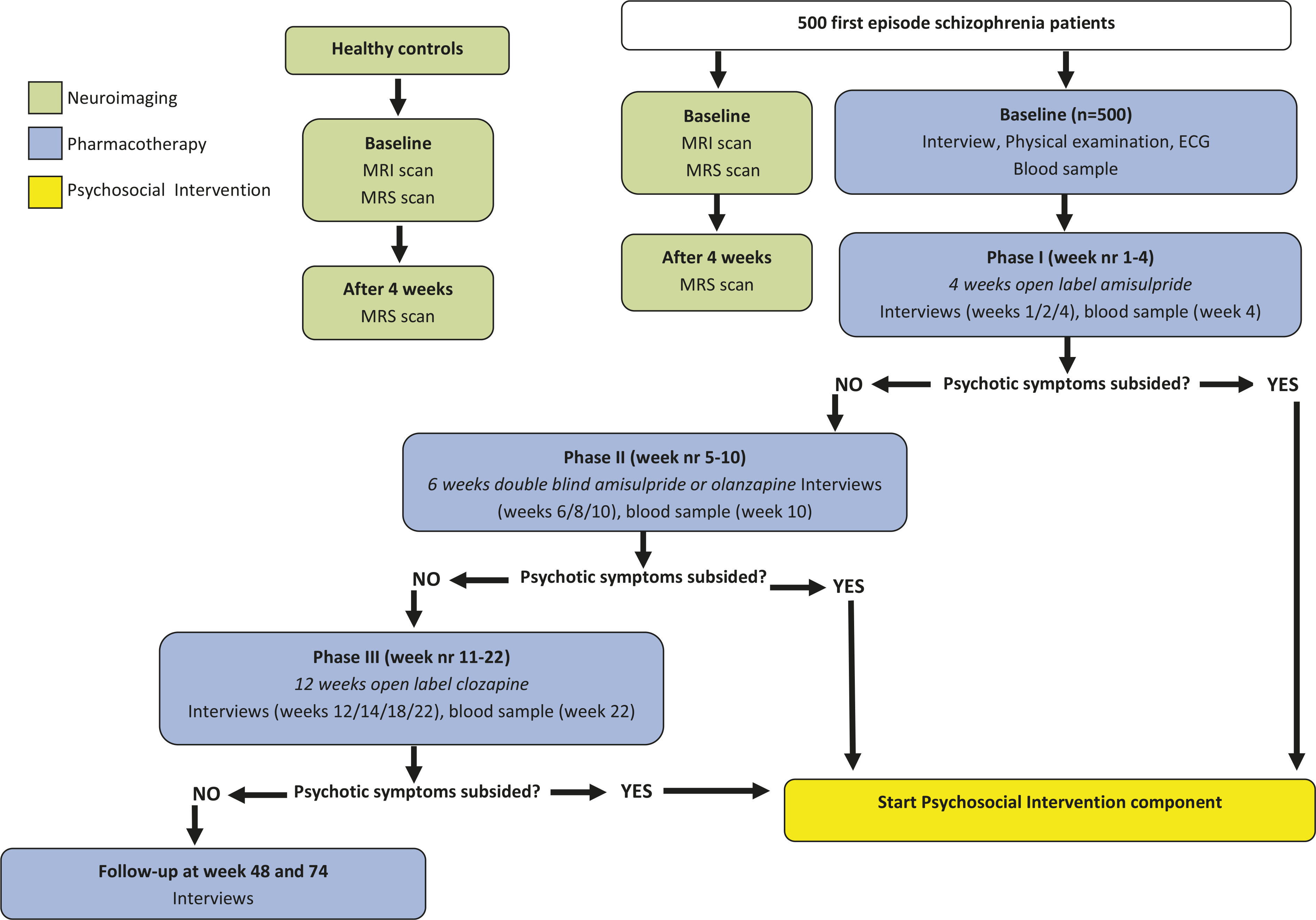
Participants
Rationale for Study Medication
Phase I.
Phase II.
Phase III.
Choice of Outcomes
Statistical Analysis
Study Progress and Outlook
References
Information & Authors
Information
Published In
History
Authors
Metrics & Citations
Metrics
Citations
Export Citations
If you have the appropriate software installed, you can download article citation data to the citation manager of your choice. Simply select your manager software from the list below and click Download.
For more information or tips please see 'Downloading to a citation manager' in the Help menu.
View Options
View options
PDF/EPUB
View PDF/EPUBGet Access
Login options
Already a subscriber? Access your subscription through your login credentials or your institution for full access to this article.
Personal login Institutional Login Open Athens loginNot a subscriber?
PsychiatryOnline subscription options offer access to the DSM-5-TR® library, books, journals, CME, and patient resources. This all-in-one virtual library provides psychiatrists and mental health professionals with key resources for diagnosis, treatment, research, and professional development.
Need more help? PsychiatryOnline Customer Service may be reached by emailing [email protected] or by calling 800-368-5777 (in the U.S.) or 703-907-7322 (outside the U.S.).

