While the precise mechanism of antipsychotic response is not known, antagonism of dopamine transmission is likely to play an important role. The dopamine system is a neuromodulatory system that arises from cells in the midbrain and has discrete projections to the mesolimbic, striatal, and cortical areas of the brain. These midbrain neurons release dopamine, which acts by means of actions on dopamine receptors. There are five known dopamine receptors, classified into “families” on the basis of similarities in gene-sequence and functional effects. The D
1 family includes the D
1 and D
5 receptors, while the dopamine D
2 family includes D
2, D
3, and D
4 receptors
(1). Of these, the dopamine D
2 family, and in particular the dopamine D
2 receptor, has been of sustained interest from the point of view of antipsychotics. In vitro studies show that all antipsychotics bind to the dopamine D
2 receptor
(2,
3), and in most cases the clinical doses correlate with their affinity for the dopamine D
2 receptor
(2,
3). This correlation is not seen for any other receptor, dopamine or otherwise. At the same time, the blockade of dopamine D
2 receptors is thought to be responsible for the antipsychotic-induced parkinsonian side effects
(4,
5) and prolactin elevation
(6). Over two dozen studies using positron emission tomography (PET)
(7–
12) and single photon emission computed tomography (SPECT)
(13,
14) with benzamide
(8,
11,
13,
14) and spiperone
(7,
9,
10) ligands have confirmed that all antipsychotics block dopamine D
2 receptors in patients, albeit to varying degrees. However, all these reports, with few exceptions
(10,
12), are based on cross-sectional or uncontrolled study designs, with open rating of clinical response and side effects and with high levels of D
2 occupancy. Thus, while there can be no doubt that antipsychotics block dopamine D
2 receptors, the quantitative relationship between in vivo blockade of dopamine D
2 receptors, clinical response, extrapyramidal side effects, and prolactin elevation is still unclear.
METHOD
The study was undertaken at the Clarke Division of the Centre for Addiction and Mental Health, Toronto. Eligible subjects, inpatients as well as outpatients, were included after they provided written consent using forms and procedures approved by the University of Toronto Human Subjects Review Committee. The study was restricted to patients between the ages of 18 and 50 who 1) met the DSM-IV criteria for schizophrenia or provisional schizophreniform disorder, 2) had never received antipsychotic treatment or had received antipsychotic treatment for less than 6 weeks and had been drug free for at least 2 weeks at inclusion, and 3) had no major medical or neurological illness or history of major head injury. Subjects were excluded if they had a lifetime history of substance dependence or a current diagnosis of substance abuse according to DSM-IV criteria. The diagnoses were established by using a structured interview of the patient by means of the Structured Clinical Interview for DSM-IV (SCID)
(15), supplemented with information from the family and the clinical staff.
The study was carried out in a double-blind fashion, and the patients were randomly assigned to doses of 1.0 or 2.5 mg of haloperidol per night. These doses were chosen on the basis of previous PET occupancy data
(16,
17) to give a wide range of dopamine D
2 occupancy without floor or ceiling effects. Clinical status was assessed by using the Clinical Global Impression Scale (CGI)
(18) for rating improvement, Positive and Negative Syndrome Scale
(19) for assessing clinical symptoms, Extrapyramidal Symptom Rating Scale
(20) for assessing drug-induced extrapyramidal side effects, and the Barnes Akathisia Rating Scale
(21) for assessing akathisia.
Only lorazepam and benztropine were allowed as adjunctive medications. The decisions regarding their use were made by the treating physicians on the basis of clinical considerations. The use of lorazepam (up to 6 mg/day) was indicated for daytime anxiety, agitation, akathisia, and insomnia. Benztropine (up to 6 mg/day) was used for extrapyramidal side effects and for akathisia.
After 2 weeks of continuous treatment, the patients each had a PET scan with [
11C]raclopride to determine their levels of D
2 occupancy. All scans were carried out 12 to 13 hours after the administration of the nightly dose of haloperidol. Clinical status and side effects were rated at this time by a rater blind to occupancy results, using the aforementioned scales. Patients who scored 2 or lower on the CGI improvement measure (i.e., were much improved or very much improved) were classified as responders, while those who scored 3 or higher (minimal improvement, no change, or worsening) were considered nonresponders. The responders had their doses held at the initial levels, while the nonresponders had their doses increased to 5.0 mg/day of haloperidol for the next 2 weeks. The purpose of this second stage of the experiment was to evaluate whether the nonresponders could be converted to responders when their doses were increased to give greater than 80% D
2 occupancy. The dose of 5.0 mg/day was chosen because previous data suggested that it would lead to greater than 80% dopamine D
2 occupancy for almost all patients
(16). The treatment was then continued, at the initial dose (for responders) or at the increased dose (for nonresponders), for a further period of 2 weeks. Ratings on the clinical scales were repeated at the end of this phase.
