However, it has been pointed out that the concept of a limbic “system” is poorly defined and that evidence for a role in emotions is often circular.
6–8 For example, whether a brain area is assumed to be part of the so-called system often depends on whether it is found to be associated with emotions. In addition, some areas, such as the hippocampus, that are clearly “limbic” on anatomical grounds have been found to be primarily related to cognitive rather than emotional functions. Furthermore, the empirical evidence relating limbic structures to emotions in humans remains indirect. The most convincing studies involve direct electrical stimulation using depth electrodes chronically implanted in patients with severe intractable epilepsy.
9–11 However, experiences reported by such patients may be related to changes in neural circuitry that are secondary to epilepsy. These experiences may also result primarily from the activation of neocortical efferents rather than from activation that remains intrinsic to limbic circuits, as local recordings cannot rule out this possibility. Finally, functional neuroimaging studies using patients with pathological emotional conditions (such as depression) or emotion-arousing stimuli have not supported the idea of an integrated limbic system responsible for all emotional phenomena. Neocortical areas are typically found to be activated as well.
12,13To go beyond the simplistic idea of “emotions” being the expression of a monolithic “limbic system,” it is necessary to determine 1) what responses—emotional or not—are associated with the selective activation of limbic structures in the normal human brain; and 2) what associations exist between particular structures (or groups of co-activated structures) and specific components of the subjective, autonomic, and endocrine aspects of emotional responses.
We used the local anesthetic procaine hydrochloride as a pharmacological probe. In animals, intravenous administration of procaine induces electrical activity in limbic structures while suppressing activity in neocortical structures.
15–17 In humans, procaine induces emotional, autonomic, and endocrine responses comparable to those of direct electrical stimulation of the amygdala.
17,18 The mechanism through which procaine
selectively activates limbic structures is not known, but it may be related to the particular distribution of mu-opiate and acetylcholine receptors
