Treatment-resistant schizophrenia is a familiar concept to today’s clinicians, and closely linked to clozapine. This is not coincidental, as seminal work involving clozapine in the late 1980s established the current framework for defining TRS. Drawing on similar criteria, a substantive body of evidence has evolved since clozapine’s reintroduction, and while modifications have been suggested, persistent symptoms and a prolonged period of poor functioning, despite several AP trials, remain fundamental. Subsequent advances demand that we revisit the concept of TRS to ensure it aligns with current thinking.
Treatment-Resistant Schizophrenia: A Historical Perspective
It is believed that the first report of TRS occurred in the context of insulin resistance.
1 However, it was following the introduction of CPZ and APs in the 1950s that the term gained momentum, and within a decade, therapy refractory was discussed.
2 Established criteria defining TRS were first used in 1966 in a paper
2 addressing therapy-resistant schizophrenia, which included the following: active psychotic symptoms, despite 2 years of AP exposure, including 6 months of phenothiazine treatment at doses greater than CPZ 600 mg or trifluoperazine 80 mg daily.
Advances in understanding related to dopamine’s role in psychosis, specifically dopamine D
2 receptor blockade in AP activity, translated to a focus on high-potency conventional APs. Notably, these drugs were not more effective than their low-potency counterparts, although their improved side effect profile from a cardiovascular perspective propagated the practice of high-dose therapy.
3 The guidelines for evaluation of clozapine in TRS captured this, with CPZ equivalents dosing almost twice that of the 1966 criteria.
2,4Treatment-Resistant Schizophrenia: Operational Criteria
By the late 1970s, evidence was being published regarding the comparison of different treatment approaches, including nonpharmacological, providing further evidence that a smaller subgroup would remain treatment resistant.
5 In the late 1980s, a text was published on this topic,
6 including a chapter by Philip May, underscoring the need for a systematic approach that would consider social adaptation as well as symptom reduction.
7 Indeed, May and Dencker,
8 one of the text’s editors, had been instrumental through the 1980s in bringing together an international group to address treatment refractoriness, a process that resulted in criteria highlighting 4 domains: positive symptoms, negative symptoms, functional deficits, and behavioural excess. They highlighted that these domains could be impacted differentially, and advocated that this framework be superimposed on criteria addressing course of illness and treatment response. Specifically, people must be ill for 2 years to define chronicity; additionally, 3 AP trials were required, each at doses of 1000 mg/day CPZ equivalents or more for 6 weeks or longer at steady state levels. An argument was made for either TDM or a trial of depot APs, as well as continuous psychosocial interventions.
Seven levels of response refractoriness were identified and defined: clinical remission, partial remission, slight resistance, moderate resistance, severe resistance, refractory, and severely refractory.
8 Scales to assess functioning were discussed, but no clear-cut thresholds were defined; rather, authors favoured clinical judgment regarding functional capacity.
Criteria for clozapine in TRS captured the essence of this definition, with slight modifications.
4 They aligned with moderate resistance but permitted 3 trials during 5 years, fitting with their prolongation of the illness’ negative impact on functioning. Criteria also specified trials from 2 chemical classes but omitted requirements related to TDM or depot APs. Subsequent treatment studies have embraced these criteria.
Two other sets of criteria have since been published, both using existing criteria for clozapine as a foundation (
Table 1).
9,10Redefining Treatment Resistance in Schizophrenia
Since clozapine was reintroduced, a new class of atypical APs thought to capture its unique pharmacology was spawned.
11 With several decades of clinical experience and research now available, it is timely to reflect on existing evidence. Although not entirely consistent, current consensus holds that clozapine is clinically superior in TRS
12; however, its effectiveness remains limited, ranging from 30% to 70%.
13 Moreover, early expectations that clozapine and subsequent APs substantially impact the multiple symptom domains characterizing schizophrenia (for example, negative symptoms or cognitive deficits) have been challenged.
14These findings necessitate changes in how treatment resistance is framed, and we recently proposed a new paradigm for classifying schizophrenia based on AP treatment response.
15 We also argued that the scope of APs was becoming blurred inasmuch as claims regarding their efficacy broadened markedly with the advent of the newer atypical agents, only to be tempered over subsequent years.