D
2 receptor occupancy was determined by using 9.7 mCi (SD=0.5) of high-specific-activity (970 Ci/mmol, SD=420) [
11C]raclopride administered as a bolus plus continuous infusion. Imaging was performed with a GE-2048-15B head scanner as described in previous publications
(8,
22). A magnetic resonance imaging (MRI) scan was obtained on each of the patients (GE Signa 1.5-T scanner, proton density maps) and was coregistered to the composite [
11C]raclopride PET scan by using automatic image registration
(23,
24) software, provided by Dr. Roger Woods, University of California, Los Angeles. Since the PET scan has a more limited axial field of view than the MRI scan and the two techniques differ in their resolution and interslice intervals, the coregistration usually resulted in a slight misalignment. Therefore, rather than draw regions of interest on the MRI scan, we drew the striatal (caudate plus putamen) and cerebellar regions of interest on two contiguous PET slices with reference to an overlapping coregistered MRI scan. The cerebellar time-activity curve was taken as an estimate of the free and nonspecific [
11C]raclopride binding
(25), while the striatal time-activity curve provided an estimate of specific binding to the D
2 receptors plus free and nonspecific binding. Under these assumptions, it can be shown that the striatal-cerebellar ratio minus one, at the time when the binding is at equilibrium (30–75 minutes in the aforementioned scans), provides an index proportional to the B
max/K
d ratio of [
11C]raclopride for dopamine D
2 receptors (referred to as the binding potential). In previous studies
(22) we have demonstrated that this ratio method correlates very well (r>0.95) with analytically derived estimates of D
2 binding potential, is highly reliable with a scan-rescan standard deviation of 6%, and has been standardized in our laboratory with interrater and intrarater reliability (intraclass correlation coefficients: r>0.95). There was no significant hemispheric asymmetry in the D
2 binding potential; therefore, data from the left and right striatum were pooled for all subsequent calculations. Since we did not have baseline measures of D
2 binding potential for these patients, we used an age-corrected estimate from a separate group of 12 antipsychotic-naive patients with schizophrenia and 15 age-matched healthy subjects who functioned as a comparison group. The pooling of patients and healthy subjects was justified as there was no significant difference in D
2 binding potential between groups (F=0.36, df=1, 24, p=0.55), nor was there a significant age-by-illness interaction (F=0.58, df=1, 24, p=0.45), findings consistent with those from previous studies that examined D
2 binding potential with raclopride in patients with schizophrenia
(26,
27).
The haloperidol levels were determined by using a gas chromatograph/mass spectrometer as described previously
(16). Prolactin levels were determined by using a two-site chemoluminometric immunoassay with a minimum detectable limit of 0.3 ng/ml and a coefficient of variance of 4.5%.
All subjects who completed 2 weeks of treatment and for whom PET data were available were included in the analysis. Since the clinical decision hinged on the CGI rating, that was taken as the primary response variable and the scores on the Positive and Negative Syndrome Scale subscales were used to provide a more detailed profile of clinical response. Four subjects developed extrapyramidal side effects or akathisia in the first 2 weeks, and adjunctive treatment was instituted for three of these four before the PET scan. As a result of this concomitant treatment, the severity of extrapyramidal side effects at the time of the PET scan was probably underestimated in these cases. For this reason, for the purposes of this analysis, extrapyramidal side effects and akathisia were rated only as present or absent, without an effort to relate the quantitative degree of extrapyramidal side effects to the degree of D2 receptor occupancy. Hyperprolactinemia for the purposes of this study was defined by reference to the normal ranges established for this assay, which are 2.1–17.7 ng/ml for men and 2.8–29.2 ng/ml for nonpregnant women. The statistical analyses were carried out in Statistica for Windows (Stat Soft, Inc., Tulsa, Okla.).
