Discussion
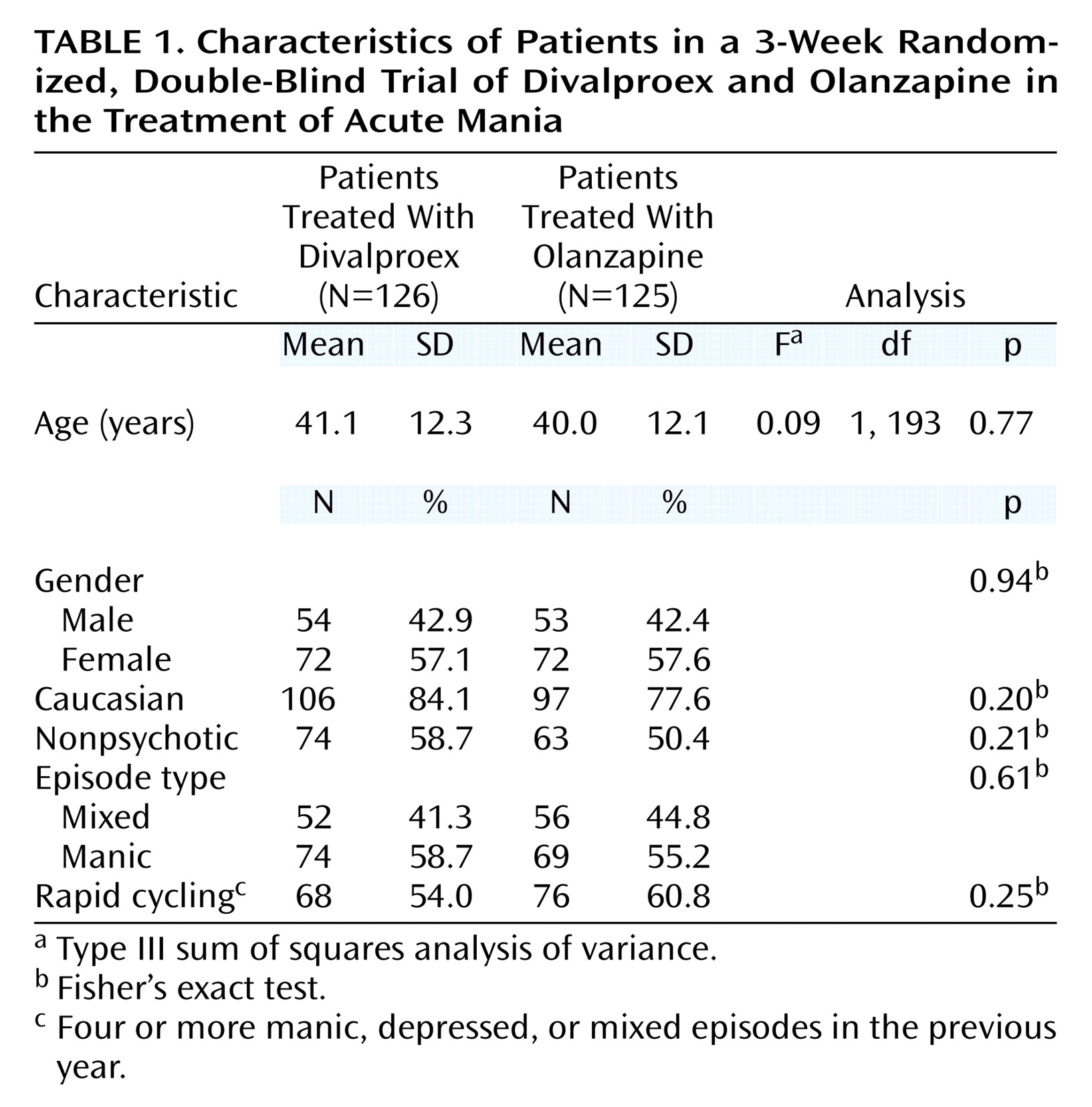
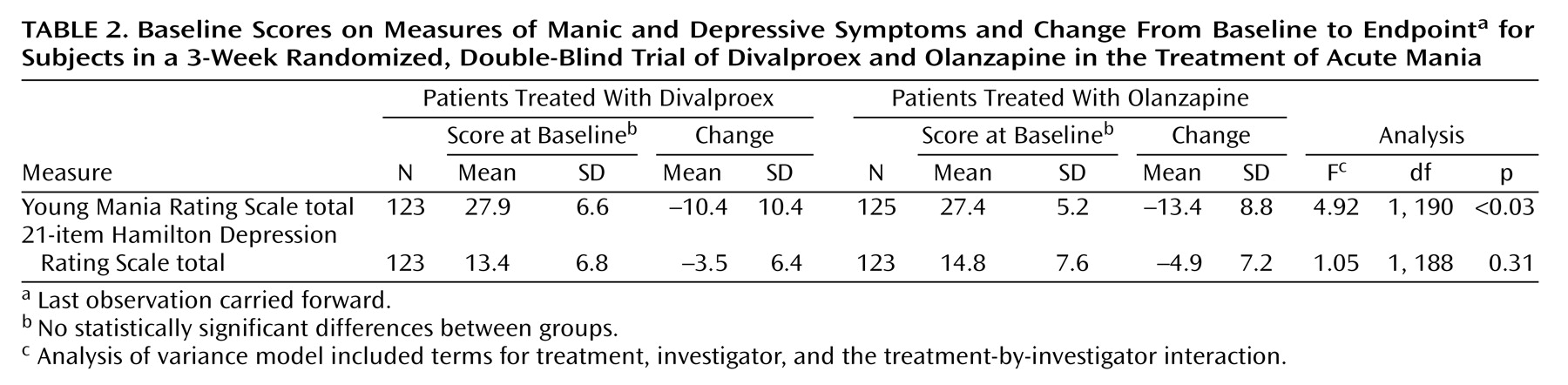
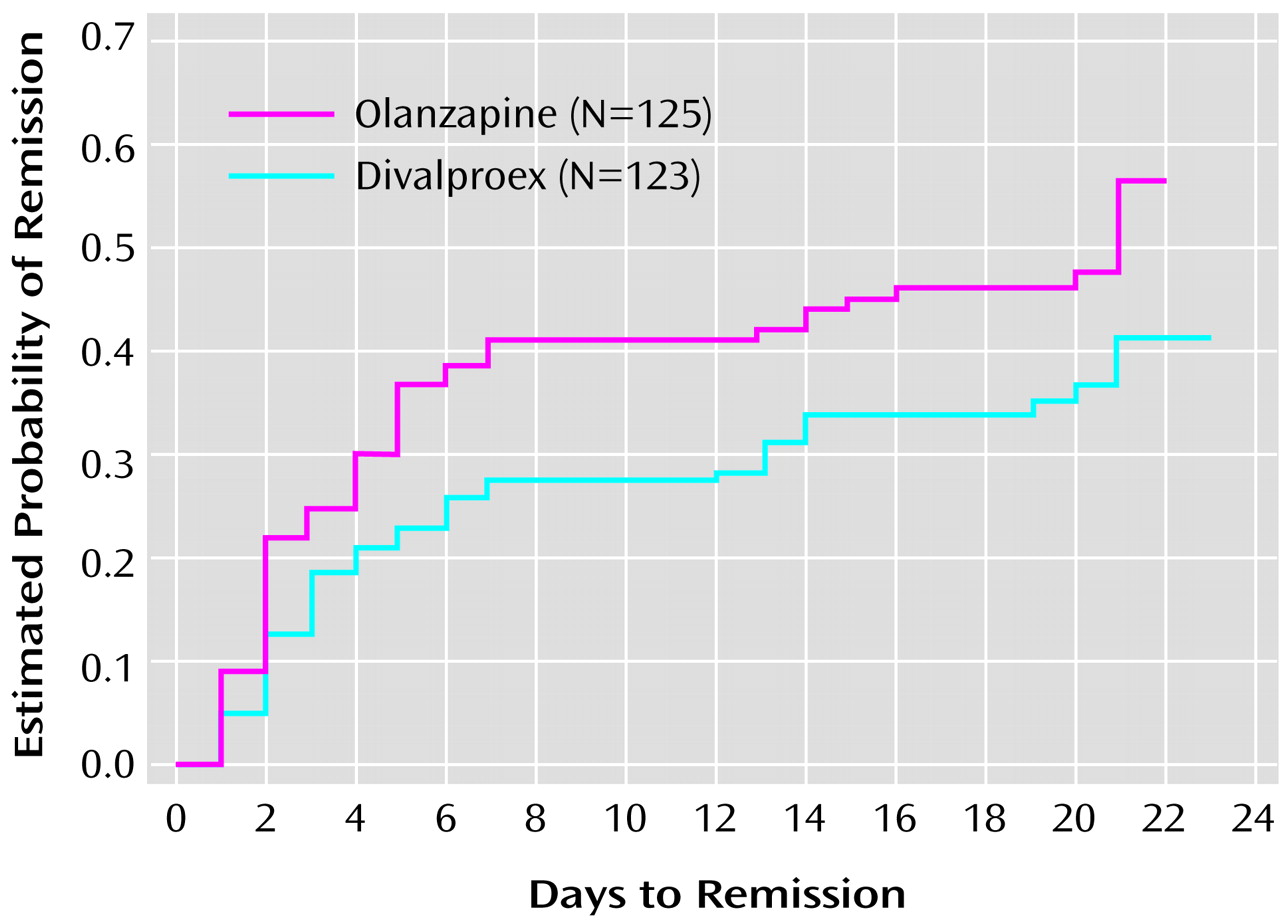
To our knowledge, this study is the largest double-blind comparison of divalproex and olanzapine in the treatment of acute mania. Olanzapine-treated patients improved significantly more on the primary efficacy variable (Young Mania Rating Scale total score).
In a recent 4-week, placebo-controlled trial that used 15 mg/day of olanzapine as the starting dose
(6), the mean reduction in the Young Mania Rating Scale score at week 3 (last observation carried forward) was 13.9 points, comparable to the 13.4-point reduction for olanzapine-treated patients in the current trial. Few available data address whether the 10.4-point mean improvement in the divalproex group is in line with expectations. A placebo-controlled valproate trial reported by Pope et al.
(4) used the Young Mania Rating Scale as the primary outcome variable, but the results may not be directly comparable to those of the current trial because the Pope et al. study was a smaller, single-center trial that enrolled lithium-refractory and/or intolerant patients. In