16 We highlighted the uniqueness of clozapine, which appears true in only one form of the illness, TRS. Finally, we raised concerns that efforts to better understand the biological underpinnings of schizophrenia risk being compromised, and the development of new treatments delayed, if these clearly identifiable clinical differences are not acknowledged in research strategies.
The model we propose revolves around positive symptoms, and the premise behind isolating them is straightforward. Available evidence indicates APs are not magic bullets; it seems naive to envisage that a single drug is capable of effectively treating the multiple symptom domains of schizophrenia.
17 Just as the field of psychopharmacology has shifted to the development of selective compounds for these other domains, the search for better drugs for positive symptoms should do likewise. We have evidence of at least 3 subtypes of positive symptoms, and discuss how this might be integrated with existing criteria specifically related to TRS and URS in the goal of accelerating AP development.
Framework for Classifying Treatment Response
While our model hinges on positive symptoms, we strongly advocate an approach that ensures different symptom domains are individually evaluated from the earliest stages of intervention. Evidence suggests that positive symptoms represent the end stage of illness, with cognitive and negative symptoms identifiable in schizophrenia’s prodrome.
20,21 We also emphasize the need to differentially evaluate measures of functional outcome and subjective well-being from the illness’ outset.
22–24Regarding positive symptoms, there are criteria defining both TRS and URS.
4,9,10,25 Regarding our model, criteria for clozapine eligibility align with people who are not AP responsive, a frequently encountered subgroup in clinical practice characterized by 2 failed AP trials, that is, clozapine eligible. More recently, criteria for URS have been proposed, and these parallel what we describe as clozapine resistant.
Building on existing literature related to TRS and URS,
tables 1 and
2 summarize published criteria and our proposal. Notably, we avoid the terms TRS and URS. TRS is somewhat of a misnomer, as these people are not necessarily treatment resistant. All are clozapine eligible and some will be clozapine responsive; URS is vague, as this subsample actually represents people who are clozapine resistant. Our approach incorporates 3 changes representing conceptual shifts in thinking supported by more recent evidence, most importantly: focus on positive symptoms, removal of functioning, and removal of duration of illness in establishing treatment eligibility at any stage.
Focus on Positive Symptoms.
While APs exert a generalized effect that impacts symptom domains beyond psychosis,
26 the magnitude of effect is not comparable across domains. The success of APs in impacting either negative or cognitive symptoms is modest, leading to efforts to develop compounds selectively addressing these other features.
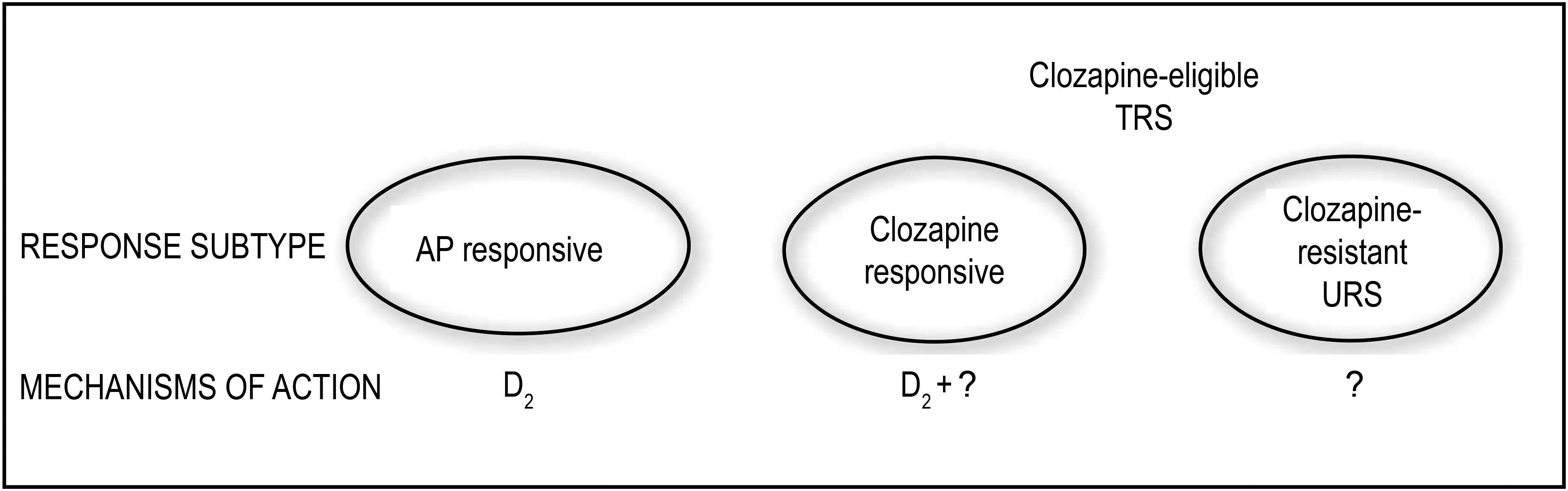
14There is a greater chance for gains in AP development if a focus on positive symptoms remains central. APs are used to control positive symptoms, the crux of TRS and highlighted in all published TRS criteria (
Table 1). It has been argued that dopamine D
2 antagonism is central to AP effects,
27 but we have at least 2 other forms of psychosis, clozapine responsive and clozapine resistant; in both cases, D
2 antagonism alone is insufficient for establishing response (
Figure 1).