6 or corticotropin-releasing hormone receptors
18 in limbic and paralimbic structures. It may also be related to the specific histological characteristics of limbic structures (which, for example, make them a specific target of the herpes virus in the central nervous system
6).
Limbic stimulation with intravenous procaine is well suited for PET studies because it is a safe procedure in healthy volunteers, produces peak effects within 2 minutes, and is of short duration (about 5 minutes). In the present study, we recorded subjective and physiological responses to procaine HCl in healthy volunteers while simultaneously measuring changes in rCBF using PET imaging of oxygen-15 water.
DISCUSSION
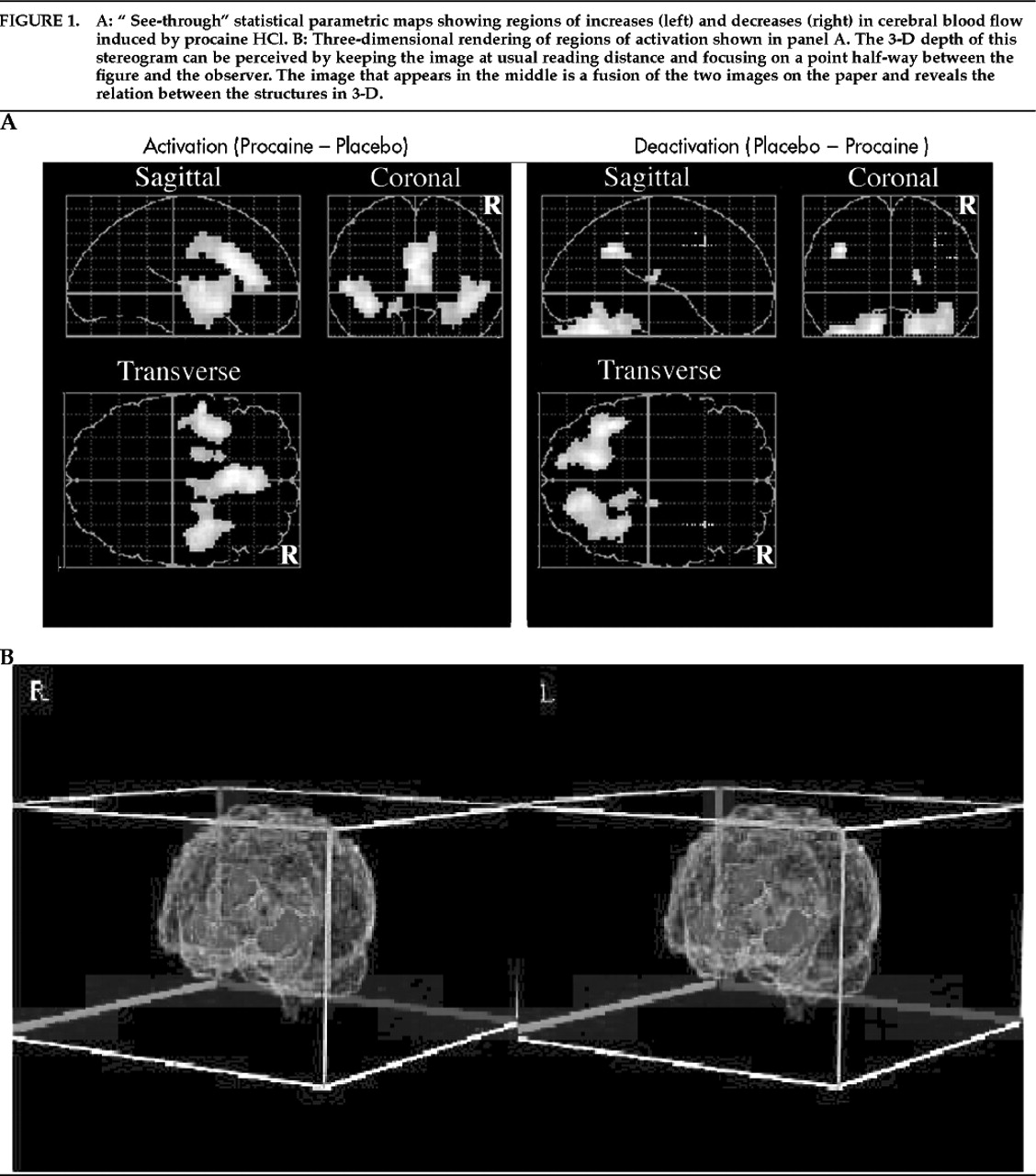
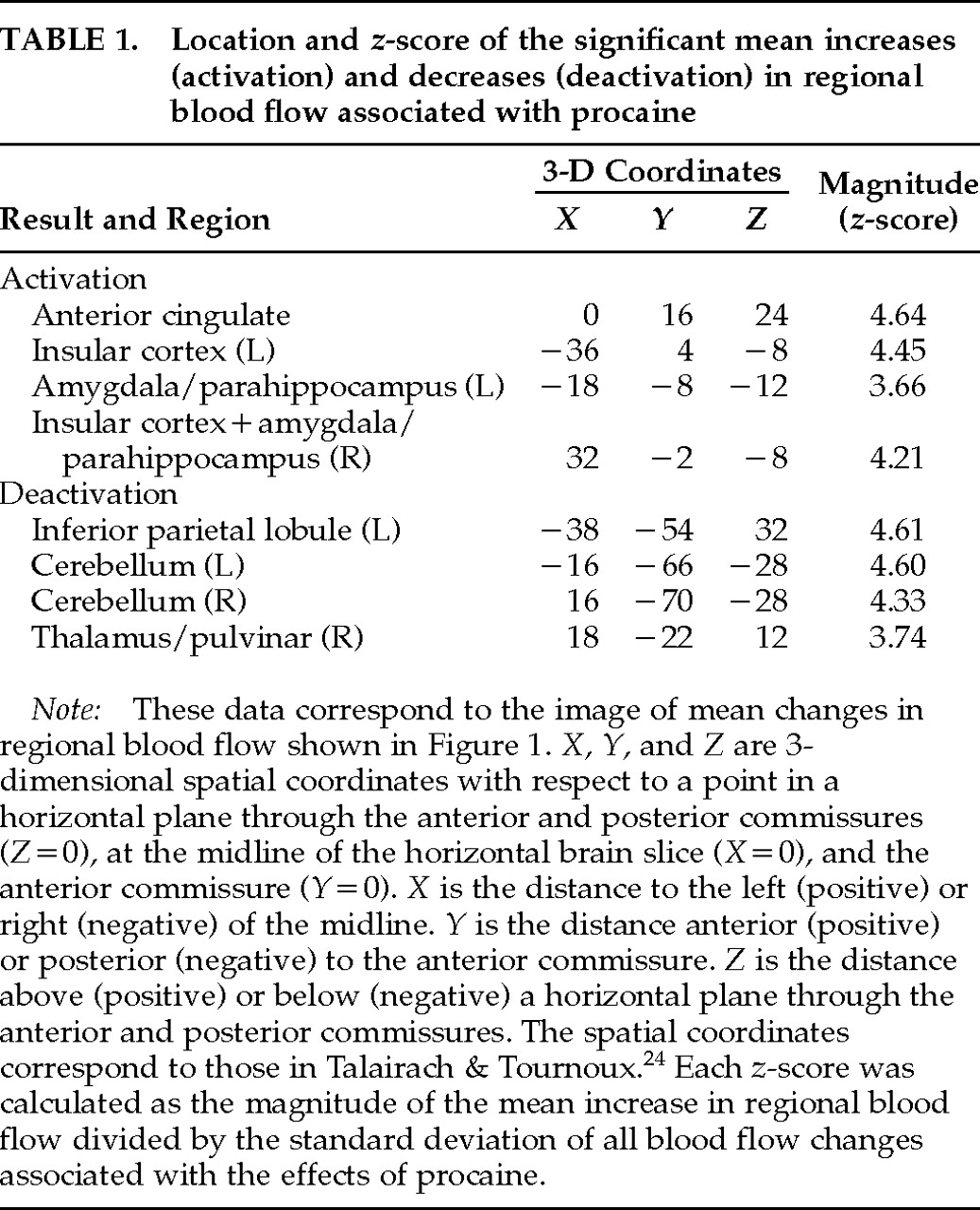
To summarize, the present study demonstrates that selective activation of an anterior limbic and paralimbic network, in the absence of significant neocortical activation, induces powerful emotional and visceral experiences in healthy subjects. Our results confirm the presumption, based on animal and clinical studies, that intense emotions and related somatic and visceral sensations are an important functional correlate of the activation of limbic and paralimbic areas in the normal human brain. Furthermore, the highly selective activation of anterior limbic/paralimbic structures with a pharmacological probe that is distributed diffusely demonstrates that such phylogenetically older brain circuits have distinctly different pharmacodynamics from those of neocortex and that they can be selectively targeted by particular drugs—a fact that has long been suspected from the selective effects of psychiatric medications.
The marked elicitation of somatovisceral phenomena is consistent with suggestions that these sensations are mediated by the insular cortex
30 and possibly by part of the cingulate cortex.
28 The insula in particular is a highly polymodal region, receiving visceral, olfactory, gustatory, and somatosensory inputs. Although poorly understood, it probably plays an important role in relating interoceptive signals to information from other modalities. Electrical stimulation of the insula has also been specifically associated with gastrointestinal sensations.