RESULTS
Of the 26 patients who entered the study, three did not complete 2 weeks: two (receiving 1.0 and 2.5 mg/day) because of clinical worsening and the third (2.5 mg/day) because of notable extrapyramidal side effects. PET data could not be obtained for one patient because of equipment failure. Of the remaining 22, five were women and 17 were men; 19 were antipsychotic naive. Their median age was 30 years (range=19–45), and they had a median duration of psychotic symptoms of 52 weeks (range=4–260 weeks). Eighteen of these patients met the criteria for schizophrenia, and four met criteria for provisional schizophreniform disorder at the time of inclusion in the study. Of those initially diagnosed with schizophreniform disorder, three were finally diagnosed with schizophrenia, and one received the diagnosis of delusional disorder.
The patients’ illnesses were of moderate to marked severity according to the CGI severity scale (mean score=4.5, SD=0.6), and positive symptoms predominated (Positive and Negative Syndrome Scale positive syndrome score: mean=20.9, SD=3.8) over negative symptoms (negative syndrome score: mean=15.9, SD=6.5). Thirteen of these patients received 1.0 mg/day of haloperidol, and nine received 2.5 mg/day. The D2 occupancy varied from 38% to 87% and was significantly related to dose (1.0-mg/day group: mean=59%, SD=11%; 2.5-mg/day group: mean=75%, SD=6%) (F=15.9, df=1, 20, p<0.001). Occupancy was highly correlated with plasma haloperidol level (F=15.9, df=1, 19, p<0.001) but was not related to any other baseline variable (age, sex, or severity of illness at baseline). As adjunctive medication four patients received lorazepam, and two others received benztropine. Ten patients were considered responders: three (23%) of the 13 who received 1.0 mg/day and seven (78%) of the nine who received 2.5 mg/day. Twelve patients were judged to be nonresponders: 10 receiving 1.0 mg/day and two receiving 2.5 mg/day. The responders showed a greater improvement in positive symptoms; they had a decrease in the Positive and Negative Syndrome Scale positive syndrome score of 8.5 (SD=4.2), as compared to 1.3 (SD=2.4) for the nonresponders (F=24.2, df=1, 20, p<0.0001). On the other hand, the overall improvement in negative symptoms was low regardless of responder status. The negative syndrome score improved by 2.2 (SD=4.4) for the responders and 1.6 (SD=1.3) for the nonresponders (F=0.2, df=1, 20, p=0.64).
