Individuals with attention deficit hyperactivity disorder (ADHD) respond to reinforcements, such as rewards or punishments, differently than healthy comparison subjects (for review, see reference
1). Some reports have shown that children with ADHD require stronger and more salient reinforcers to regulate their behavior than do healthy children, that they are less sensitive to changes in reinforcement contingencies
(2), and that negative reinforcers are more effective than rewards in enhancing their cognitive performance
(3–
6). Other reports on the effects of reinforcement contingencies
(7) and on the effectiveness of punishment
(8) have not supported these findings.
Despite inconsistencies in findings from laboratory studies of reward processes in ADHD (for review see reference
9), the first lines of treatment for this disorder directly affect reward-related systems
(10). The behavioral approach is based on increasing the frequency and immediacy of consequences to behavior, and stimulant treatment enhances dopaminergic activity, a key neurochemical substrate of reward systems. The reward-related nature of these treatments constitutes strong evidence for a role of reward processes in ADHD and supports the reward-dysfunction hypothesis as a means for understanding the etiology and treatment of ADHD.
A number of functional neuroimaging studies have examined different aspects of reward-related processes in healthy adults
(11–
15). Their findings, together with those from electrophysiological research in animals
(16,
17), support the involvement of the ventral prefrontal cortex, dorsolateral prefrontal cortex, anterior cingulate, insula, amygdala, hippocampus, and ventral striatum.
Method
Subjects
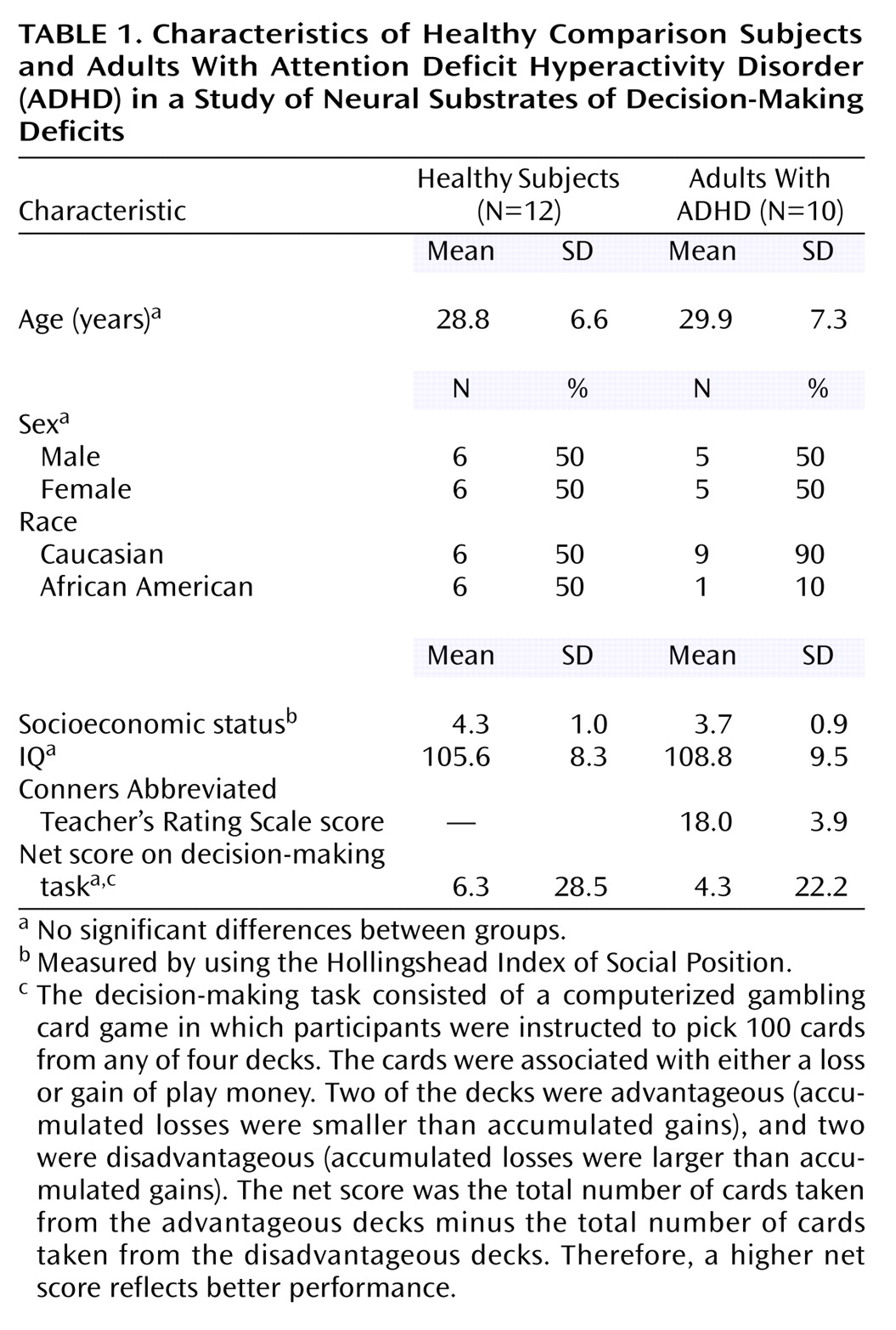
Men and women were recruited through newspaper advertisements. All participants gave written informed consent after receiving an explanation of the study. Inclusion criteria were age between 21 and 45 years, IQ >80 (Shipley Institute of Living Scale
[19]), and right-handedness. Exclusion criteria were current psychopathology (as assessed with the SCL-90
[20]), history of psychiatric disorders (as assessed with the NIMH Diagnostic Interview Schedule
[21]) except for childhood-onset ADHD, and evidence of acute or chronic medical problems (as indicated by means of medical history, physical examination, and routine blood screening). All patients fulfilled the DSM-IV criteria for ADHD in childhood (as assessed retrospectively with the NIMH Diagnostic Interview Schedule) and currently in adulthood (as assessed with the NIMH Diagnostic Interview Schedule), fulfilled the Utah Criteria for ADHD in adults
(22), and had a score >12 on the 10-item Conners Abbreviated Teacher’s Rating Scale
(23). Patients treated with stimulants were asked to discontinue treatment for a minimum of 48 hours before scanning.