31,32 Functional imaging studies have only begun to address the function of insular cortex. Activation has been observed in studies of phobic patients stimulated with phobic stimuli
13,33 as well as during practice of a cognitive task.
34 On the basis of our results, we suspect that insular cortex activation in these other studies reflects the presence of somatovisceral symptoms—either associated with anxious anticipation or with a long, protracted PET scanning session.
We believe that robust activation of the anterior cingulate cortex in our study is related to the motivational and attentional function often attributed to this structure.
28,35,36 The anterior cingulate cortex has been implicated in tasks that demand attention (such as the Stroop task) and in the response to motivational states (for example, spontaneous vocalizations as opposed to evoked vocalizations). It is clear that the strong effects of procaine on mood, sensory perception, and somatovisceral sensation powerfully mobilized subjects' attention and could therefore be expected to activate anterior cingulate cortex. (Indeed, one subject graphically described this combined state of attention and somatovisceral sensations as “being grabbed by the balls.”)
Finally, the amygdala has been implicated in emotional responses in general
37 and in the fear response in particular.
38–40 Given the significant number of subjects who experienced anxiety or fear in response to procaine, it may not be surprising that we observed activation in the amygdalae/parahippocampal region bilaterally. Yet, surprisingly few functional imaging studies have documented amygdala activation in relation to emotional states.
It is important to note that several PET studies of induced emotions have failed to document limbic or paralimbic activation in the presence of very strong emotional responses, including cocaine-induced euphoria and panic anxiety.
41–44 These results suggest that limbic or paralimbic activity may be sufficient but not necessary for certain emotional experiences to occur (see, for example, Lane et al.
45) or that some emotional experiences do induce activity in such structures, but below the threshold detectable with PET. Such a failure to document limbic or paralimbic activity in studies of induced emotion or of emotional disorders might also reflect a number of methodological problems.
