Traumatic Brain Injury (TBI) is an often devastating and common problem that affects today's society. Only rough estimates of incidence and sequelae can be obtained, as many of the milder injuries go initially unreported. Current estimates note an incidence of 1.5 to 2 million a year in the United States, most commonly from motor vehicle accidents.
1 It occurs bimodally in age groups from the teens through the twenties and after age 70. 80% are mild injury; 10%–13% moderate; and 7%–10% severe.
2 A large portion of these surviving patients will have cognitive or emotional sequelae and will need neuropsychiatric interventions years after the injury. For example, 10%–75% of these patients have been reported to have depression.
3 It is therefore essential to try to understand the pathophysiology of brain injury, be able to perform adequate diagnostic evaluations, and intervene as early as possible, in hopes of limiting continual injury processes.
In the last 20 years, improved clinical management has significantly reduced mortality, and for those who survive, new research has identified secondary injury processes that continue in the first hours and days postinjury that may be amenable to therapeutic intervention. Much of the current work in TBI focuses these processes and their therapeutic management potential. An excellent review of the history of research in acute brain injury was recently written.
4 Beginning with a review of the anatomy of the neuron, its subcellular structure, and normal function, the authors discuss the biomechanics of the forces of TBI (rotational and translational), the implications of injury impact location, and the microscopic sequalae of the initial impact. Primary, secondary, and tertiary processes of injury to axons are emphasized. Difficulties in creating laboratory models of acceleration/deceleration injury, the problems associated with current research strategies, and how the theories of axonal injury have changed over the years are also addressed. The review closes with a consideration of imaging techniques for the acute TBI patient as well as potential interventions that may be on the horizon.
4 This current communication focuses on three new concepts—new information regarding microscopic pathophysiology of secondary axonal injury; new diagnostic tests to quantify injury; and new imaging techniques that better identify axonal injury in TBI patients.
DIFFUSE AXONAL INJURY—MICROSCOPIC EVALUATION
Diffuse Axonal Injury (DAI) is a term applied to TBI-induced scattered destruction of white matter tracts. It was first described in 1955 by Lindenberg in patients with post traumatic hemorrhage of the corpus callosum.
5 DAI can only be definitively diagnosed postmortem. DAI, as detected via histological assessment, is generally associated with shearing injuries especially those involving acceleration/deceleration. Typically, these patients show significant morbidity without evidence of mass lesions on early imaging. Correlation between axonal injury and the presence of neuropsychological abnormalities, even years after injury, have been described [for review see
6].
Recent research has determined that initial axonal injury is an ongoing process, evolving over several hours to days. When first identified almost 50 years ago in postmortem analysis using silver salts, the finding of grossly swollen axonal bulbs in humans suggested that the axons were mechanically torn at the moment of injury, with an expulsion of their axoplasm to form the axonal bulb.
4 Experimental studies in animals as well as humans, by Povlishock and colleagues in the early eighties, rejected this premise, showing that the forces of injury focally perturbed the axon, leading to impaired axonal transport that then led to focal axonal swelling, followed over several hours, by axonal disconnection.
7 These findings have been confirmed by multiple laboratories leading to universal acceptance of the phenomenon that diffuse axonal injury involves progressive/evolving change and ultimately leads to axonal disconnection many hours postinjury.
In addition to the recognition of the evolving changes associated with DAI, Povlishock and colleagues have also followed the downstream consequences of the axonal disconnection showing that the diffuse axonal injury translates into diffuse synaptic terminal loss that then sets the stage for either adaptive or maladaptive neuroplasticity, perhaps explaining some of the morbidity found in these patients.
7 Recognizing the evolving nature of diffuse axonal injury and its linkage to focally impaired axonal transport, more contemporary studies seeking to identify DAI in both the laboratory and forensic setting have relied on antibodies targeting amyloid precursor protein (APP), which is known to be delivered by axonal transport. Its anterograde passage is impeded by axonal injury, with subsequent accumulation in the axonal swelling. At present, antibodies to APP are the gold standard in both the experimental and forensic setting as well as in the evaluation of many preclinical drug studies focusing on the ability of various drugs to attenuate APP-positive swellings.
4While all of the research and clinical community have agreed that axonal swellings and disconnection are the consequence of injury and, as such, contribute to the adverse consequences of traumatic injury, new information arising from the experimental setting suggests that not all traumatically injured axons may go on to swell.