that 3-week study, 17 valproate-treated patients had a mean improvement on the Young Mania Rating Scale of 11.4 points, slightly higher than that observed in the current study.
It is noteworthy that the reported average divalproex plasma levels in a study by Bowden et al.
(3) were 77 μg/ml at day 8 and 93 μg/ml at day 21, compared to 82 μg/ml at day 7 and 84 μg/ml at day 21 in the current trial. The slightly higher mean levels at endpoint in the Bowden et al. trial may reflect differences in methods; in that study, six upward titrations of divalproex were mandated, unless the subject’s blood level was more than 150 μg/ml or contravening side effects developed. In the study by Pope et al.
(4), blood levels of valproate were not reported. However, the authors indicated that most of the clinical improvement was observed within 1–4 days of achieving serum levels of at least 50 mg/liter.
A report recently presented by Zajecka et al.
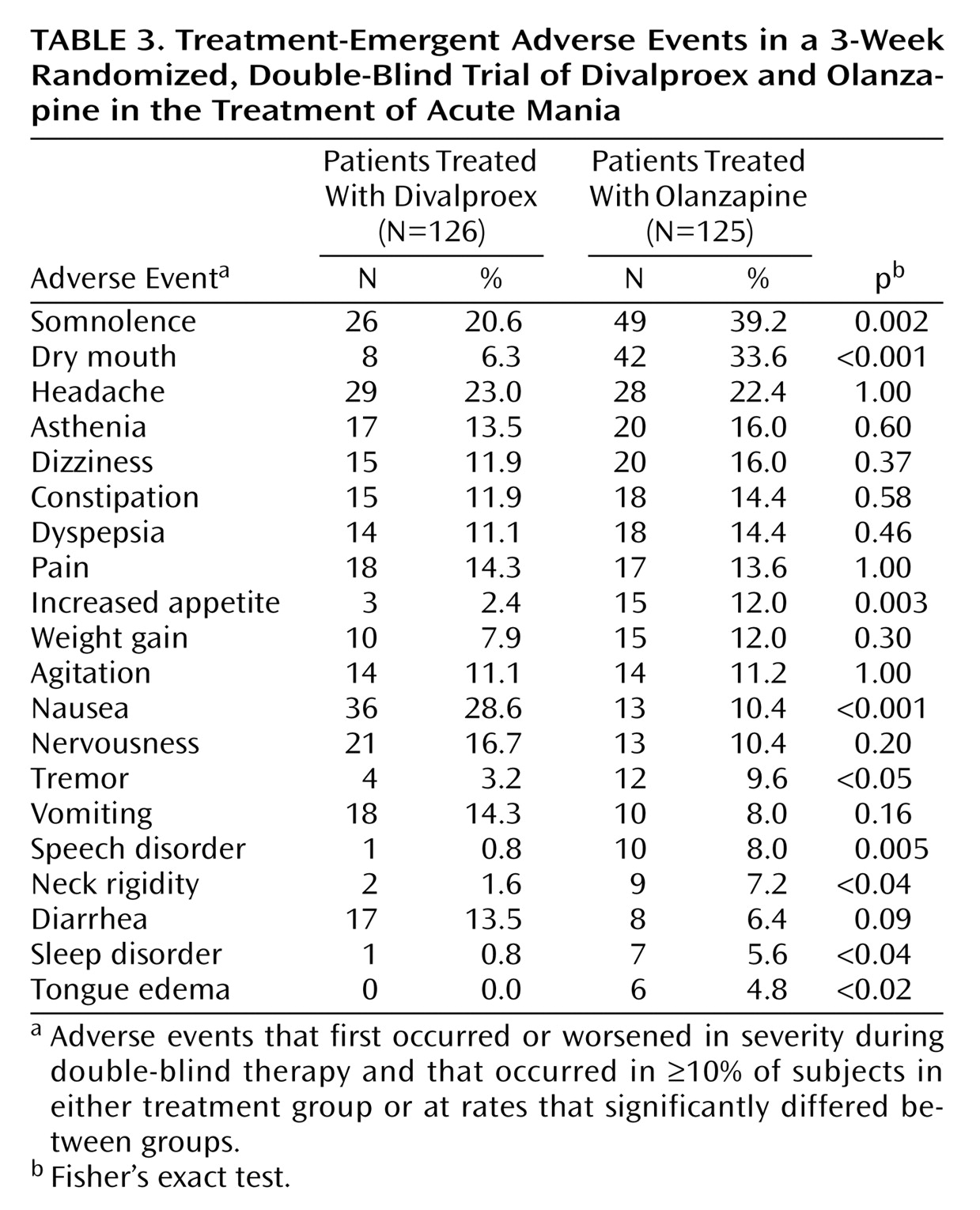
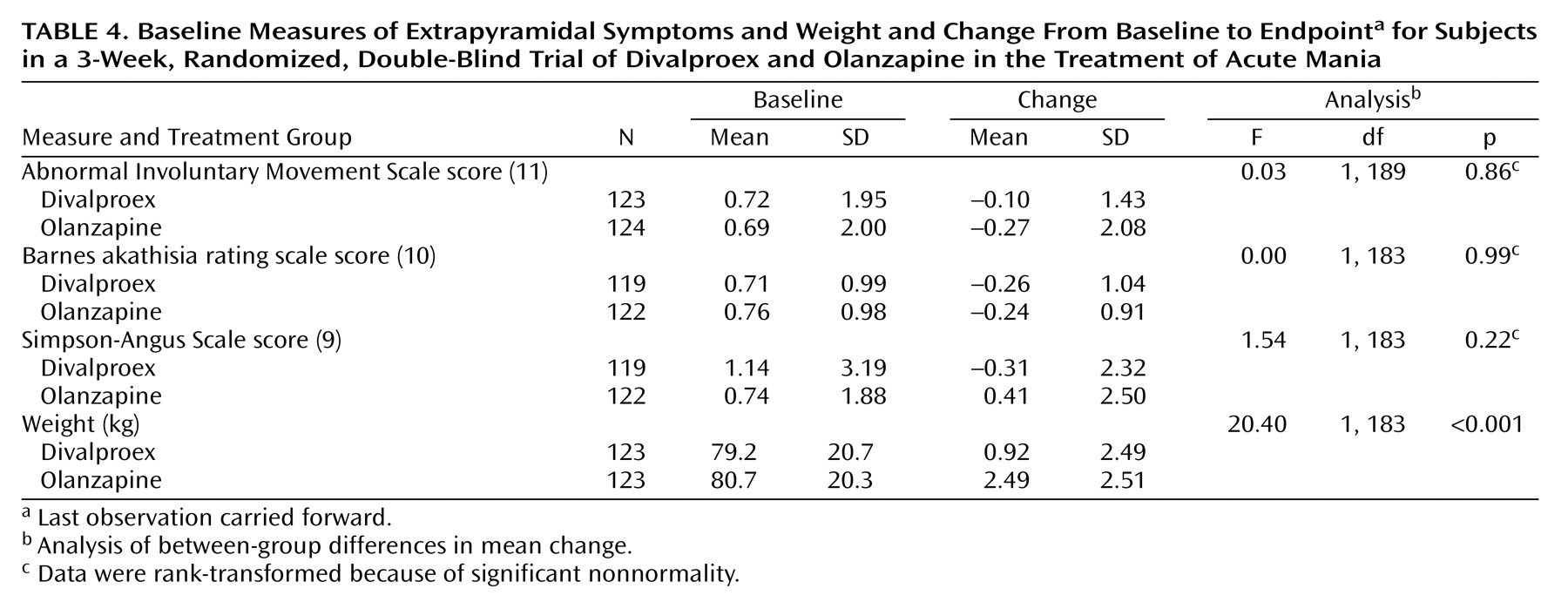
