Experimental Therapies for Treatment-Resistant Depression: Deciding When to Go to an Unproven or Experimental Therapy
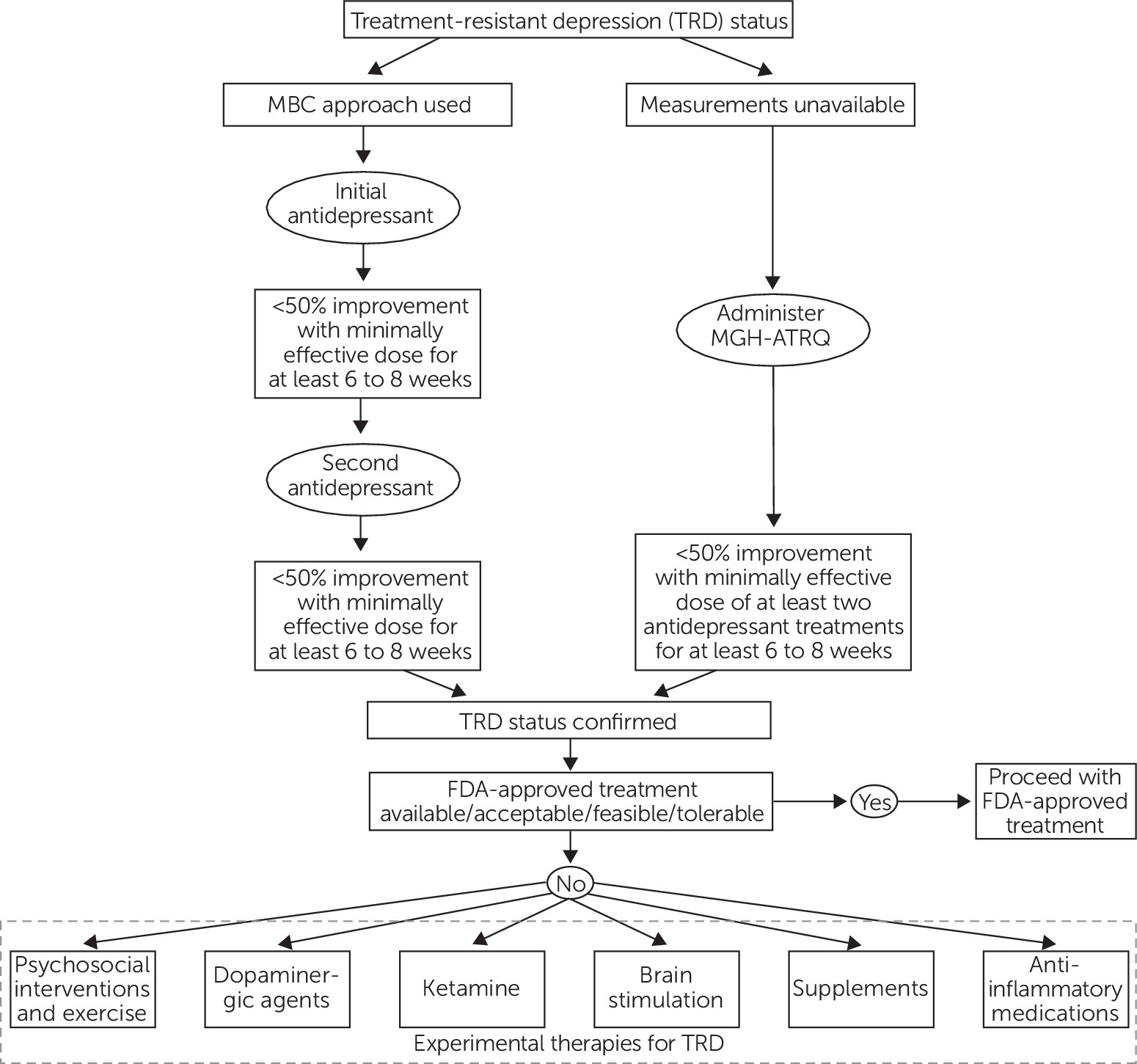
Confirming the Presence of TRD
Ketamine
Dopaminergic Agents
Supplements
Brain Stimulation
Psychosocial Interventions and Exercise
Anti-Inflammatory Medications
Conclusions
CLINICAL CONSIDERATIONS BEFORE, DURING, AND AFTER OFF-LABEL USE OF KETAMINE FOR PATIENTS WITH TREATMENT-RESISTANT DEPRESSION (TRD)
Preinfusion
Psychiatric Evaluation
Medical Evaluation
Restrictions and Prohibitions
Informed Consent
Measurement-Based Care
During Infusion
Monitoring
PRN Treatments
Postinfusion
Monitoring
Predischarge
Discharge Instructions
References
Information & Authors
Information
Published In
History
Keywords
Authors
Funding Information
Metrics & Citations
Metrics
Citations
Export Citations
If you have the appropriate software installed, you can download article citation data to the citation manager of your choice. Simply select your manager software from the list below and click Download.
For more information or tips please see 'Downloading to a citation manager' in the Help menu.
View Options
View options
PDF/EPUB
View PDF/EPUBLogin options
Already a subscriber? Access your subscription through your login credentials or your institution for full access to this article.
Personal login Institutional Login Open Athens loginNot a subscriber?
PsychiatryOnline subscription options offer access to the DSM-5-TR® library, books, journals, CME, and patient resources. This all-in-one virtual library provides psychiatrists and mental health professionals with key resources for diagnosis, treatment, research, and professional development.
Need more help? PsychiatryOnline Customer Service may be reached by emailing [email protected] or by calling 800-368-5777 (in the U.S.) or 703-907-7322 (outside the U.S.).