Study Design
Positron emission tomography (PET) sessions included six 1-minute scans at 12-minute intervals, each following the intravenous injection of 10 mCi of [15O]H2O to assess regional cerebral blood flow (rCBF). Before the PET study, subjects were familiarized with the task in a 1-minute practice. During each scan, participants performed the decision-making task (active task), a control task, and a visual fixation task. The fixation task, used for quality control, was not part of the analysis. Each task was performed twice, and the order of tasks was counterbalanced across subjects.
Decision-Making Task
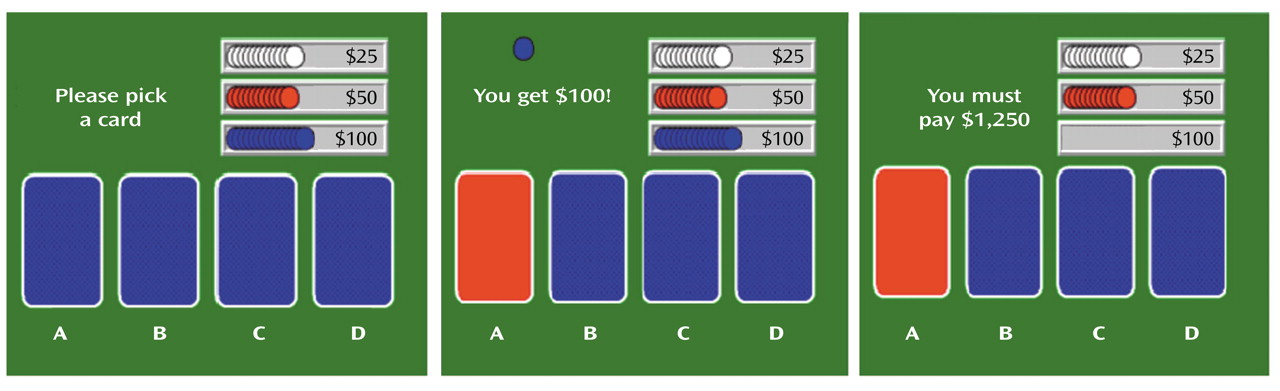
The decision-making task (
Figure 1) is a computerized gambling card game that tests the ability to choose between high gains with a risk for even higher losses and low gain with a risk for smaller losses. It was designed to assess patients who have ventromedial prefrontal lesions and exhibit poor decision making
(18). Participants were instructed to accumulate as much (play) money as possible by picking one card at a time from each of four decks (A, B, C, D) until they were told to stop (after selection of the 100th card). They were also told that some decks were better than others and that they would receive one cent for each dollar accumulated (maximum possible $20). Cards could be selected from the decks in any order. Once a card was selected, the message “You get $50” (or $100) immediately appeared on the screen and remained for 1.5 seconds until the subject was prompted to play again by the message, “Pick a card.” If a loss was also attached to that choice, a message lasting 1.5 seconds was added on the screen (e.g., “You must pay $75”). At the end of the 1.5 seconds, the subject was prompted to make the next selection. Each outcome was associated with a sound that was positive for the gains and negative for the losses. The cumulative amount of money was updated on the screen after each card pick. The task was initiated 40 seconds before the injection of the tracer (i.e., an average of 85 seconds before scanning, which started when the tracer entered the brain [∼45 seconds after injection]) to ensure that subjects had selected at least 20 cards before data collection. Scanning lasted 1 minute, during the selection of the next 20 cards. Subjects continued to play until 100 cards were selected.
The decks differed along two dimensions: immediate gain and risk of penalties (
Figure 1). The accumulated penalties were larger than the accumulated gains in decks A and B (disadvantageous decks), and the accumulated penalties were smaller than the accumulated gains in decks C and D (advantageous decks).
Control Task
The control task was designed to replicate the decision-making task in all aspects except for decision making. The four decks used for the control task were equal in gains and losses to those in the active task. Therefore, the tasks were similar in sensorimotor demands and in exposure to gains and losses. For the control task, however, the participants did not decide from which deck they would select the cards. Rather, they were instructed to pick cards from the decks sequentially in the fixed order of A-B-C-D-A-B-C-D, etc.