28In focusing on positive symptoms, we believe there is value in employing the CGI-SCH scale.
29,30 The CGI itself has been shown to be as sensitive as the BPRS in detecting efficacy differences between APs.
31 As recommended by different sources,
32,33 the CGI-SCH also permits independent evaluation of multiple key symptom domains associated with schizophrenia (positive, negative, depressive, cognitive, and overall severity), simultaneously allowing for extraction of a value specific to positive symptoms. It is comparable with the PANSS in terms of sensitivity to change, and easier to administer, crucial if the objective includes clinical use.
29 The CGI-SCH permits scoring of severity and change, the latter also accommodating relative values, compared with absolute thresholds. It is meant to include delusions, hallucinations, and bizarre behaviour, comparable with the positive symptom dimensions of psychoticism (reality distortion) and disorganization captured in more recent remission criteria.
32Removal of Functioning in Evaluating Treatment Response.
All available criteria for TRS and URS incorporate functional impairment (
tables 1 and
2). It seems intuitive that clinical recovery or remission
32 would translate to functional recovery, although some evidence refutes this; for example, patients with first-episode schizophrenia meeting criteria for remission continue to manifest marked deficits in functional outcome.
34 Similarly, patients meeting criteria for TRS frequently demonstrate a favourable response to clozapine, but this is not necessarily matched by improvements in functional measures.
22,35 Longer-term, follow-up studies have also demonstrated that clinical and functional recovery do not parallel each other across time.
23More recently, it has been argued that other symptom domains may play a more critical role in functional outcome.
36,37 Because APs fall short in effective treatment of these domains,
14 we recommend that treatment response be confined to AP efficacy (positive symptoms) only.
Removal of Duration of Illness in Evaluating Treatment Response.
Existing criteria for TRS and URS incorporate duration of illness (2 to 5 years) in establishing candidacy for clozapine (
tables 1 and
2). Earlier, TRS and chronicity were seen as inextricably linked
4,8; however, safety concerns regarding clozapine in the early stages of reintroduction framed it as a treatment of last resort.
4 More recently, focus has shifted to earlier diagnosis and treatment, bolstered by evidence that early, effective treatment improves outcome.
34 Suggesting that TRS requires a minimum 2-year duration is contrary to this evidence and current arguments that clozapine use is, in fact, inappropriately delayed.
38,39 By current guidelines, clozapine can be implemented as early as 2 to 3 months after treatment onset.
40,41Strengths and Limitations
We have previously spoken of the model’s advantages.
15 Arguably, this is the most ecologically valid model currently available for advancing AP development, in that it uses existing clinical evidence as its foundation. It reminds us why we still prescribe APs, highlighting the central role of positive symptoms. Finally, it acknowledges clinical reality; the profound impact of persistent psychosis despite existing treatments, including clozapine, on people and resources demands a continued focus on better APs, even as we turn our attention to other symptom domains. The criteria we propose have fidelity in this regard and best optimize our chances to advance AP development.
That we distinguish our groups clinically, rather than biologically (at least at this point), represents the greatest limitation to subtyping. Nowhere is this more evident than in decision making regarding thresholds for response. One means of dealing with this has been the establishment of response thresholds, although as a strategy this has been challenged.