46 However, our results significantly overlap with the pattern of rCBF evoked in normal subjects following infusion of the panic-inducing agent cholecystokinin tetrapeptide (CCK
4), reported by Benkelfat et al.
47 In that study, activation of left anterior cingulate, and of insular cortex and amygdala bilaterally, was observed in the absence of cortical activation.
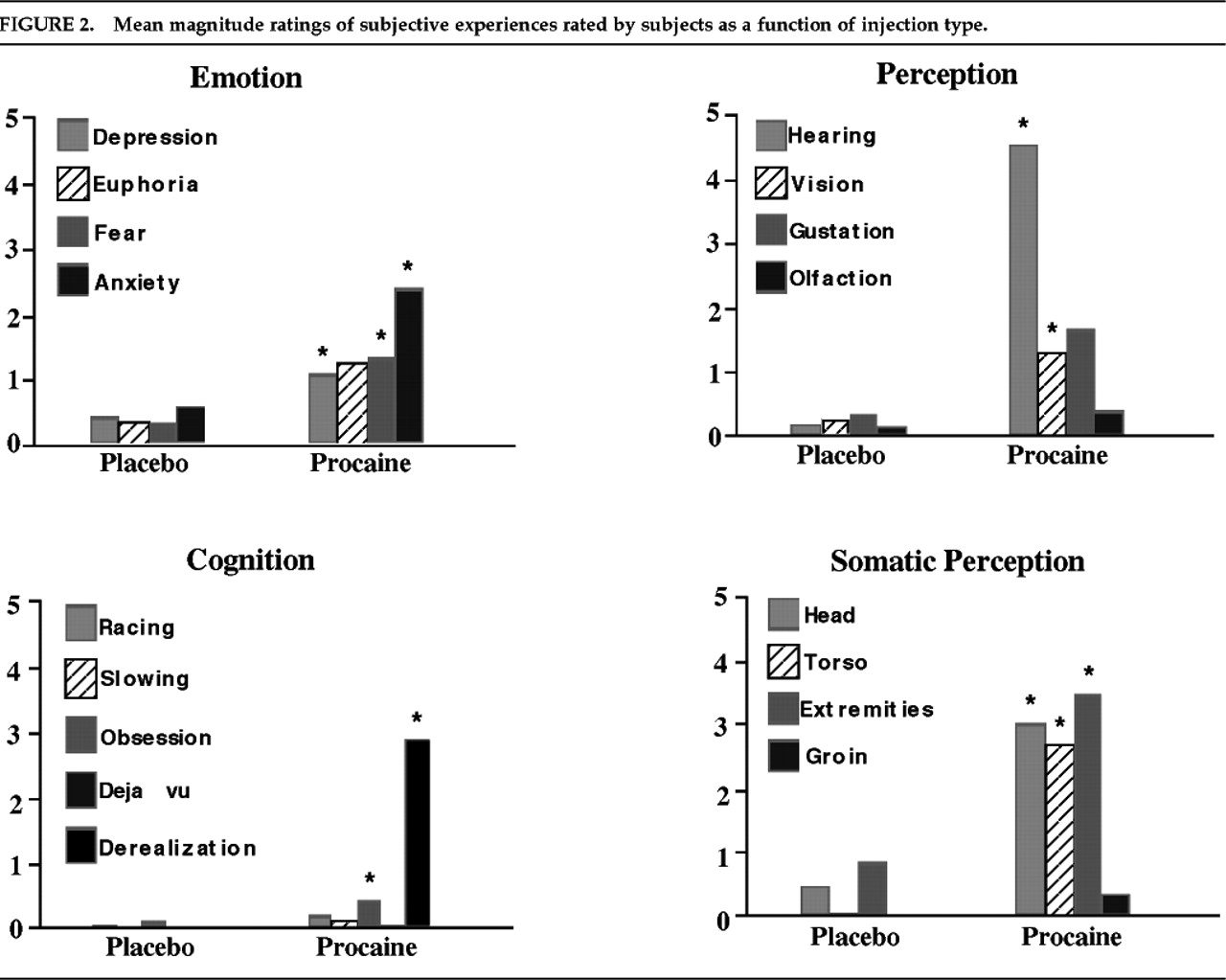
Both the nature and the variability of the subjective experiences we observed are perhaps easiest to describe in comparison to the clinical manifestations of partial complex seizures (PCS), as previously noted by Stark-Adamec et al.