PET
PET scans were performed on a Siemens ECAT EXACT HR+ scanner (Knoxville, Tenn.). This instrument records images of 63 contiguous transaxial slices, with a 15.5-cm field of view that covers the entire brain and a 4.6-mm transaxial resolution. A thermoplastic mask, custom-made for each subject, was used to minimize head movement. Counts of radioactivity were recorded for 1 minute, starting after the tracer had reached the brain (an average 45 seconds after bolus injection). Scans were reconstructed by using a Hann filter with a 0.5 cutoff frequency. As arterial blood was not sampled, absolute rates of rCBF were not determined.
Data Analysis
Demographic, cognitive, and subjective variables
Demographic and cognitive (net score) data are described by using means and standard deviations. The net score was the total number of cards (100 cards) taken from the advantageous decks (C+D) minus the total number of cards taken from the disadvantageous decks (A+B).
Brain imaging analysis
The Statistical Parametric Mapping program (SPM 99)
(24) was used for most of the processing and for the analysis of the PET data. To correct for head motion between scans, PET scans for each subject were coregistered by using the Automated Image Registration Program
(25) and the SPM 99 realignment module. Scans were then spatially normalized to a standard space corresponding to the Montreal Neurologic Institute brain template (Montreal Neurologic Institute space)
(25). Data were smoothed with a 10×10×10 mm Gaussian filter to reduce high-frequency noise and the effects of individual differences in gyral anatomy. A two-stage procedure was implemented for statistical analyses. In the first stage, PET scans from each participant were used to create an adjusted mean image representing the decision-making effect (the two active scans minus the two control scans). In the computation of these adjusted images, proportional scaling (regional signal/global signal) was used to control for within-session variations in the global signal. In the second stage, the single adjusted image from each participant was entered into a two-sample t test analysis or into a simple regression analysis by using a random-effects model