(16) described another study comparing divalproex sodium (N=63) to olanzapine (N=57) in the treatment of acute mania. Zajecka and collaborators used a starting loading dose of 20 mg/kg per day for divalproex sodium (mean maximum dose=2115 mg/day) and 10 mg/day for olanzapine (mean maximum dose=14.7 mg/day). As in the present study, more improvement in the Young Mania Rating Scale was observed during olanzapine treatment than during divalproex treatment. However, unlike our findings, the difference observed by Zajecka et al. was not statistically significant. This difference may be explained by the smaller study group size of 120 patients, compared to 251 in the present study. The results indicated a reduction of 17.2 points for olanzapine and 14.8 for divalproex (p=0.21) on the Young Mania Rating Scale from baseline to 21 days of treatment. Dropout rates due to adverse events were similar between treatments, and more individual adverse event terms—including somnolence, weight gain, rhinitis, edema, and speech disorder—were observed significantly more often during treatment with olanzapine.
In the current study, olanzapine produced greater improvement, compared to divalproex, as measured by the change in score on the Young Mania Rating Scale at days 2, 14, and 21. It is possible that the brief early separation reflects slower achievement of therapeutic dosing for divalproex than for olanzapine. Divalproex dosing was titrated in accordance with the manufacturer’s recommendation, starting at 750 mg on day 1 and was thereafter adjusted on the basis of clinical response and serum levels. Improvement in the early stages of divalproex therapy appears more likely if the serum level has reached at least 45 μg/ml
(17). Therefore, standard dose initiation at 750 mg/day potentially achieves efficacy less promptly than divalproex “loading” strategies, with first day doses of 20–30 mg/kg. Several open studies have supported the feasibility and utility of loading
(18–
21), and one double-blind trial focusing on safety and tolerability found more efficacy for loaded than for standard titration after 3 days of treatment
(22). Based on the findings of Hirschfeld et al.
(22), olanzapine’s transient separation during week 1 may have been less likely if divalproex had been loaded.
It is interesting to note that olanzapine’s antimanic advantage in this 3-week study cannot be solely ascribed to treatment of psychotic symptoms. Without correcting for multiple comparisons, the treatments separated on only three of the 11 items of the Young Mania Rating Scale, with olanzapine superior on sleep, increased motor activity, and language–thought disorder items. Furthermore, Young Mania Rating Scale improvement among these psychotic patients was similar between the two treatments, and olanzapine treatment produced significantly greater improvement for the manic patients without psychotic features. A treatment-by-subgroup interaction in Young Mania Rating Scale improvement was observed when patients were categorized on the basis of presence or absence of psychotic features. Among olanzapine-treated patients, mean Young Mania Rating Scale improvement was similar in the psychotic and nonpsychotic subgroups (mean change of –12.6 and –14.1 points, respectively). Among divalproex-treated patients, Young Mania Rating Scale score reduction was greater in the psychotic subgroup (–12.8 points) than in the nonpsychotic subgroup (–8.7 points). To our knowledge, previous controlled trials of divalproex have not reported whether antimanic efficacy varied according to the presence or absence of psychotic features.