48 These phenomena are also comparable to the responses observed with depth electrode stimulation of limbic structures, such as the amygdala and parahippocampal gyri. Fear is the most common experiential phenomenon described during such experiments.
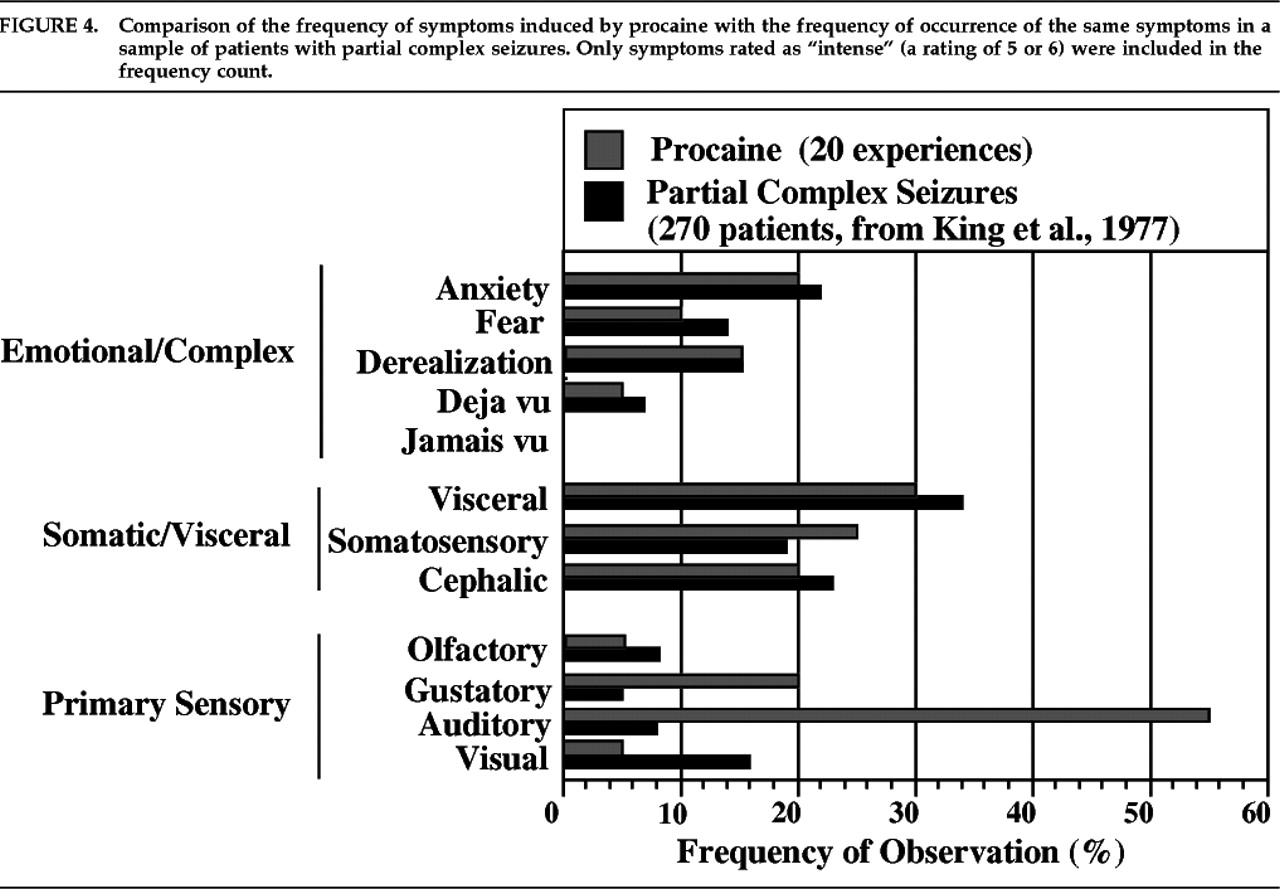
10,11 Figure 4 illustrates the similar distribution of subjective experiences in PCS and those following procaine injections in our study. Experiential phenomena during the aura phase of PCS are taken from King and Marsan.
49 Only PCS phenomena that were reported in that study and were directly inquired about in our questionnaire were included for comparison. As in that report,
emotions refers to subjective feeling of an unexpected and self-limiting emotional state, and
visceral sensation refers to localized warmth, pressure, or a rising or sinking feeling experienced inside the chest or abdomen.