8 This suggests that studies focusing on APP-positive swellings may underestimate the total number of axons injured by the traumatic event. This potential was first recognized by Povlishock and colleagues in studies seeking to better understand the subcellular mechanisms initiating traumatically induced axonal injury and swelling. Previous studies had shown that the traumatic episode perturbed the interaxonal cytoskeleton and impaired axonal transport. The assumption was advanced that these events were casually interrelated within the same axon. Recently, however, more critical assessment of these issues using double label immunofluorescent approaches, suggested that this premise was overly simplistic, underestimating the complexity of the pathobiology of DAI.
9 Specifically, via double label approaches, some axons were recognized to demonstrate cytoskeletal alteration and detachment, without the subsequent formation of an axonal swelling. In contrast in other axons, axonal swelling occurred, sometimes independent of overt cytoskeletal damage. These studies have been subsequently confirmed via other approaches, collectively illustrating the complexity of the pathobiology of traumatically induced axonal change and the danger of relying on one individual marker of axonal injury to estimate the degree and extent of the traumatically induced axonal change.
9,10 Preliminary studies conducted by Stone and colleagues in humans suggest that these same events described in animals are also ongoing in man, thereby suggesting the need for caution in human postmortem and forensic analyses that rely exclusively on markers targeting axonal swelling as the sole endpoint.
9IMAGING STUDIES
Although it has been known for some time that MR imaging is more sensitive than CT in detecting axonal injury, both methods are widely used.
5,6 CT examination, still the standard for life-threatening acute hemorrhage, is robust and relatively inexpensive. In contrast, while MR is much better for detection of white matter lesions, the exams take longer, are more expensive, and require special nonmagnetic ventilators, cardiac monitors, and other medical equipment. Both conventional and developing MR techniques can add value to the clinical assessment of TBI. Gradient echo (GE) images are sensitive to magnetic susceptibility (T2*) and can demonstrate even very small areas of hemorrhage.
23–27 Such lesions are more easily visualized with this technique because the hemorrhagic blood creates a local magnetic field disturbance, causing a loss of signal. The number of small hemorrhagic lesions identified on T2* weighted GE images correlated with GCS score in several studies.
23,26,27 In addition, lesion locations were appropriate to explain all focal neurological signs and symptoms in the acute phase (≤ 3 weeks) in one study.
26 Nonhemorrhagic lesions are better visualized on T2 weighted spin echo sequences, especially those obtained using fluid attenuated inversion recovery (FLAIR), a technique that incorporates suppression of the bright signal from CSF. In a prospective study, 33 patients with normal CT scan but abnormal neurologic status underwent MR examination within 48 hours of injury.
5 This group obtained T1 weighted echo planar, T2* weighted GE, T2 weighted FLAIR, and T2 weighted turbo spin echo images, and found that MR demonstrated more nonhemorrhagic lesions than CT. The authors noted that presence of nonhemorrhagic lesions was associated with a relatively good clinical outcome. However, outcome was measured only with the GOS, a relatively insensitive measure. It does not, for instance, include in-depth neuropsychological or neuropsychiatric evaluation. In another prospective study, a group of 21 patients with DAI underwent MR imaging within 24 hours of injury, then again on days 1, 3, 7, and 14.
28 The signal intensity of the corpus callosum was measured on FLAIR images at each time point. The study found a positive correlation between the duration of unconsciousness and the maximum signal intensity of the corpus callosum, which occurred most commonly on day 7 (range 3–14). In addition, higher signal intensity was associated with an unfavorable outcome at 6 months, as determined by the GOS.