(26). Areas of activation were identified by comparison with a standard atlas
(27).
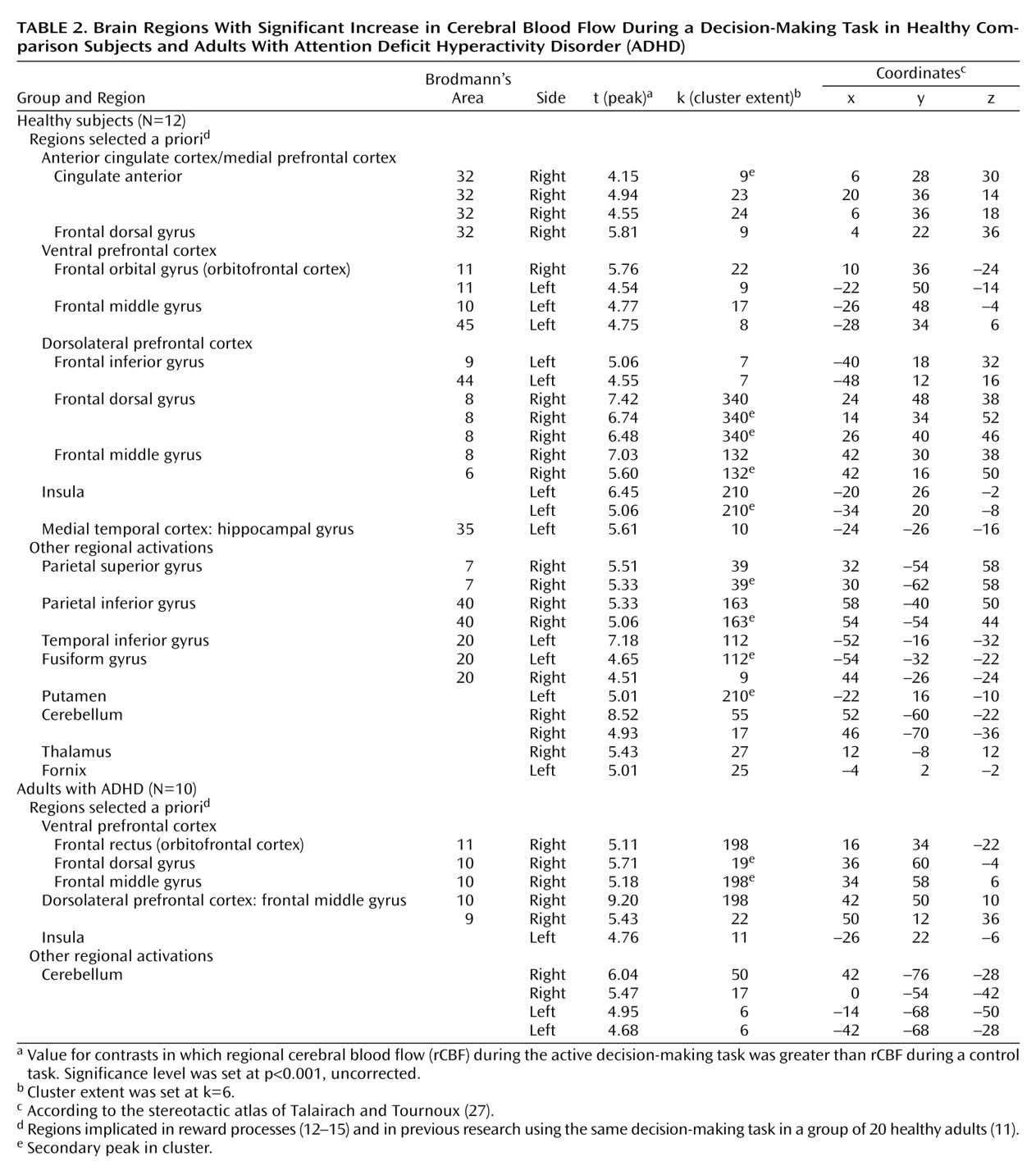
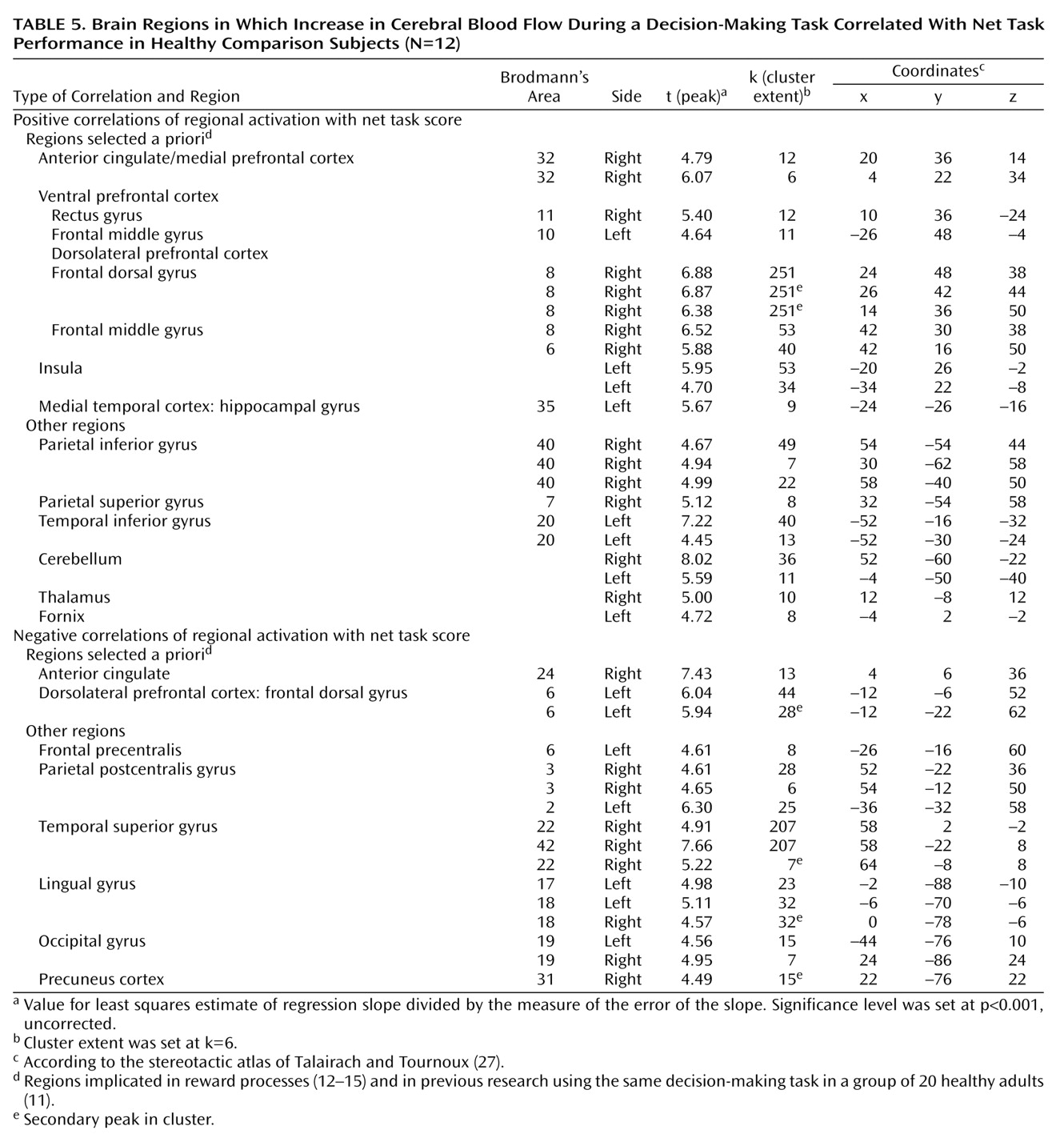
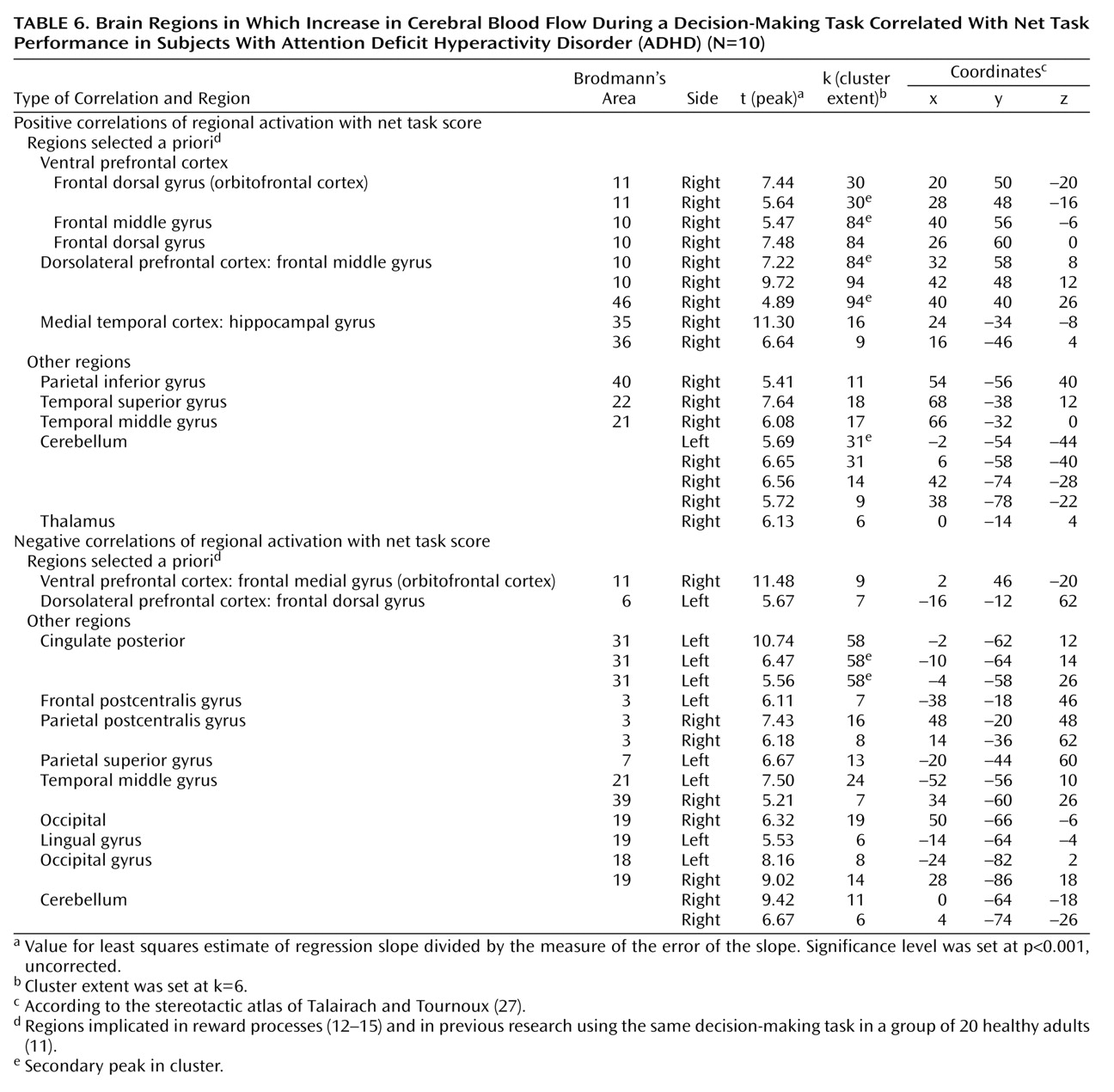
We focused our analysis on the regions that have been implicated in reward processes
(12–
15) and in our previous research in which a group of 20 healthy adults completed the same decision-making task
(11). These regions include the ventral and medial prefrontal cortex, anterior cingulate cortex, insular cortex, nucleus accumbens, amygdala, and hippocampus. The statistical threshold for these a priori regional analyses was set at p<0.001 (uncorrected) for height and six contiguous voxels for extent (i.e., 6 times 2×2×2 mm, or 2–3 times the spatial resolution of the scanner). Although the discussion centers almost exclusively on these regions, the tables present all loci of activations for the sake of completeness.