Cephalic sensations observed were pressure, warmth, tingling, or numbness localized to the head or face.
Derealization refers to a feeling of unreality of immediate surroundings.
Somatosensory sensations included primarily tingling and numbness but also increased acuity of touch sensations.
Visual phenomena were illusions such as changes in light, color vision, or object shadows.
Auditory phenomena were unformed hallucinations, typically described as a loud buzzing or ringing heard equally in both ears and an enhancement of auditory acuity.
Olfactory symptoms consisted of a hallucination of unpleasant smell (“acrid pungent smell”).
Gustatory symptoms consisted of hallucinations of a bitter or metallic taste. The qualitative and quantitative similarity to experiences of epileptic patients was most impressive with respect to emotional and viscerosomatic phenomena. In contrast, procaine may have induced primary sensory illusions and hallucinations more frequently than is typically reported in temporal lobe epilepsy.
The striking similarity of the subjective experiences we recorded and the phenomenology of epileptic auras demonstrates that many of the symptoms experienced by patients with PCS can be a direct consequence of the activation of normal anterior limbic and paralimbic structures, in the absence of an epileptic disease process. We observed these phenomena in almost all of our healthy subjects, none of whom had a history of seizure disorder or psychiatric disorder. Furthermore, the comparable frequency of different subjective phenomena after procaine injection and during PCS auras suggests that anterior limbic stimulation tends to elicit particular symptoms with a comparable frequency independently of the nature of the stimulus (chemical or electrical).
We should note, however, that the emotional phenomena induced by procaine did not include the elicitation of vivid memories often reported by epileptic patients following limbic stimulation. Bancaud et al.
9 have suggested that such experiences are related to temporal neocortical activation, which was not observed in our blood-flow maps following procaine injections. Our study therefore tends to support their conclusions about the anatomical origin of vivid memory phenomena in PCS.
The elicitation of panic by intravenous procaine has not been reported previously (except by Ketter et al.
14) and has likely been underestimated in the present study, since these symptoms were not directly probed by our questionnaire. Notably, there have been several reports of an association of panic with temporal lobe abnormalities