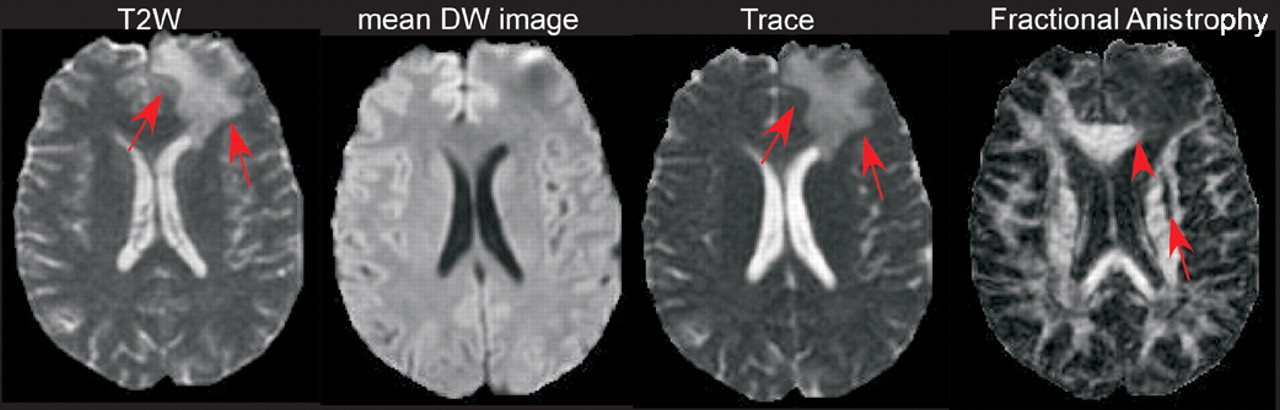
Newer types of MR imaging provide quantitative measures and show promise for increasing identification of DAI. A recent retrospective study examined 25 patients within 48 hours of TBI, comparing lesion visualization on T2 weighted fast spin echo, FLAIR, T2* weighted GE, and diffusion weighted (DW) images.
25 Of 427 lesions identified on at least one type of image, 58% were detected on T2/FLAIR, 47% were detected on GE, and 22% were detected on both. DW imaging was the most sensitive, allowing visualization of 72%, including all nonhemorrhagic lesions. Importantly, 16% of lesions were visible only on DW imaging. All of these were hyperintense in appearance. The apparent diffusion constant (ADC) was decreased in the majority of lesions visible on DWI, indicating more restricted diffusion, perhaps an indication of cytotoxic edema. Approximately one-quarter displayed increased ADC, which may indicate the development of vasogenic edema. Consistent with expectation, many hemorrhagic lesions (23%) were seen only on the GE images. No follow up data was presented, so the clinical significance of the lesions identified could not be assessed.
Three studies used diffusion tensor imaging (DTI) to evaluate DAI. One measured diffusion anisotropy of the corpus callosum (splenium, body, genu) in 10 patients 2 weeks to 8 months after TBI (initial GCS scores ranged from 3 to 14) compared to normal individuals.
29 Diffusion anisotropy was decreased by an average of approximately 25% in at least one region of the corpus callosum in all 10 patients, indicating disruption of the normally highly ordered structure. Decreased anisotropy was found even in areas that looked normal on both FLAIR and GE images. No correlation with functional measures was provided. A case report of a child with hemiparesis subsequent to a severe TBI (comatose for 3 days) reported that abnormalities on conventional MR did not correlate with clinical symptoms.