Discussion
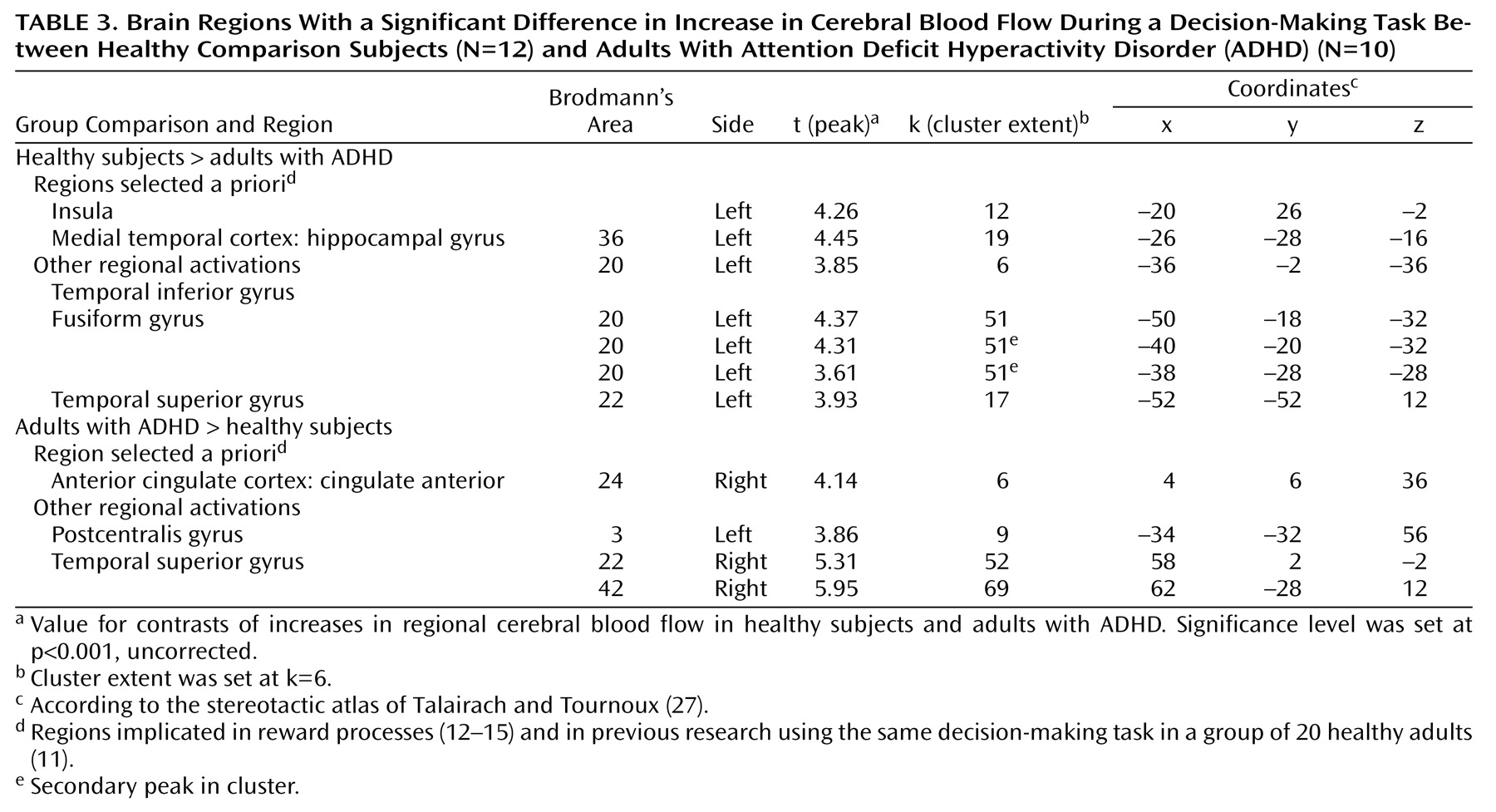
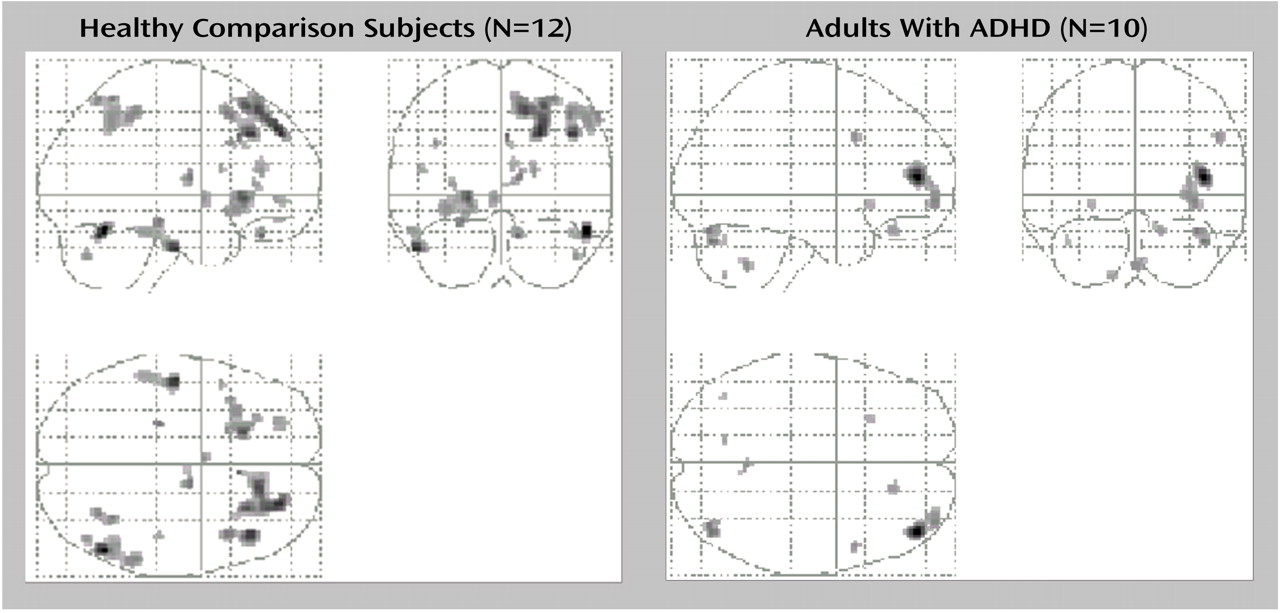
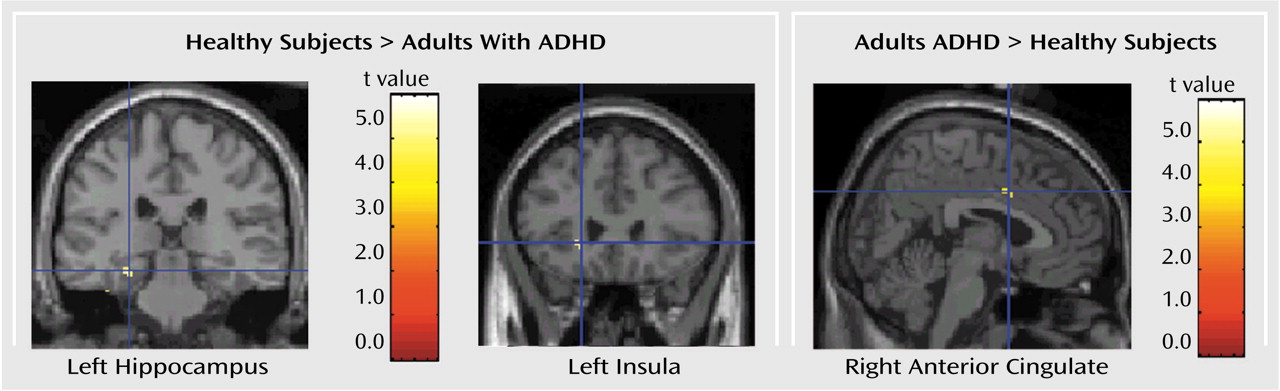
The neural circuits engaged in decision making differed in adults with ADHD compared to healthy adults. The decision-making task engaged the expected regions of the ventral prefrontal cortex, insula, and dorsolateral prefrontal cortex in both groups. However, activation was overall more extended in the comparison group than in the ADHD group (
Figure 2). Furthermore, in contrast to the comparison group, the ADHD group failed to activate other expected limbic areas, i.e., the hippocampus and anterior cingulate (Brodmann’s area 32). The functional significance of these findings can be understood in a number of ways on the basis of the various roles associated with these brain structures. The following discussion represents only one of several possible interpretations of the data. The key point of our proposed model is that the deficits in decision making observed in individuals with ADHD arise from the weaker engagement of brain structures involved in complex cognitive-emotional processes in favor of structures engaged in more primary processes, such as the coding of sensorimotor aspects of stimuli.
Although primarily associated with learning and memory function
(28), the hippocampus has been seen as playing a key role in several functions essential to decision making (for review see reference
29). These functions include encoding novelties of stimuli/situations
(30), processing spatial information
(31), and activating perceptual and emotional representations of past experience to influence output processes
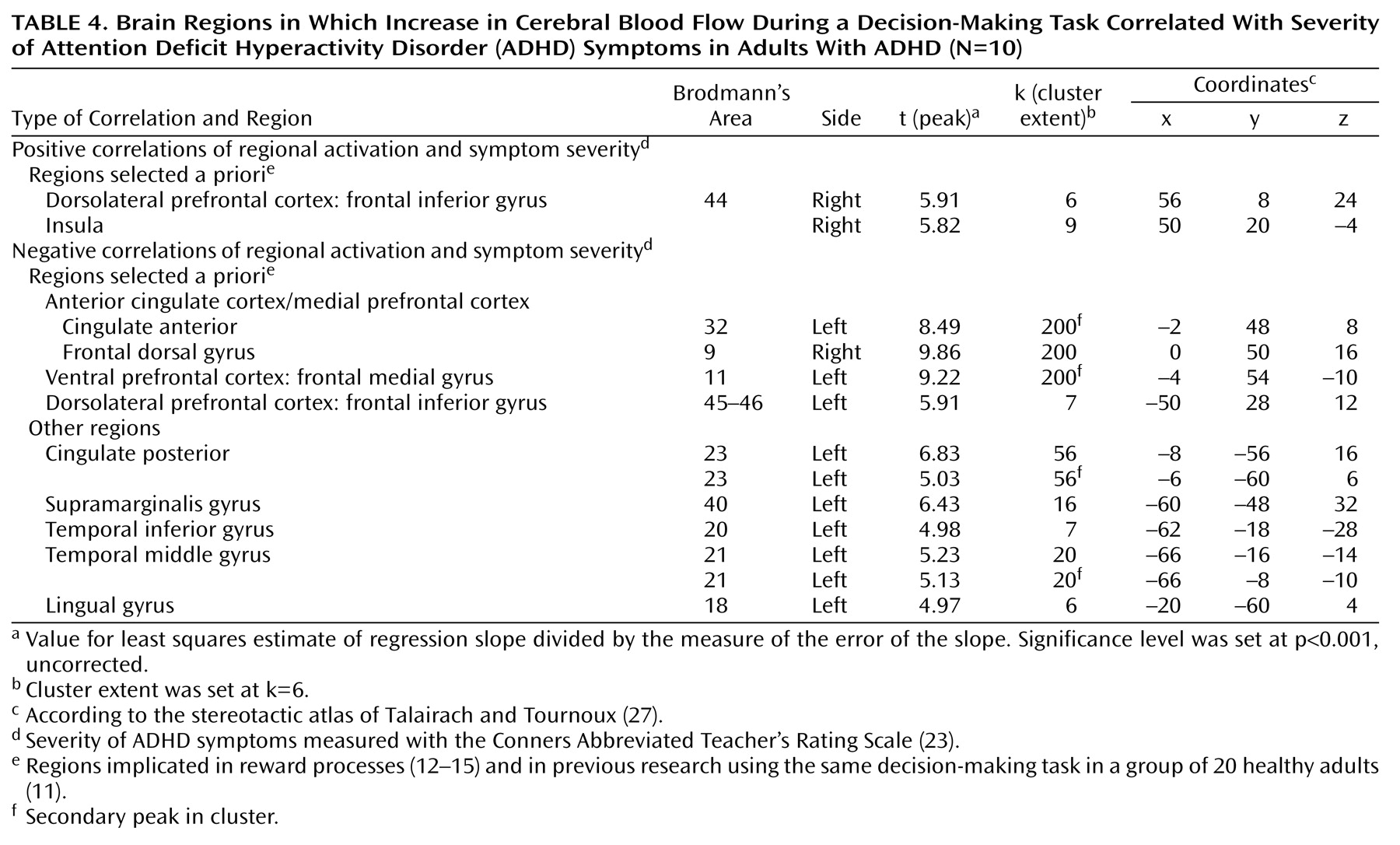
(32). Most relevant is the proposed role of the hippocampus in attentional monitoring of online processes
(33), which can subserve the continuous recording of conditions of novelty and uncertainty and contribute to the coding of the reinforcing property of stimuli. The positive correlation of performance scores with hippocampal activation in the comparison group suggests a direct role of the hippocampus in the decision-making process tested here. The deficit in hippocampal engagement in the ADHD group, manifested both by the absence of significant hippocampal activation while subjects perform the task and by its significantly greater activation in the comparison subjects than in the ADHD subjects, may explain in part the clinical observation of overall decreased efficacy of reinforcements in this disorder (for review see references