50–52 and with PCS.
53–55 There are also reports of successful treatment of panic disorder with limbic anticonvulsants.
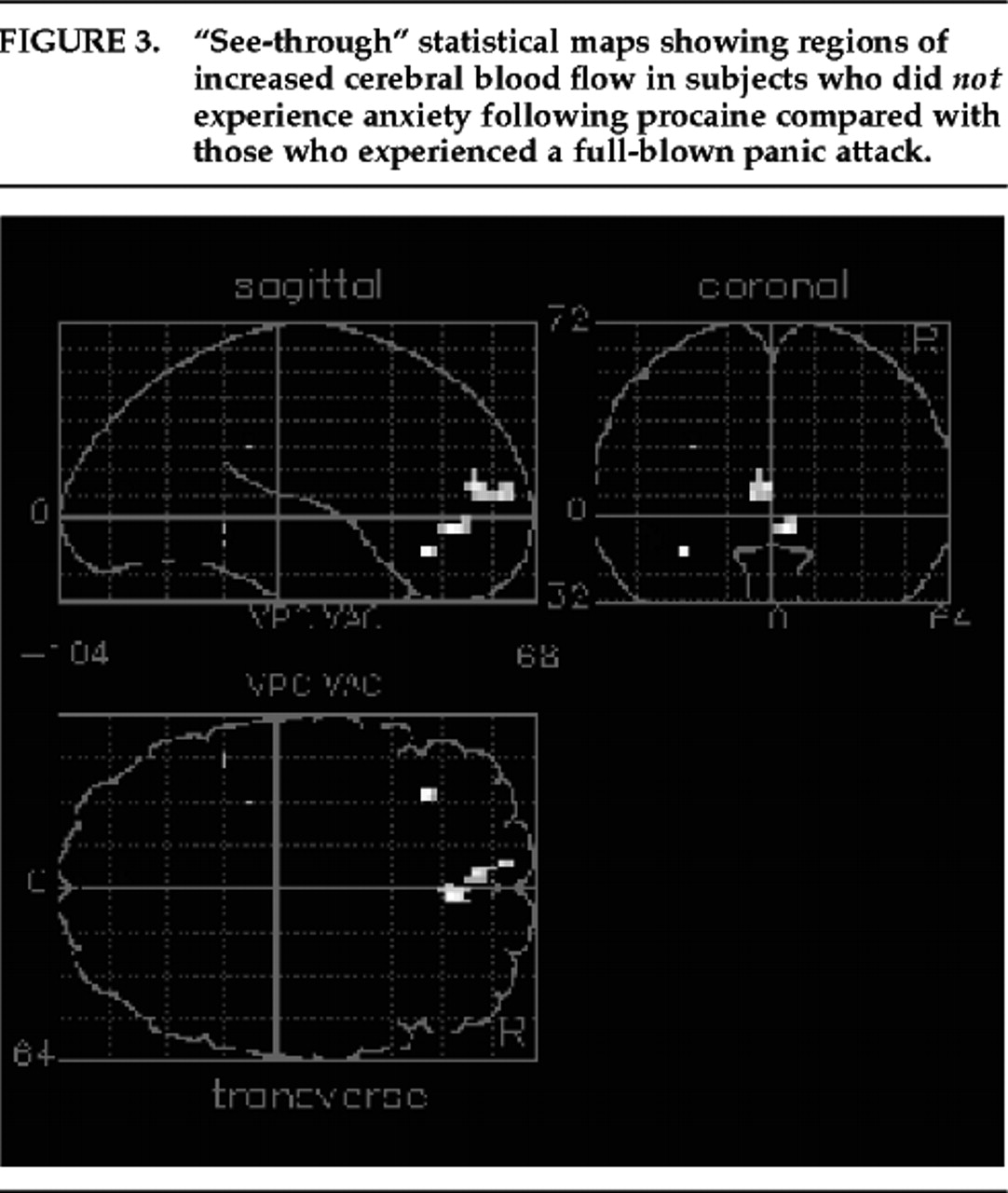
56 As in Ketter et al.,
14 we found a positive correlation between anxiety and blood flow in the left amygdala but not the right. This unilateral finding confirms that, in humans in particular, there may be a lateralization of function between the two amygdalae. Our exploratory analysis of panic versus no-anxiety scans also suggests that subjects who did not experience anxiety may have activated areas of the frontal lobes (tip of the anterior cingulate and left inferior frontal cortex) that could be relevant to inhibiting an anxiety response. Several authors
57–59 have proposed that inferior frontal regions contribute to the modulation of emotional responses and that the left frontal cortex in particular is associated with positive affect or inhibition of negative affect.
58A possible concern with our results is that procaine may have a direct vasoconstricting or vasodilating effect on CNS vasculature that could alter activity in the vascular tree directly rather than modify neural activity. Indeed, the areas of activation observed in our study overlap in part with the course of the anterior and middle cerebral arteries. However, there are a number of reasons to believe that the blood flow changes we observed are related to changes in neural activity rather than to vascular effects. First, the impact of an initial bolus of arterial activity on the PET image has been considerably lessened in our study by using the minor modifications in the technique described by Raichle et al.
20 The original approach initiates scanning when tracer activity first enters the head (approximately 14–16 seconds after injection), which maximizes the imaging of high arterial activity. Our approach, in contrast, delays onset of scanning until 20 seconds, thus avoiding this initial arterial phase. Also, the scan has been lengthened to 60 seconds from the original 40 seconds, which further dilutes any remaining vascular effect by about 50%. Second, procaine is known to induce electrical activity in limbic structures, as recorded from intracranial electrodes in animals and from intracranial
10,11,53 and scalp electrodes in humans.
60 Third, the subjective reports of patients were almost identical to those of patients experiencing epileptic activity in limbic structures, and did not include complaints of headaches or nausea as seen in patients with vascular CNS changes.