42 Here we propose criteria on 2 dimensions: true response (severity) and relative response (change). However, this is necessary to also capture clinical reality, especially with clozapine, which is confined to people who are more ill. There are people who demonstrate complete, or almost complete, resolution of symptoms, even after multiple AP trials, but there are also those who remain quite symptomatic despite substantially greater benefits following clozapine. Without better options, these people remain on clozapine and are, relatively speaking, responders. More recent literature on response trajectories speaks to the notion of differential treatment response and comes closest to what is observed clinically.
43–46At first glance, our proposal to subtype schizophrenia clinically may appear to contradict the recent removal of clinical subtypes in DSM-5
33; however, these 2 approaches of clinical subtyping have notable differences. The previous clinical subtypes (paranoid, disorganized, catatonic, undifferentiated, and residual) relied on symptom presentation at a specific point, and symptoms have been known to vary across various phases of the illness; hence it suffered from a lack of diagnostic stability, reliability, and validity. Conversely, the proposed subtyping by treatment response, with a specific focus on positive symptoms, is based on decades of clinical and research experience with APs, and is in line with DSM-5’s dimensional approach for symptoms. Further, a growing body of biological evidence supports such an approach. For example, recent positron emission tomography imaging studies
47,48 have reported normal dopamine but elevated glutamate levels in the brains of patients with TRS, while genetic studies,
49,50 too, have identified promising markers for TRS. Taken together, these research findings strongly suggest TRS to be a biologically distinct subtype.
The CGI-SCH has not gained the widespread use of scales such as the BPRS and PANSS, and undoubtedly the use of these other scales will continue. While we advocate for further studies comparing each scale, at least one report has proposed comparative values between the CGI, BPRS, and PANSS based on several large databases arising from clinical trials.
31,51,52 In terms of CGI–Severity, it was proposed that CGI of 4 or more is roughly equivalent to a total BPRS of 40 or more, and PANSS of 80 or more, while for CGI–Change a CGI of more than 2 represents improvement of less than 50% on the BPRS and PANSS, respectively.
Finally, the focus on a single symptom domain flies in the face of a progressive shift to view schizophrenia in the context of multiple symptom domains,
30,32,33 just as it seems at odds with the increased use of APs in other diagnoses and even off label.
53 However, the notion of focusing drug development on specific symptoms is also gaining momentum,
14 and measurement tools, such as the DSM-5
33 and CGI-SCH,
30 provide a means of achieving both. Again, we do not disavow the benefits of APs in symptoms beyond positive features; what we underscore is the magnitude of response (or lack thereof) and the logic of focusing on positive symptoms if our goal is one of better APs. Evidence has not supported their stand-alone efficacy in other key symptom domains, nor in clinical practice where extensive use of additional psychotropics in schizophrenia (for example, anxiolytics, antidepressants, or mood stabilizers) further highlights the limitations of APs beyond positive symptoms.
54Conclusions
What we have detailed builds on existing criteria for TRS and URS, with minor modifications to accommodate advances in the field. Current practice patterns actually reflect several of these; clozapine is advocated and used in people much earlier in the course of illness, with decision making regarding initiation of clozapine and evaluation of response guided by psychopathology.
55,56 Our choice to focus decision making purely on positive symptoms is shaped by the belief that AP development would best be served using this as the yardstick as it homes in on AP activity per se. We highlight pharmacotherapy here, but note that nonpharmacological interventions can be evaluated by the same standard.
Subtyping schizophrenia by treatment response affords us a distinct advantage, and there is already evidence of this. Neuroimaging data confirm that suboptimal response to conventional APs is not simply a function of suboptimal dopamine blockade.
57 Regarding clozapine response, the role of dopamine D
2 antagonism has been challenged based on its low affinity
58; however, it has also been identified that clozapine’s level of D
2 occupancy may exceed proposed thresholds associated with clinical response, albeit short-lived.
59 This does not absolutely prove a role for dopamine in clozapine’s unique efficacy, although there is additional preclinical work that indicates modifying the D
2 profile of clozapine can attenuate its atypical profile.
60Unfortunately, there has not yet been a systematic effort to isolate those demonstrating poor response to clozapine for comparison purposes with AP and clozapine responders. This clearly represents an important next step. Clozapine augmentation strategies have been ineffective, indicating that this third type of psychosis requires a novel approach. Given how little we know about this population, it is even presumptuous at present to assume it represents a single population in terms of pathophysiology. As a starting point, though, we have clear evidence of 3 subtypes of psychosis, which may represent the best opportunity currently available for advancing our understanding and treatment of positive symptoms.