1,
2, and
9). Alternatively, the deficit in hippocampal recruitment may translate as defective memory of the incentive value of the stimuli.
Like the hippocampus, the anterior cingulate (Brodmann’s area 32) was activated in the comparison group and not in the ADHD group. This finding is consistent with a previous report of anterior cingulate dysfunction in ADHD
(34). It also supports the role of this region in attention that regulates cognitive and emotional/motivational processing
(35). On the basis of cytoarchitecture, anatomical connectivity, and function, the anterior cingulate comprises a number of specialized subdivisions, which subserve functions involved in cognitive, emotional, motor, nociceptive, and visuospatial processing
(36,
37). Two major subdivisions—a dorsal cognitive section and a rostroventral emotional section—have been isolated. The cingulate activation in the comparison subjects included two peaks situated at the juncture of these two subdivisions and one other peak in the dorsal cognitive territory, which has been shown to be involved in reward-based decision making
(35). A more caudal area of the right anterior cingulate (Brodmann’s area 24) was the only region significantly more activated in the ADHD subjects than in the comparison subjects. This activation may reflect a compensatory recruitment of the right dorsal caudal part of the anterior cingulate in ADHD for decision making, or it may reflect a primary faulty engagement of this region, which prevents the expected neural circuits from assuming their function. Supporting the latter hypothesis is the negative correlation of this exact regional activation with performance in the comparison group, suggesting that its activation may disrupt the process of decision making in healthy subjects.
The left insula is the other region that was significantly more activated in the comparison subjects than in the ADHD subjects. Greater activation in this region has also been shown to predict better performance in healthy subjects. The insula is a multimodal sensory integration region that contributes to the integration of sensory events with emotional responses