30 DTI demonstrated decreased anisotropy in the cerebral peduncle of the midbrain. A preliminary study evaluated diffusion anisotropy of areas that appeared normal on standard CT and MR imaging in 5 patients with mild TBI (initial GCS score 13 to 15).
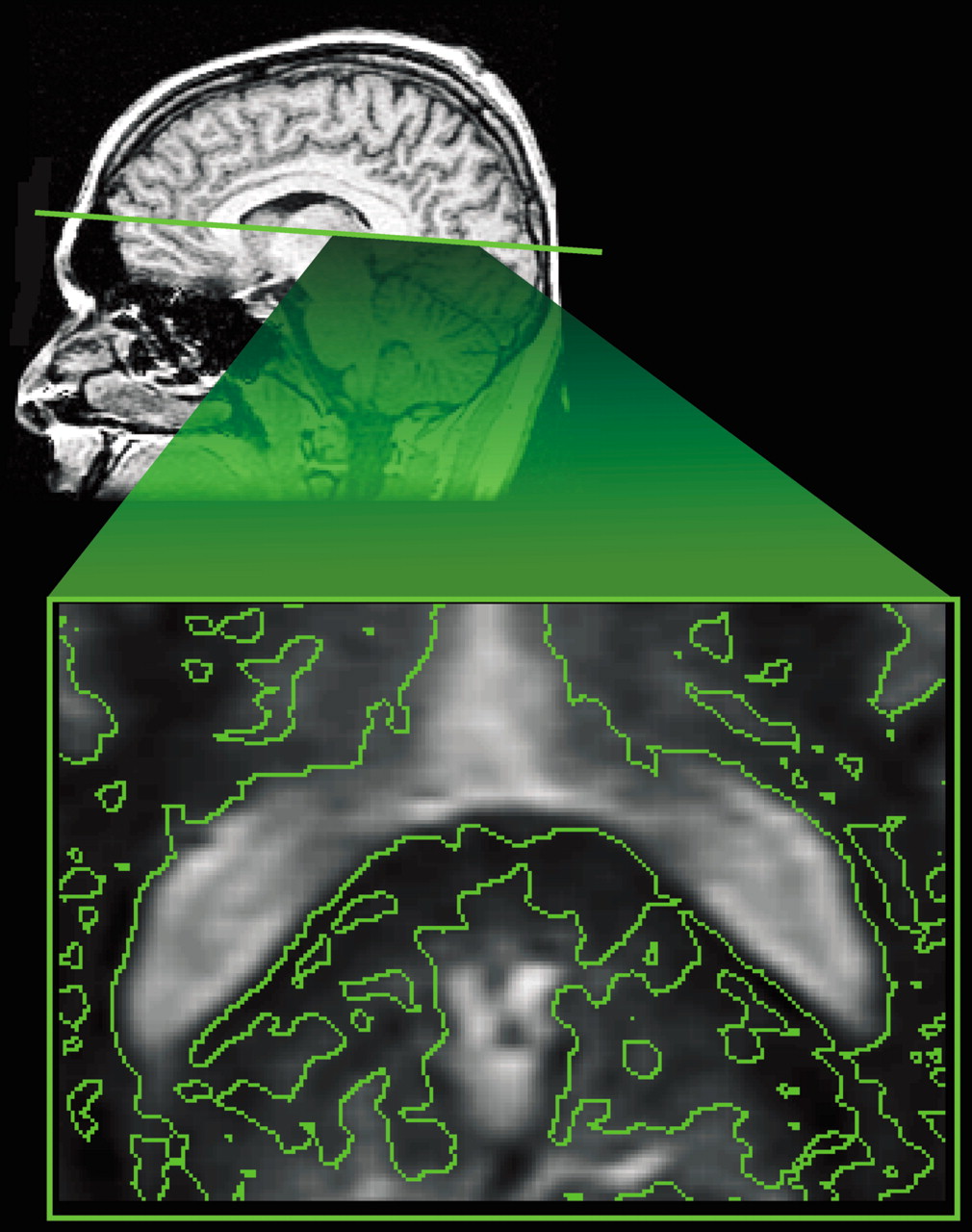
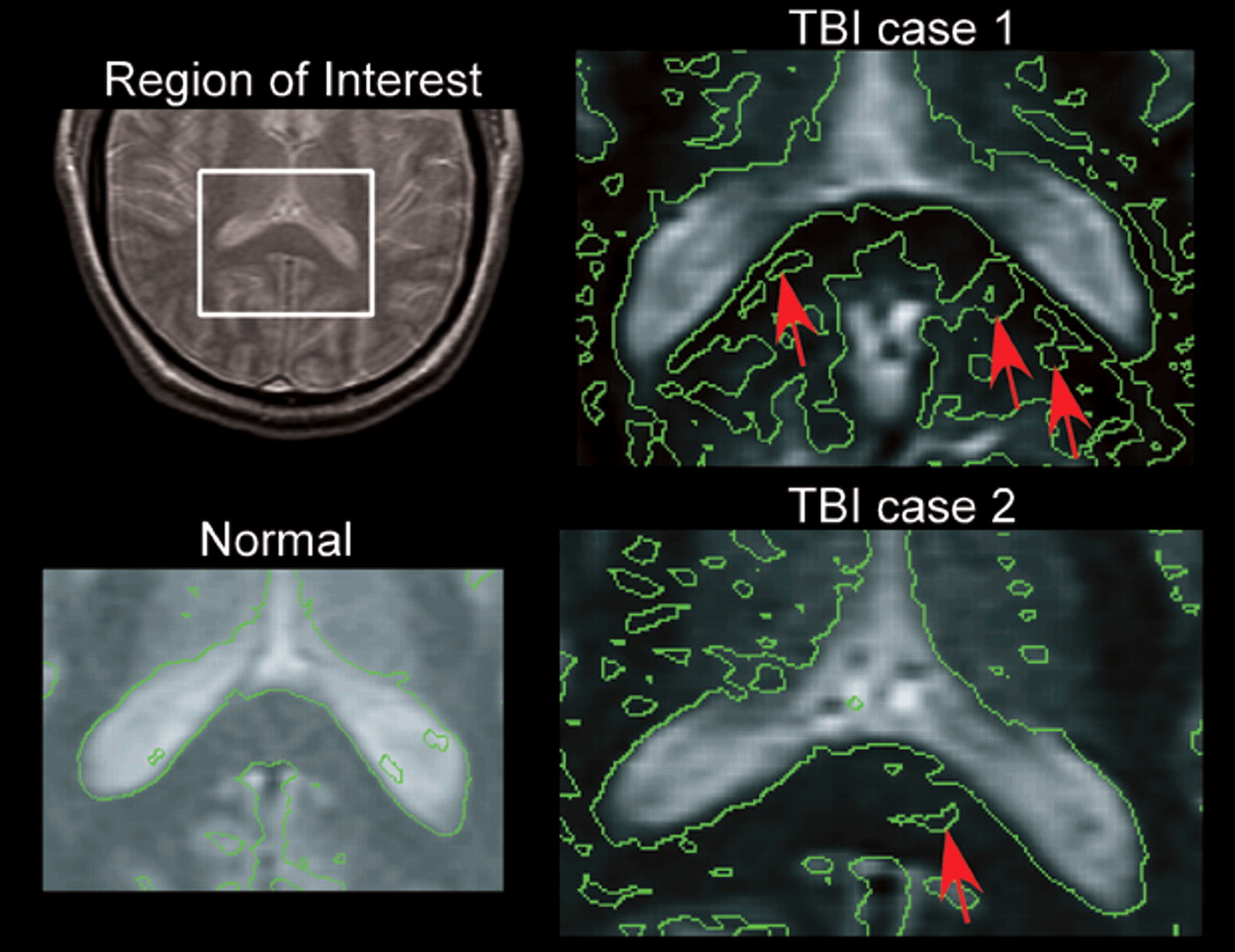
31 All were imaged within the first 24 hours and 2 were imaged again at one month. There were areas of reduced anisotropy in regions that appeared normal by other imaging techniques in all 5 patients, most often in the internal capsule and corpus callosum (
Figure 1). The authors noted that the decreased anisotropy in areas without edema or hemorrhage (which would have been seen on the other types of images gathered) indicate that diffusion parallel to the main axis of the axons has become impeded. They suggested misalignment of axonal membranes and of the cytoskeletal network as possible causes. By 30 days, some areas had returned to normal values, while others were still decreased. The authors suggested that these patterns may correspond to areas in which repair processes have been successful versus areas in which injury has progressed. All these studies indicate that various measures of water diffusion may be more sensitive to DAI than conventional MR imaging.
Magnetization transfer (MT) imaging is another quantitative MR technique that has been found to be related to tissue structure and thus can be used to assess structural integrity. In one study of DAI in an animal model, MT imaging results were found to be related to pathologically-proven injury.
32 In a later study conducted by the same group, 28 patients who had experienced loss of consciousness as a result of head injury were compared to 15 controls.