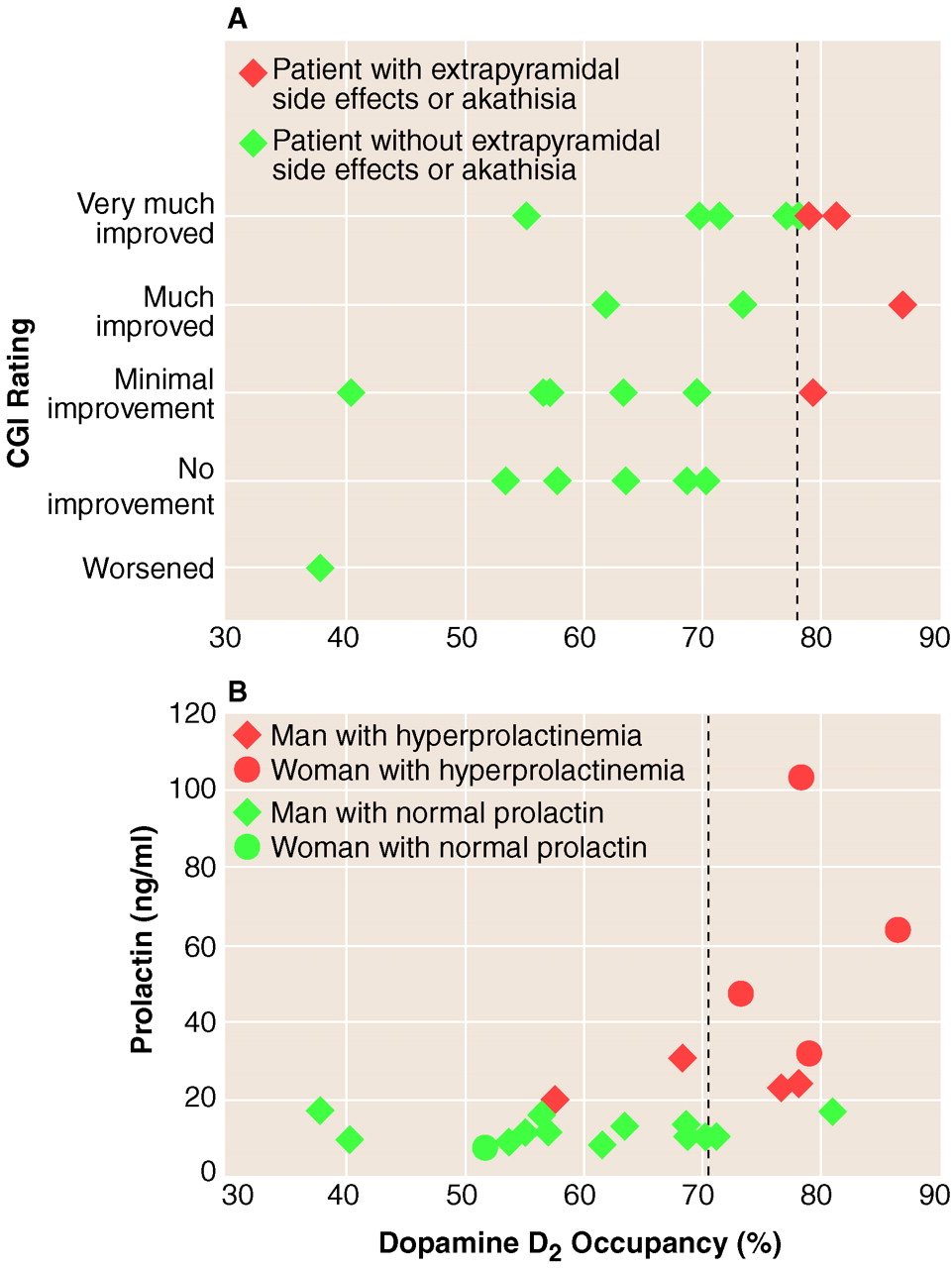
The responders showed significantly higher D
2 occupancy (mean=73%, SD=9%) than the nonresponders (mean=60%, SD=12%) (F=8.3, df=1, 20, p<0.009) (
figure 1A). The D
2 occupancy significantly predicted the degree of clinical response as measured with the CGI (F=9.3, df=1, 20, p=0.006) (
figure 1A). The relationship between D
2 occupancy and improvement in CGI rating was equally maintained in a multiple regression model that included baseline severity as a covariate (omnibus F=4.4, df=2, 19, p=0.02); the relationship between baseline severity and improvement was nonsignificant (t=0.02, df=19, p=0.98), and the relationship between D
2 occupancy and improvement was highly significant (t=2.86, df=19, p=0.009). However, the relationships of D
2 occupancy to improvement in the scores on the Positive and Negative Syndrome Scale positive syndrome scale (F=2.8, df=1, 20, beta=0.35, p=0.10) and negative syndrome scale (F=0.0, df=1, 20, beta=0.11, p=0.90) did not achieve significance.
We were interested in determining whether a threshold for clinical response existed in the data. Ideally, a threshold would be a level of D2 occupancy such that there are no responders below it and everyone above that level obtains equivalent response. We did not find such an absolute threshold. However, a cutoff at 65% D2 occupancy provided optimal separation: 80% of the responders were above it while 67% of the nonresponders were below it (p=0.04, Fisher’s exact test). Furthermore, when we restricted the analysis to subjects who were above this threshold of 65%, we did not find a relationship between clinical response and D2 occupancy (multiple regression, baseline severity as measured with the CGI and D2 occupancy as independent variables; omnibus F=0.9, df=2, 9, p=0.45; test for the effect of D2 occupancy: t=1.20, df=9, p=0.25).
Of the 10 responders, nine had maintained or improved responses over the next 2 weeks, while one patient (with 62% D2 occupancy) showed a decline in response. Of the 12 nonresponders, whose doses were increased to 5.0 mg/day, one had an acute dystonic reaction after the first dose of 5.0 mg of haloperidol and left the study. Of the remaining 11, seven achieved responder status while four continued to be nonresponders. Six of the seven patients who had occupancies below 65% before the dose increase showed improvement, while only one of the four who had occupancies above 65% showed improvement.