(38), and it has been shown to be recruited during negative mood states
(39). Its reduced involvement in decision making in ADHD subjects concurs with the relatively weaker autonomic response reported in ADHD youths in both nonspecific skin conductance responses
(40) and skin conductance responses in the context of reward paradigms
(41). Overall, this interpretation of findings is consistent with ADHD subjects’ having deficits in attributing motivational value to stimuli, resulting in lower motivation and less willingness to exert effort to attain goals, both of which characterize ADHD behavior.
The lack of a difference in performance on the decision-making task between ADHD and comparison subjects most likely reflects the small sample size. This task, however, has strong face validity, as it probes the specific cognitive processes, such as sensitivity and responses to contingent stimuli
(1), that are clinically altered in ADHD individuals
(2,
42). We recently found impaired performance on this task in a group of 33 adolescents with externalizing behavioral disorders, including ADHD, compared to 31 healthy adolescents
(43).
Finally, the activation maps associated with the task included a number of regions that were not discussed in this work because they were not part of our a priori hypotheses. Of interest, however, is the greater involvement of primary somatosensory areas (Brodmann’s area 3, 42) in the ADHD subjects than in the comparison subjects. This difference may indicate that individuals with ADHD use primary processes more readily than complex processes (involving the hippocampus and insula) in decision making, preventing effective filtering of environmental stimuli for adaptive responses.
Two caveats should be mentioned. First, the functional abnormalities might be confounded with structural differences between the ADHD subjects and healthy subjects. Previous studies have reported size differences in various brain regions
(44,
45). Second, the present findings are applicable only to adults with ADHD. Studies of children are needed for generalization of the findings to this age group.
Overall, the findings support the idea that deficits in reward systems may play an important role in the pathology of ADHD. A better understanding of the nature of these deficits can be applied to refine treatment strategies.