33 Most were first imaged within 2 weeks of injury (range 1 to 29 days). Follow up scans were obtained in 10 patients. Magnetization transfer ratios (MTRs) were calculated for all white matter areas that were abnormal on the T2 weighted images as well as for areas of normal appearing white matter. Outcome measures (GOS) were obtained 3 months to 3 years after injury. Decreased MTR was found in normal appearing white matter only in patients that had persistent functional deficits at follow up. However, absence of decrease in MTR in normal appearing white matter was found in patients with outcomes that ranged from very good to poor. In a subsequent related study, 13 patients who had experienced mild TBI were examined with conventional MR imaging and MT imaging several months to several years following injury.
34 Neuropsychological testing was done in 9 patients, all of whom were found to be impaired. Most of the patients in the study (12/13) had negative conventional MR imaging but all were symptomatic. Contour mapping of the MTR revealed areas of profoundly decreased MTR in the splenium of the corpus callosum in 4 of these patients (
Figure 2). A moderate correlation was found between a test of verbal learning and MTR for the splenium. Results of this study suggested that MT imaging may be sensitive to mild TBI when other modalities fail. In another study from this group, MT imaging and
1H (proton) MR spectroscopy of the splenium of the corpus callosum were obtained in 30 patients with TBI.
35 Initial GCS scores ranged from 3 to 15 (mean score, 11). Assessment was done between 2 and 1129 days after injury. Outcome (GOS) was assessed a minimum of 3 months after injury. No correlation between MT imaging and outcome was found. However, a decreased N-acetylaspartate to creatine ratio (a possible indication of neuronal loss) was associated with poorer outcome. These results are consistent with prior suggestions that abnormal MR spectroscopy results may predict a poor clinical outcome.
36 Limitations of this study include the wide range of times from injury to examination and injury to assessment of outcome.