Only three of the 23 patients who completed 2 weeks showed extrapyramidal side effects at 2 weeks. Patients who showed drug-induced extrapyramidal side effects had significantly higher D
2 occupancies (mean=80%, SD=0%, N=3) than patients without extrapyramidal side effects (mean=64%, SD=12%, N=20) (Mann-Whitney U Test: z=2.4, exact p=0.009). For clinical considerations, benztropine was prescribed for two of these three patients, and lorazepam was prescribed for one. Four patients showed symptoms of akathisia; three of them were the same ones who experienced the extrapyramidal side effects just noted. The patients with akathisia as a group showed higher occupancies (mean=81%, SD=4%, N=4) than those without (mean=63%, SD=12%, N=19) (Mann-Whitney U Test: z=3.1, exact p=0.0002). None of the patients with D
2 occupancies below 78% showed extrapyramidal side effects or akathisia (
figure 1A).
Among the nonresponders whose doses were increased to 5.0 mg/day, there were several new cases of extrapyramidal side effects. One patient developed an acute dystonic reaction, two developed extrapyramidal side effects, two developed akathisia, and another two patients exhibited both extrapyramidal side effects and akathisia at the 4-week mark. Thus, seven of the 12 patients who received 5.0 mg/day of haloperidol manifested either extrapyramidal side effects or akathisia, as compared to only three of the 23 patients who were treated with 1.0 or 2.5 mg/day (p=0.008, Fisher’s exact test).
Finally, D
2 occupancy significantly predicted an abnormally high prolactin level at the time of PET scanning (F=7.3, df=1, 20, p<0.01). The likelihood of hyperprolactinemia was 15% with a D
2 occupancy below 72%, and the likelihood was 86% at occupancies higher than 72% (p=0.002, Fisher’s exact test) (
figure 1B).
DISCUSSION
The results of our study show that striatal D2 occupancy, as induced by haloperidol, predicts antipsychotic response, drug-induced extrapyramidal side effects, akathisia, and prolactin elevation. There is also evidence for a stepped increase in response beyond 65% D2 occupancy; prolactin elevation became prominent beyond 72%, while extrapyramidal side effects and akathisia were evident beyond 78%.
To our knowledge, only one previous study has systematically explored the relationship between D
2 occupancy and clinical response. Nordstrom et al.
(12) studied 12 patients with schizophrenia treated with an experimental antipsychotic, raclopride, and reported a significant relationship between D
2 occupancy and clinical response. Our data confirm this initial report in much larger study group and extend its applicability to a clinically relevant typical antipsychotic. Since 65%–70% D
2 occupancy was obtained with 2.5 mg/day of haloperidol in a majority of the patients, our data suggest that a dose in the range of 2–3 mg/day should be the optimal starting dose for first-episode patients. This dose contrasts to those in previous clinical studies
(28,
29), which started treatment at haloperidol doses in the range of 10–20 mg/day for first-episode patients. Such doses would lead to occupancies beyond the cutoff for extrapyramidal side effects discerned in this study, and in keeping with that, 78%–85% of the patients in those high-dose studies experienced extrapyramidal side effects
(28μ30). To our knowledge, only two controlled studies of first-episode patients have tested lower doses
(31,
32). These studies indicated that 1–4 mg/day
(31) or 2–5 mg/day
(32) of haloperidol is the optimal dose for patients with a first episode of schizophrenia. Higher doses in both studies were associated with a higher incidence of extrapyramidal side effects without a significant improvement in clinical response
(30–
32).
While previous cross-sectional neuroimaging studies have suggested a relationship between D
2 occupancy and extrapyramidal side effects
(11,
12,
14,
33), our study not only confirms this relationship with a prospective design but provides evidence that clinical extrapyramidal side effects emerge only beyond 78% D
2 occupancy. This situation is similar to idiopathic Parkinson’s disease, in which a significant loss of dopaminergic transmission occurs before clinical symptoms become evident
(34,
35). Our data also confirm previous associations between D
2 occupancy and prolactin elevation
(7,
36) and extend them by showing that a high level of blockade (approximately 72%) is necessary before prolactin elevation becomes manifest. The extrapyramidal side effects and hyperprolactinemia findings suggest that there is a functional reserve in the D
2-mediated systems. As a result, these side effects are not a linear function of receptor blockade but become evident only after a high proportion of receptors have been blocked.
The aim of an optimal treatment strategy is to achieve the highest rate of response with the lowest incidence of side effects. The therapeutic window suggested by our data is rather narrow: from 65% (for response) to 72% (when side effects emerge). This window corresponds to less than 0.5 mg/day of haloperidol for a given patient. Given this narrow therapeutic window and the wide interindividual variation in D
2 occupancy at a given dose
(8,
16), one cannot recommend a single dose of haloperidol that will get most patients above 65% without pushing them into the occupancies associated with side effects. This explains why clinically it has been difficult to obtain clinical response without a high incidence of side effects in the use of haloperidol-like agents.