Acknowledgments
The authors acknowledge the following institutions and individuals who participated in this clinical trial: Jay Amsterdam, M.D., University of Pennsylvania, Philadelphia; George Bartzokis, M.D., Little Rock Veterans Affairs Medical Center, North Little Rock, Ark.; Louise Dabiri-Beckett, M.D., IPS Research Company, Oklahoma City; Gary Booker, M.D., IPS Research Company, Shreveport, La.; Ron Brenner, M.D., St. John’s Episcopal Hospital, Far Rockaway, N.Y.; David Brown, M.D., Community Clinical Research, Inc., Austin, Tex.; Timothy Byrd, M.D., Charter Springs Behavioral Clinic, Ocala, Fla.; Franca Centorrino, M.D., McLean Hospital, Belmont, Mass.; James Chou, M.D., Bellevue Hospital, New York; Anthony Claxton, M.D., and Hermant Patel, M.D., University of Oklahoma Health Sciences Center, Oklahoma City; Lori Davis, M.D., Veterans Affairs Medical Center, Tuscallosa, Ala.; Jose E. De La Gandara, M.D., North Broward Neurological Institute Memory Disorder Center, Pompano Beach, Fla.; G. Michael Dempsey, M.D., Memorial Hospital, Albuquerque, N.M.; Rif El-Mallakh, M.D., University of Louisville School of Medicine, Louisville, Ky.; Louis Fabre, M.D., Fabre Research Clinics, Houston; David Feifel, M.D., University of California at San Diego Medical Center, San Diego; Brent Forester, M.D., Mental Health Center of Greater Manchester, Manchester, N.H.; Arthur Freeman III, M.D., Louisiana State University Medical Center, Shreveport, La.; Mark Frye, M.D., UCLA Neuropsychiatric Institute and Hospital, Los Angeles; Alan Green, M.D., Massachusetts Mental Health Center, Boston; Sanjay Gupta, M.D., Psychiatric Network, P.C., Olean, N.Y.; Robert Horne, M.D., Lake Meade Hospital, Las Vegas; Fuad Issa, M.D., Clinical Research Center of Northern Virginia, Falls Church, Va.; Robert Jamieson, M.D., Psychiatric Consultants, P.C., Nashville, Tenn.; Christopher Kelsey, M.D., San Diego Center for Research, San Diego; Louis Ari Kopolow, M.D., Associated Psychotherapy Centers, Gaithersburg, Md.; Jeff Mitchell, M.D., Laureate Psychiatric Clinic and Hospital, Tulsa, Okla.; Cesar Munoz, M.D., University of Alabama at Birmingham, Birmingham, Ala.; Fred Petty, M.D., Ph.D., Veterans Affairs Medical Center, Dallas; Michael, Plopper, M.D., Mesa Vista Hospital, San Diego; Joanchim Raese, M.D., Knollwood Psychiatric and Chemical Dependency Center of Charter Hospital, Murietta, Calif.; Jeffery Rausch, M.D., Medical College of Georgia, Augusta, Ga.; Robert Riesenberg, M.D., Atlanta Center for Medical Research, Decatur, Ga.; Leon Rubenfaer, M.D., Harbor Oaks Hospital, Farmington Hills, Mich,; John Schmitz, M.D., Midwest Psychiatric Consultants, P.C., Kansas City, Mo.; Taylor Segraves, M.D., Ph.D., MetroHealth Medical Center, Cleveland; Philip Seibel, M.D., Contemporary Behavioral Research, L.L.C., Washington, D.C.; G. Michael Shehi, M.D., Mountain View Hospital, Gladsden, Ala.; Marshall Thomas, M.D., University of Colorado Health Sciences Center, Denver; Kathleen Toups, M.D., Bay Area Research Institute, Lafayette, Calif.; Dan Wilson, M.D., Pauline Warfield Lewis Center, Cincinnati; Tai P. Yoo, M.D., Mercy Hospital, Detroit; and John Zajecka, M.D., Rush Presbyterian St. Lukes, Chicago.