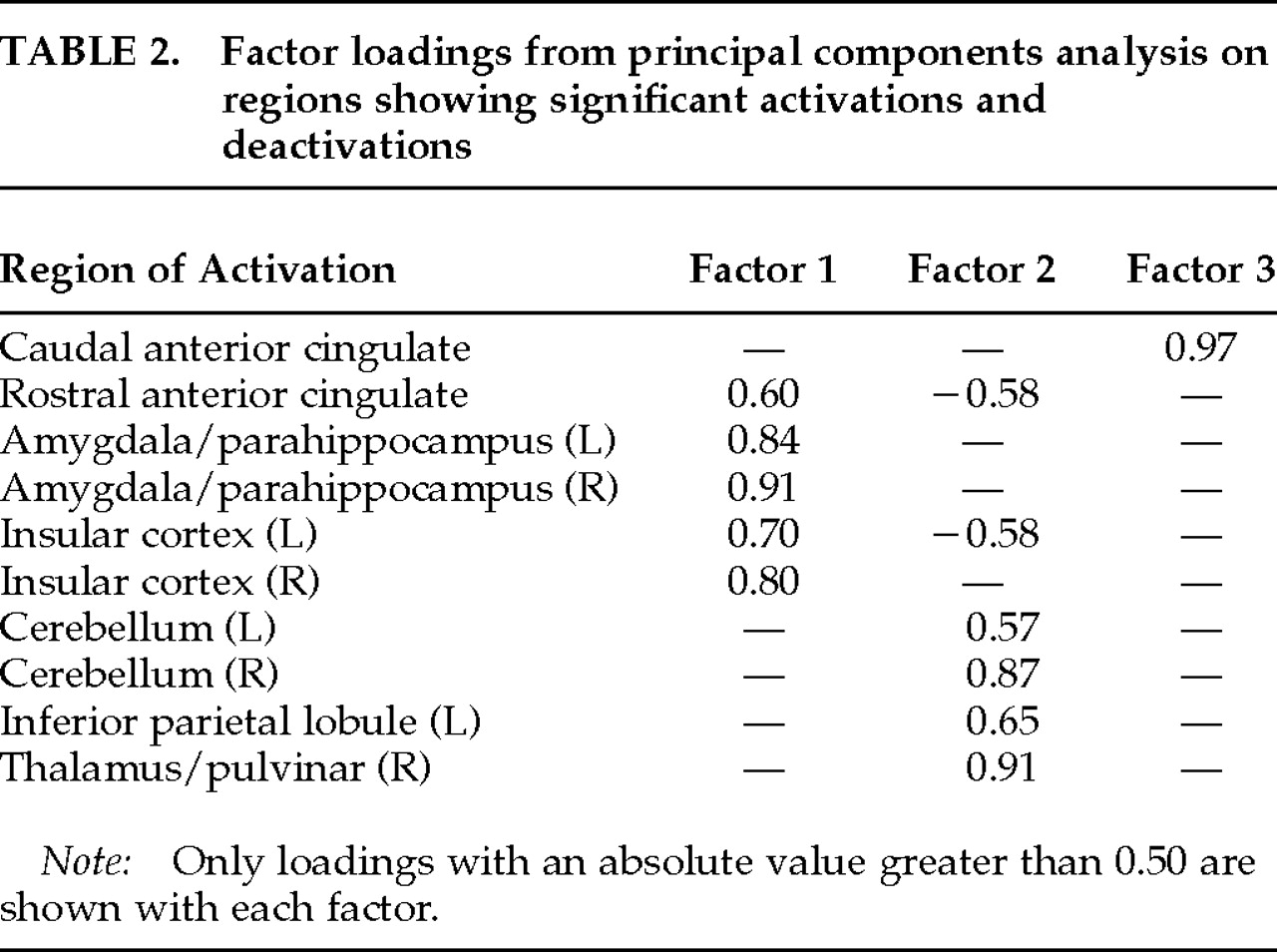
Our PET deactivation results could suggest that emotional experiences are related to a relative bilateral deactivation of the cerebellum. Cerebellar deactivation has not, however, been implicated previously in emotional experiences, either in healthy volunteers or in psychiatric patients. Our exploratory PCA did not aid in interpreting this cerebellar deactivation, since both the first (anterior limbic and paralimbic structures) and second (cerebellum, inferior parietal lobule and thalamus) factors were correlated with emotional and somatosensory changes associated with procaine (data not shown). However, it is possible that cerebellar deactivation contributed significantly to the marked sense of derealization reported by our subjects following procaine injections. Parietal cortex has been associated with perception of extrapersonal space, and the thalamus is critical to the integration of sensory signals.
6 Deactivation of these structures may thus be directly related to the experience of derealization. An alternative hypothesis is that cerebellar deactivation reflects a “steal phenomenon” without any true relationship to derealization. It is also possible that the relative cerebellar deactivation is an artifact of the global normalization process used prior to analysis, which removes the robust increase in whole-brain absolute blood flow seen with procaine.
14 Investigation of depersonalization and derealization has received little attention in biological psychiatry, however, and the possibility of a relationship between these symptoms and parietal, thalamic, and cerebellar function may warrant further research.