Several limitations qualify the preceding results. First, mesolimbic D
2 receptors are thought to be more relevant to the induction of clinical response
(37), and pituitary D
2 receptors mediate prolactin levels
(6). However, we could not measure D
2 occupancy in these regions as extrastriatal regions cannot be reliably quantified with [
11C]raclopride and PET. Two studies
(38,
39) have shown no significant difference between striatal and extrastriatal D
2 occupancy in patients treated with haloperidol. Therefore, striatal occupancy is a reasonable surrogate of mesolimbic D
2 occupancy, at least in the case of haloperidol. Second, this study is limited in that patients were given only 2 weeks to respond at the lower doses. This duration reflects a compromise. To increase the validity of this study we wanted to expose patients to low D
2 occupancies, lower than what had been tested before. On the other hand, there was concern that low doses/occupancies may not be optimally antipsychotic, and therefore we wanted to keep the duration of such exposure short. The short duration increases the risk of missing a relationship because of limited power. Since we found significant positive relationships in this study, this is less of a concern. However, the question of whether patients with lower D
2 occupancies would have responded if given a longer period of time still remains.
In addition to the aforementioned limitations, our study does not address several important questions. Clinical studies suggest that patients who have been previously treated may need higher doses than first-episode patients
(31). It is unclear why this is the case. Whether the chronic patients achieve lower occupancies for a given drug/plasma level because of better drug disposition or up-regulation of receptors, or whether chronic treatment alters the basic relationship between occupancy and clinical correlates, is a question for the future. Finally, our data apply mainly to antipsychotics whose predominant mode of action is D
2 blockade. Several newer, “atypical” antipsychotics (e.g., clozapine, risperidone, olanzapine) show a higher affinity for the serotonin, adrenergic, and histaminergic receptors than for D
2 receptors
(40), and these additional properties may enhance their efficacy
(41,
42) or diminish the side effects induced by D
2 blockade
(43–
45). Thus, the relationship of D
2 blockade to clinical response and side effects in this study may be different with these newer medications
(14,
46–
48), a hypothesis that has yet to be directly tested. Most of the current studies of typical versus atypical antipsychotics have used doses of haloperidol (10–20 mg/day) that give inordinately high levels of D
2 occupancy and have compared these doses to doses of the atypical agent that give 60%–80% D
2 occupancy. These studies have routinely shown more extrapyramidal side effects with haloperidol than with the atypical agents, but it is unclear whether this difference reflects the different levels of D
2 occupancy induced by the drugs or some other fundamental difference between the typical and atypical agents
(48).
The more interesting question that this relationship raises is one of causality and mechanism. How does D2 blockade mediate response? At this stage one can offer only logical speculations. While D2 blockade is essential, it does not temporally correspond with clinical response. The D2 occupancy is achieved within hours after the start of treatment, while response takes days. Conversely, when the drug is stopped, the occupancy declines in days while the rate of relapse has a half-life of many months. This suggests that D2 blockade induces response by invoking downstream changes, which are delayed but enduring. And these alterations lead to the physiological changes that are the ultimate mediator of clinical improvement. These downstream changes are neurochemically nonspecific (e.g., the changes in cAMP, immediate early genes, and glucose metabolism, etc., that can also be observed with other modulators) but are distinguished by their anatomically specificity (happen only in circuits linked to cells with D2 receptors). Furthermore, these intracellular and gene expression changes are associated with changes in D2 occupancy, as opposed to sustained high D2 occupancy, i.e., they are triggered by D2 occupancy rather than being maintained by continuous occupancy. Thus, we believe that blockade of D2 receptors targets the appropriate anatomical circuits and triggers within them intracellular changes that lead to clinical improvement. The precise circuits and the crucial intracellular changes are a matter of active research, but the target-and-trigger conception of the role of D2 occupancy helps in narrowing the possible candidates.
In summary, we find that dopamine D2 occupancy predicts short-term clinical response, extrapyramidal side effects, and hyperprolactinemia. The likelihood of clinical response increases as D2 occupancies exceed 65%–70%, while the risks of hyperprolactinemia and extrapyramidal side effects or akathisia increase at occupancies higher than 72% and 78%, respectively. Therefore, low doses of typical antipsychotics, in the range of 1–3 mg/day of haloperidol, should be used as initial doses for first-episode patients.